Abstract
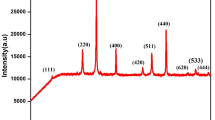
Polycrystalline magnetite hollow spheres with diameter of about 200 nm and shell thickness of 30–60 nm were prepared via a facile solution route. For the reaction, ethylene glycol (EG) served as the reducing agent and soldium acetate played the role of precipitator. In addition, polyvinylpyrrolidone (PVP) served as a surface stabilizer. The morphologies and structures were characterized by scanning electron microscopy, transmission electron microscopy and X-ray diffraction. The intermediate products at different stages were also studied to shed light on the evolution of phase formation. It revealed that the hollow structure formed via self-assembly of nanocrystallites (about 15 nm) using sodium acetate as mild precipitator. Evidences further pointed out that the Ostwald ripening process well explained the growth mechanism of the hollow structure. Magnetization measurements showed that the coercivity of magnetite hollow spheres at low temperature is about 200 Oe and the saturation magnetization is about 83 emu g−1, roughly 85% that of the bulk phase, close to the value of its solid counterpart. In addition, a freezing transition was observed at 25 K.









Similar content being viewed by others
References
Aldea N, Turcu R, Nan A, Craciunescu I, Pana O, Yaning X, Wu Z, Bica D, Vekas L, Matei F (2009) Investigation of nanostructured Fe3O4 polypyrrole core-shell composites by X-ray absorbtion spectroscopy and X-ray diffraction using synchrotron radiation. J Nanopart Res 11(6):1429–1439
Butter K, Philipse AP, Vroege GJ (2002) Synthesis and properties of iron ferrofluids. J Magn Magn Mater 252:1–3
Cong YH, Wang GL, Xiong MH, Huang Y, Hong ZF, Wang DL, Li JJ, Li LB (2008) A facile interfacial reaction route to prepare magnetic hollow spheres with tunable shell thickness. Langmuir 24(13):6624–6629
Dadarlat D, Neamtu C, Streza M, Turcu R, Craciunescu I, Bica D, Vekas L (2008) High accuracy photopyroelectric investigation of dynamic thermal parameters of Fe3O4 and CoFe2O4 magnetic nanofluids. J Nanopart Res 10(8):1329–1336
Deng Y, Qi D, Deng C, Zhang X, Zhao D (2008) Superparamagnetic high-magnetization microspheres with an Fe3O4@SiO2 core and perpendicularly aligned mesoporous SiO2 shell for removal of microcystins. J Am Chem Soc 130(1):28–29
Doyle PS, Bibette J, Bancaud A, Viovy JL (2002) Self-assembled magnetic matrices for DNA separation chips. Science 295(5563):2237
Franger S, Berthet P, Dragos O, Baddour-Hadjean R, Bonville P, Berthon J (2007) Large influence of the synthesis conditions on the physico-chemical properties of nanostructured Fe3O4. J Nanopart Res 9(3):389–402
Ge JP, Hu YP, Zhang TR, Huynh T, Yin YD (2008) Self-assembly and field-responsive optical diffractions of superparamagnetic colloids. Langmuir 24(7):3671–3680
Guo L, Liang F, Wen XG, Yang SH, He L, Zheng WZ, Chen CP, Zhong QP (2007) Uniform magnetic chains of hollow cobalt mesospheres from one-pot synthesis and their assembly in solution. Adv Funct Mater 17(3):425–430
Hong RY, Pan TT, Li HZ (2006) Microwave synthesis of magnetic Fe3O4 nanoparticles used as a precursor of nanocomposites and ferrofluids. J Magn Magn Mater 303:60–68
Huang J, Chen WM, Zhao W, Li YQ, Li XG, Chen CP (2009) One-dimensional chainlike arrays of Fe3O4 hollow nanospheres synthesized by aging iron nanoparticles in aqueous solution. J Phys Chem C 113(28):12067–12071
Hung Y, Hsiao JK, Yao M, Chung TH, Lin YS, Wu SH, Hsu SC, Liu HM, Mou CY, Yang CS, Huang DM, Chen YC (2007) Bifunctional magnetic silica nanoparticles for highly efficient human stem cell labeling. Nano Lett 7(1):149–154
Jia BP, Gao L (2008) Morphological transformation of Fe3O4 spherical aggregates from solid to hollow and their self-assembly under an external magnetic field. J Phys Chem C 112(3):666–671
Karmakar S, Chakravorty A (1996) Carboxyl-bonded low-spin iron(III) chemistry of a family of coordination type cis-FeN4O2. Inorg Chem 35(7):1935–1939
Kovalenko MV, Bodnarchuk MI, Lechner RT, Hesser G, Schaffler F, Heiss W (2007) Fatty acid salts as stabilizers in size- and shape-controlled nanocrystal synthesis: the case of inverse spinel iron oxide. J Am Chem Soc 129(20):6352–6353
