Abstract
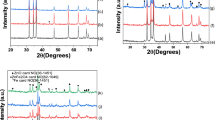
Zn/Fe oxide compound powders were obtained by the hydrothermal method using ferric nitrate Fe(NO3)3·9H2O and zinc nitrate Zn(NO3)2·6H2O at 200 °C and different precursor molar ratios x = Fe3+/Zn2+ equal to 2.8/0.2, 2.5/0.5, 1.8/1.2 and 1.5/1.5. The samples were characterized by X-ray diffraction (XRD) and scanning electron microscopy/energy dispersive X-ray analysis (SEM–EDAX). Room temperature measurements of the frequency dependence of the complex magnetic permeability and complex dielectric permittivity, over the frequency range from 0.1 to 6 GHz, were performed. For precursor molar ratios x = 2.8/0.2, x = 1.8/1.2 and x = 1.5/1.5 the obtained samples showed a ferromagnetic-like resonance behaviour. This behaviour was assigned to the prevalent compounds in the obtained samples, Fe2O3 (for x = 2.8/0.2) and ZnFe2O4 (for x = 1.8/1.2 and x = 1.5/1.5). Based on the magnetic and dielectric measurements, the microwave absorbent properties of the four samples were analysed, and the sample containing mostly of ZnFe2O4 (for x = 1.8/1.2) was found to be the best electromagnetic absorber in the frequency range 1.36–6 GHz.








Similar content being viewed by others
References
Cheng YL, Dai JM, Wu DJ, Sun YP (2010) Electromagnetic and microwave absorption properties of carbonyl iron/La0.6Sr0.4MnO3 composites. J Magn Magn Mater 322(1):97–101
Chu X, Liu X, Meng G (1999) Preparation and gas sensitivity properties of ZnFe2O4 semiconductors. Sens Actuators B 55(1):19–22
Collin RE (1966) Foundation for microwave engineering. McGraw Hill, New York
Duan Y, Ma H, Li X, Liu S, Ji Z (2010) The microwave electromagnetic characteristics of manganese dioxide with different crystallographic structures. Physica B 405(7):1826–1831
Gao D, Zhang Z, Fu J, Xu Y, Qi J, Xue D (2009) Room temperature ferromagnetism of pure ZnO nanoparticles. J Appl Phys 105(11):113928. doi:10.1063/1.3143103
Ghasemi A, Morisako A (2008) Static and high frequency magnetic properties of Mn–Co–Zr substituted Ba-ferrite. J Alloys Compd 456(1–2):485–491
El-Shobaky GA, Turky AM, Mostafa NY, Mohamed SK (2010) Effect of preparation conditions on physicochemical, surface and catalytic properties of cobalt ferrite prepared by coprecipitation. J Alloys Compd 493(1–2):415–422
Fannin PC, Vincent D, Noyel G (1999) On the measurement of the complex susceptibility and permittivity of magnetic fluids by means of two different measurement techniques. J Magn Magn Mater 201(1–3):116–118
Fannin PC, Malaescu I, Marin CN, Stefu N (2009) Microwave propagation parameters in magnetic fluids. Eur Phys J E 29(3):299–303
Franco A Jr, Celma de Oliveira LE, Novak MA, Wells PR Jr (2007) Synthesis of nanoparticles of CoxFe(3–x)O4 by combustion reaction method. J Magn Magn Mater 308(2):198–202
Hofmann M, Campbell SJ, Ehrhardt H, Feyerherm R (2004) The magnetic behaviour of nanostructured zinc ferrite. J Mater Sci 39(16–17):5057–5065
Jeyadevan B, Tohji K, Nakatsuka K (1994) Structure analysis of coprecipitated ZnFe2O4 by extended X-ray absorption fine structure. J Appl Phys 76(10):6325. doi:10.1063/1.358255
Jin H-b, Cao M-s, Zhou W, Agathopoulos S (2010) Microwave synthesis of Al-doped SiC powders and study of their dielectric properties. Mater Res Bull 45(2):247–250
Kamiyama T, Haneda K, Sato T, Ikeda S, Asano H (1992) Cation distribution in ZnFe2O4 fine particles studied by neutron powder diffraction. Solid State Commun 81(7):563–566
Liu J, Lu G, He H, Tan H, Xu T, Xu K (1996) Studies on photocatalytic activity of zinc ferrite catalysts synthesized by shock waves. Mater Res Bull 31(9):1049–1056
