Abstract
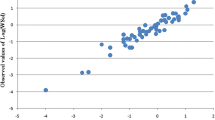
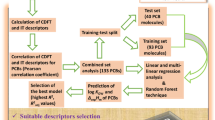
Fullerenes are sparingly soluble in many solvents. The dependence of fullerene’s solubility on molecular structure of the solvent must be understood in order to manage efficiently this class of compounds. To find such dependency ab initio quantum-chemical calculations in combination with quantitative structure–property relationship (QSPR) tool were used to model the solubility of fullerene C60 in 122 organic solvents. A genetic algorithm and multiple regression analysis (GA-MLRA) were applied to generate correlation models. The best performance is accomplished by the four-variable MLRA model with prediction coefficient r 2test = 0.903. This study reveals a correlation of highest occupied molecular orbital energy (HOMO), certain heteroatom fragments, and geometrical parameters with solubility. Several other important parameters of solvents that affect the C60 solubility have been also evaluated by the QSPR analysis. The employed GA-MLRA approach enhanced by application of quantum-chemical calculations yields reliable results, allowing one to build simple, interpretable models that can be used for predictions of C60 solubility in various organic solvents.




Similar content being viewed by others
References
Abraham MH, Green CE, Acree WE (2000) Correlation and prediction of the solubility of Buckminsterfullerene in organic solvents; estimation of some physicochemical properties. J Chem Soc Perkin Trans 2:281–286
Antipin IS, Arslanov NA, Palyulin VA, Konovalov AI, Zefirov NS (1991) Solvation topological index. Topological description of dispersion interaction (in Russian). Dokl Akad Nauk SSSR 316:925–927 (Chem Abstr 115, 91390)
Balaban AT (1983) Topological indexes based on topological distances in molecular graphs. Pure Appl Chem 55:199–206
Beck MT, Mandi G (1997) Solubility of C60. Fuller Sci Technol 5:291–310
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652
Cook SM, Aker WG, Rasulev BF, Hwang H-M, Leszczynski J, Jenkins JJ, Shockley V (2010) Choosing safe dispersing media for C60 fullerenes by using cytotoxicity tests on the bacterium Escherichia coli. J Hazard Mater 176:367–373
Davis L (1991) Handbook of genetic algorithms. Van Nostrand Reinhold, New York, USA
de Oliveira DB, Gaudio AC (2001) BuildQSAR: a new computer program for QSAR analysis. Quant Struct Act Relat 19:599–601
Devillers J (1996) Genetic algorithms in molecular modeling. Academic Press Ltd, London
Duchowicz PR, Talevi A, Bruno-Blanch LE, Castro EA (2008) New QSPR study for the prediction of aqueous solubility of drug-like compounds. Bioorg Med Chem 16:7944–7955
Estrada E, Gutman I (1996) A topological index based on distances of edges of molecular graphs. J Chem Inf Comp Sci 36:850–853
Foresman JB, Frisch A (2000) Exploring chemistry with electronic structure methods. Gaussian, Inc., Pittsburgh
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR et al (2004) Gaussian 03, Revision C.02. Gaussian, Inc., Wallingford
Hansen CM, Smith AL (2004) Using Hansen solubility parameters to correlate solubility of C60 fullerene in organic solvents and in polymers. Carbon 42:1591–1597
Hehre WJ, Radom L, Schleyer P, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Herbst MH, Dias GHM, Magalhaes JG, Torres RB, Volpe PLO (2005) Enthalpy of solution of fullerene [60] in some aromatic solvents. J Mol Liq 118:9–13
Katritzky AR, Lobanov VS, Karelson M (1994) Comprehensive descriptors for structural and statistical analysis. Version 2.0. Semichem, Inc., Gainesville (reference manual)
Katritzky AR, Lobanov VS, Karelson M (1995) Chem Soc Rev 24:279–287
Kiss IZ, Mandi G, Beck MT (2000) Artificial neural network approach to predict the solubility of C60 in various solvents. J Phys Chem A 104:8081–8088
Korobov MV, Smith AL (2000) Solubility of the fullerenes. In: Kadish KM, Ruoff RS (eds) Fullerenes: chemistry, physics, and technology. John Wiley and Sons Inc, New York, pp 53–90
Liu H, Yao X, Zhang R, Liu M, Hu Z, Fan B (2005) Accurate quantitative structure-property relationship model to predict the solubility of C60 in various solvents based on a novel approach using a least-squares support vector machine. J Phys Chem B 109:20565–20571