Li XH, Zhang DH, Chen JS (2006) Synthesis of amphiphilic superparamagnetic ferrite/block copolymer hollow submicrospheres. J Am Chem Soc 128(26):8382–8383
Ling F, Lei J, Mai ZH, Zhu DB (2004) Polymer-controlled synthesis of Fe3O4 single-crystal nanorods. J Colloid Interf Sci 278(2):372–375
Liu SH, Xing RM, Lu F, Rana RK, Zhu JJ (2009) One-pot template-free fabrication of hollow magnetite nanospheres and their application as potential drug carriers. J Phys Chem C 113(50):21042–21047
Lou XW, Wang Y, Yuan CL, Lee JY, Archer LA (2006) Template-free synthesis of SnO2 hollow nanostructures with high lithium storage capacity. Adv Mater 18(17):2325–2329
Oliveira LCA, Petkowicz DI, Smaniotto A, Pergher SBC (2004) Magnetic zeolites: a new adsorbent for removal of metallic contaminants from water. Water Res 38(17):3699–3704
Ostwald W (1900) On the assumed isomerism of red and yellow mercury oxide and the surface-tension of solid bodies. Z Phys Chem-Stoch Ve 34(4):495–503
Ottaviano L, Kwoka M, Bisti F, Parisse P, Grossi V, Santucci S, Szuber J (2009) Local surface morphology and chemistry of SnO2 thin films deposited by rheotaxial growth and thermal oxidation method for gas sensor application. Thin Solid Films 517(22):6161–6169
Puntes VF, Krishnan KM, Alivisatos AP (2001) Colloidal nanocrystal shape and size control: the case of cobalt. Science 291(5511):2115–2117
Raj K, Moskowit B, Casciari R (1995) Advances in ferrofluid technology. J Magn Magn Mater 149:174–180
Sun YG, Xia YN (2002) Shape-controlled synthesis of gold and silver nanoparticles. Science 298(5601):2176–2179
Teo JJ, Chang Y, Zeng HC (2006) Fabrications of hollow nanocubes of Cu2O and Cu via reductive self-assembly of CuO nanocrystals. Langmuir 22(17):7369–7377
Wang H, Zhu T, Zhao K, Wang WN, Wang CS, Wang YJ, Zhan WS (2004) Surface spin glass and exchange bias in Fe3O4 nanoparticles compacted under high pressure. Phys Rev B 70(9):092409
Wang RM, Chen YF, Fu YY, Zhang H, Kisielowski C (2005) Bicrystalline hematite nanowires. J Phys Chem B 109(25):12245–12249
Wang N, Guo L, He L, Cao X, Chen CP, Wang RM, Yang SH (2007) Facile synthesis of monodisperse Mn3O4 tetragonal nanoparticles and their large-scale assembly into highly regular walls by a simple solution route. Small 3(4):606–610
Wang DB, Song CX, Zhao YH, Yang ML (2008a) Synthesis and characterization of monodisperse iron oxides microspheres. J Phys Chem C 112(33):12710–12715
Wang N, Cao X, Kong DS, Chen WM, Guo L, Chen CP (2008b) Nickel chains assembled by hollow microspheres and their magnetic properties. J Phys Chem C 112(17):6613–6619
Zeng H, Li J, Liu JP, Wang ZL, Sun SH (2002) Exchange-coupled nanocomposite magnets by nanoparticle self-assembly. Nature 420(6914):395–398
Zhang LY, Zhang YF (2009) Fabrication and magnetic properties of Fe3O4 nanowire arrays in different diameters. J Magn Magn Mater 321(5):L15–L20
Zhang WM, Wu XL, Hu JS, Guo YG, Wan LJ (2008) Carbon coated Fe3O4 nanospindles as a superior anode material for lithium-ion batteries. Adv Funct Mater 18(24):3941–3946
Zhang GX, Liu YB, Zhang CF, Hu WQ, Xu WB, Li Z, Liang S, Cao JQ, Wang YX (2009) Aqueous immune magnetite nanoparticles for immunoassay. J Nanopart Res 11(2):441–448
Zhong LS, Hu JS, Liang HP, Cao AM, Song WG, Wan LJ (2006) Self-assembled 3D flowerlike iron oxide nanostructures and their application in water treatment. Adv Mater 18(18):2426
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 50671003, 50971011 and 10874006), Beijing Natural Science Foundation (No. 1102025), the National Basic Research Program of China (Nos. 2009CB939901 and 2010CB934601), the Program for New Century Excellent Talents in University (NCET-06-0175) and Research Fund for the Doctoral Program of Higher Education of China (20091102110038).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sun, Q., Ren, Z., Wang, R. et al. Magnetite hollow spheres: solution synthesis, phase formation and magnetic property. J Nanopart Res 13, 213–220 (2011). https://doi.org/10.1007/s11051-010-0020-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-010-0020-5