Morin FJ (1950) Magnetic susceptibility of αFe2O3 and αFe2O3 with added titanium. Phys Rev 78(6):819–820
Owens FJ (2009) Ferromagnetic resonance observation of a phase transition in magnetic field-aligned Fe2O3 nanoparticles. J Magn Magn Mater 321(15):2386–2391
Qiu J, Wang C, Gu M (2004) Photocatalytic properties and optical absorption of zinc ferrite nanometer films. Mater Sci Eng B 112(1):1–4
Sharma Prashant K, Dutta Ranu K, Pandey Avinash C, Samar Layek, Verma HC (2009) Effect of iron doping concentration on magnetic properties of ZnO nanoparticles. J Magn Magn Mater 321(17):2587–2591
Sunny V, Kurian P, Mohanan P, Joy PA, Anantharaman MR (2010) A flexible microwave absorber based on nickel ferrite nanocomposite. J Alloys Compd 489(1):297–303
Tanaka K, Makita M, Shimizugawa Y, Hirao K, Soga N (1998) Structure and high magnetization of rapid quenched zinc ferrite. J Phys Chem Solids 59(9):1611–1618
Toledo-Antonio JA, Nava N, Martínez M, Bokhimi X (2002) Correlation between the magnetism of non-stoichiometric zinc ferrites and their catalytic activity for oxidative dehydrogenation of 1-butene. Appl Catal A 234(1–2):137–144
Tsukada M, Abe K, Yonemochi Y, Ameyama A, Kamiya H, Kambara S, Moritomi H, Uehara T (2008) Dry gas cleaning in coal gasification systems for fuel cells using composite sorbents. Powder Technol 180(1–2):232–238
Valenzuela MA, Bosch P, Jiménez-Becerrill J, Quiroz O, Páez AI (2002) Preparation, characterization and photocatalytic activity of ZnO, Fe2O3 and ZnFe2O4. J Photochem Photobiol A 148(1–3):177–182
Vonsovski SV (1974) Magnetism. Wiley, New York
Wang L, Song J, Zhang Q, Huang X, Xu N (2009) The microwave magnetic performance of Sm3+ doped BaCo2Fe16O27. J Alloys Compd 481(1–2):863–866
Wesselinowa JM (2010) Size and anisotropy effects on magnetic properties of antiferromagnetic nanoparticles. J Magn Magn Mater 322(2):234–237
Xie G, Yuan L, Wang P, Zhang B, Lin P, Lu H (2010) GHz microwave properties of melt spun Fe–Si alloys. J Non-Cryst Solids 356(2):83–86
Yuan Z-H, Zhang L-D (2001) Synthesis, characterization and photocatalytic activity of ZnFe2O4/TiO2 nanocomposite. J Mater Chem 11(4):1265. doi:10.1039/b006994i
Zhang W-H, Zhang W-D, Zhou J-F (2010) Solvent thermal synthesis and gas-sensing properties of Fe-doped ZnO. J Mater Sci 45(1):209–215
Zhao L, Zhang H, Xing Y, Song S, Yu S, Shi W, Guo X, Yang J, Lei Y, Cao F (2008) Studies on the magnetism of cobalt ferrite nanocrystals synthesized by hydrothermal method. J Solid State Chem 181(2):245–252
Zhao D-L, Lv Q, Shen Z-M (2009) Fabrication and microwave absorbing properties of Ni–Zn spinel ferrites. J Alloys Compd 480(2):634–638
Zhu CL, Chen YJ, Wang RX, Wang LJ, Cao MS, Shi XL (2009) Synthesis and enhanced ethanol sensing properties of α-Fe2O3/ZnO heteronanostructures. Sens Actuators B 140(1):185–189
Zysler RD, Vasquez Mansilla M, Fiorani D (2004) Surface effects in α-Fe2O3 nanoparticles. Eur Phys J B 41(2):171–175
Acknowledgements
P. C. Fannin acknowledges ESA for part funding of this study, whilst the other authors gratefully acknowledge partial financial support from CNMP grant no. 1-32155/2008. Acknowledgements are also due to A. Bucur for the support in performing the XRD measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fannin, P.C., Marin, C.N., Malaescu, I. et al. Effect of the concentration of precursors on the microwave absorbent properties of Zn/Fe oxide nanopowders. J Nanopart Res 13, 311–319 (2011). https://doi.org/10.1007/s11051-010-0032-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-010-0032-1