Marcus Y (1997) Solubilities of buckminsterfullerene and sulfur hexafluoride in various solvents. J Phys Chem 101:8617–8623
Marcus Y, Smith AL, Korobov MV, Mirakyan NV, Avramenko NV, Stukalin EB (2001) Solubility of C60 fullerene. J Phys Chem B 105:2499–2506
Mohar B (1989) Laplacian matrices of graphs. In: Graovac A (ed) MATH/CHEM/COMP 1988. Studies in physical and theoretical chemistry, vol 63, Elsevier, Amsterdam, pp 1–8
Randic M, Krilov G (1999) On a characterization of the folding of proteins. Int J Quantum Chem 75:1017–1026
Randic M, Kleiner AF, De Alba LM (1994) Distance/distance matrices. J Chem Inf Comp Sci 34:277–286
Ruoff RS, Tse DS, Malhotra R, Lorents DC (1993) Solubility of fullerene (C60) in a variety of solvents. J Phys Chem 97:3379–3383
Simon J (1987) Molecular graphs as topological objects in space. J Comp Chem 8:718–726
Sivaraman N, Dhamodaran R, Kaliappan I, Srinivasan TG, Rao PRV, Mathews CK (1992) Solubility of C60 in organic solvents. J Org Chem 57:6077–6079
Sivaraman N, Dhamodaran R, Kaliappan I, Srinivasan TG, Rao PRV, Mathews CK (1994) Solubility of C60 and C70 in organic solvents. In: Kadish KM, Ruoff RS (eds) Recent advances in the chemistry and physics of fullerenes and related materials. The Electrochemical Society, Pennington, pp 156–165
Smith AL, Wilson LY, Famini GR (1996) A quantitative structure-property relationship study of C60 solubility. In: Recent advances in the chemistry and physics of fullerenes and related materials, vol 3. Proceedings of Electrochemical Society, Philadelphia, pp 53–62
Stukalin EB, Korobov MV, Avramenko NV (2003) Solvation free energies of the fullerenes C60 and C70 in the framework of polarizable continuum model. J Phys Chem B 107:9692–9700
Todeschini R, Consonni V (2000) Handbook of molecular descriptors. Wiley-VCH, Weinheim
Todeschini R, Consonni V (2003) DRAGON software for the calculation of molecular descriptors Version 3.0
Toropov AA, Leszczynska D, Leszczynski J (2007a) QSPR study on solubility of fullerene C60 in organic solvents using optimal descriptors calculated with SMILES. Chem Phys Lett 441:119–122
Toropov AA, Leszczynska D, Leszczynski J (2007b) Predicting water solubility and octanol water partition coefficient for carbon nanotubes based on the chiral vector. Comput Biol Chem 31:127–128
Toropov AA, Rasulev BF, Leszczynska D, Leszczynski J (2007c) Additive SMILES based optimal descriptors: QSPR modeling of fullerene C60 solubility in organic solvents. Chem Phys Lett 444:209–214
Toropov AA, Rasulev BF, Leszczynska D, Leszczynski J (2008) Multiplicative SMILES-based optimal descriptors: QSPR modeling of fullerene C60 solubility in organic solvents. Chem Phys Lett 457:332–336
Toropov AA, Toropova AP, Benfenati E, Leszczynska D, Leszczynski J (2009) Additive InChI-based optimal descriptors: QSPR modeling of fullerene C60 solubility in organic solvents. J Math Chem 46(4):1232–1251
Vanin AA, Piotrovskaya EM, Piotrovsky LB (2008) Investigation of fullerene solutions by molecular dynamics method. Fuller Nanotub Car N 16(5–6):555–562
Acknowledgments
The authors would like to thank for support the National Science Foundation for the “Interdisciplinary Center for Nanotoxicity” support—NSF HRD #0833178; for NSF EPSCoR Grant no. 362492-190200-01\NSFEPS-0903787 and Department of Defense through the U. S. Army Engineer Research and Development Center (Vicksburg, MS) for the grant “Development of Predictive Techniques for Modeling Properties of NanoMaterials Using New OSPR/QSAR Approach Based on Optimal NanoDescriptors”—Contract #W912HZ-06-C-0061. The authors are grateful to the Mississippi Center for Supercomputing Research (MCSR) for providing state-of-the-arts high performance computing facilities and excellent services for supporting this research.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Petrova, T., Rasulev, B.F., Toropov, A.A. et al. Improved model for fullerene C60 solubility in organic solvents based on quantum-chemical and topological descriptors. J Nanopart Res 13, 3235–3247 (2011). https://doi.org/10.1007/s11051-011-0238-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-011-0238-x