Abstract
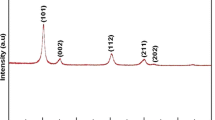
Nanocrystalline zirconium oxide was synthesized by hydrothermal treatment of ZrO(NO3)2 and ZrOCl2 aqueous solutions at different temperatures and time in presence of hydrogen peroxide. Hydrothermal treatment of zirconium salts (0.25 and 0.50 mol L−1) produced nanocrystalline monoclinic ZrO2 powders with narrow size distribution, which were formed by the attachment of the smaller particles with crystallites size of 3.5 nm, estimated by means of the Scherrer’s equation and confirmed by transmission electronic microscopy. Typical monoclinic zirconium oxide X-ray powder diffraction patterns and Raman spectra were obtained for all the crystalline powders. It was observed that the crystallization depends strongly on the temperature, resulting in amorphous material when the synthesis was realized at 100 °C, and crystalline with monoclinic phase when synthesized at 110 °C, independently of the salt used. Zirconium oxide colloidal nanoparticles were formed only at hydrothermal treatments longer than 24 h. The stability of the colloids was successfully characterized of zeta potential, showing an initial value of + 59.2 mV in acid media and isoelectric point at pH = 5.2, in good agreement with previous studies.





Similar content being viewed by others
References
Chen F, Zhong Z, Gan GJ (2005) Hydrothermal synthesis of anisotropic monoclinic zirconia particles in the presence of 1, 12-Diaminododecane. J Am Ceram Soc 88:1985–1987
Deville S, Guénin G, Chevalier J (2004a) Martensitic transformation in zirconia Part I. Nanometer scale prediction and measurement of transformation induced relief. Acta Mater 52:5697–5707
Deville S, Guénin G, Chevalier J (2004b) Martensitic transformation in zirconia Part II. Martensite growth. Acta Mater 52:5709–5721
Di Maggio R, Fambri L, Mustarelli P, Campostrini R (2003) Physico-chemical characterization of hybrid polymers obtained by 2-hydroxyethyl(methacrylate) and alkoxides of zirconium. Polymer 44:7311
El Ghzaoui A (1999) Measurement of the diffuse double-layer forces between zirconia and α-alumina. J Appl Phys 86:5894–5897
Gao Y, Masuda Y, Ohta H, Koumoto K (2004) Room-temperature preparation of ZrO2 precursor thin film in an aqueous peroxozirconium-complex solution. Chem Mater 16:2615–2622
Guo GY, Chen YL (2005) A nearly pure monoclinic nanocrystalline zirconia. J Solid State Chem 178:1675
Hu MZC, Harris MT, Byers CH (1998) Nucleation and growth for synthesis of nanometric zirconia particles by forced hydrolysis. J Colloid Interface Sci 198:87
Kelly PM, Francis Rose LR (2002) The martensitic transformation in ceramics—its role transformation toughening. Prog Mater Sci 47:463–557
Kolen’koa Y, Maximova VD, Burukhinb AA, Muhanovb VA, Churagulovb BR (2003) Synthesis of ZrO2 and TiO2 nanocrystalline powders by hydrothermal process. Mater Sci Eng C 23:1033
Kosmambetova GR, Strizhak PE, Moroz EM, Konstantinova TE, Gural’skii AV, Kol’ko VP, Gritsenko VI, Danilenko IA, Gorban OA (2007) Influence of the conditions of manufacture of nanomeric zirconium dioxide, stabilized with yttrium oxide, on its catalytic properties in the oxidation of CO. Theor Exp Chem 43:102
Kumari L, Li W, Wang D (2008) Monoclinic zirconium oxide nanostructures synthesized by a hydrothermal route. Nanotechnology 19:195602–195609
Li M, Feng Z, Xiong G, Ying P, Xin Q, Li C (2001) Phase transformation in the surface region of zirconia detected by UV Raman spectroscopy. J Phys Chem B 105:8107–8111
Millesime L, Amiel C, Michel F, Chaufer B (1996) Surface modification of zirconium oxide particles with charged polymers. Langmuir 12:3377–3382
Mottet B, Pichavant M, Bény JM, Alary JA (1992) Morphology of zirconia synthesized hydrothermally from zirconium oxychloride. J Am Ceram Soc 75:2515–2519
Purohit RD, Saha SA, Tyagi K (2006) Combustion synthesis of nanocrystalline ZrO2 powder: XRD, Raman spectroscopy and TEM studies. Mater Sci Eng B 130:57–60
Rashad MM, Baioumy HM (2008) Effect of thermal treatment on the crystal structure and morphology of zirconia nanopowders produced by three different routes. J Mater Process Technol 195:178
Shen P, Lee WH (2001) (111)-specific coalescence twinning and martensitic transformation of tetragonal ZrO2 condensates. Nano Lett 1:707–711
Stadelmann PA (1987) EMS—a software package for electron diffraction analysis and HREM image simulation in materials science. Ultramicroscopy 21:131–145
Valmalette JC, Isa M (2002) Size effects on the stabilization of ultrafine zirconia nanoparticles. Chem Mater 14:5098
Wang H, Li G, Xue Y, Li L (2007) Hydrated surface structure and its impacts on the stabilization of t-ZrO2. J Solid State Chem 180:2790–2797
Xie S, Iglesia E, Bell AT (2000) Water-assisted tetragonal-to-monoclinic phase transformation of ZrO2 at low temperatures. Chem Mater 12:2442–2447
Yashima M, Tsunekawa S (2006) Structures and the oxygen deficiency of tetragonal and monoclinic zirconium oxide nanoparticles. Acta Cryst B62:161
Zhao S, Ma F, Song Z, Xu K (2008) Thickness-dependent structural and optical properties of sputter deposited ZrO2 films. Opt Mater 30:910
Acknowledgments
This work was supported by the Brazilian agencies FAPESP through the Project 2007/58.991-7, CNPq, CAPES, and the CMDMC/Cepid/INCT.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arantes, T.M., Mambrini, G.P., Stroppa, D.G. et al. Stable colloidal suspensions of nanostructured zirconium oxide synthesized by hydrothermal process. J Nanopart Res 12, 3105–3110 (2010). https://doi.org/10.1007/s11051-010-9906-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-010-9906-5