Abstract
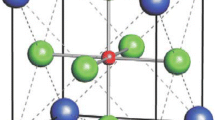
Crystalline (Ba x Sr1−x TiO3) (x = 0, 0.5 0.8 and 1) nanosized powders is synthesized by an ambient pressure and low temperature sol process. The influence of Ba/Sr ratio, temperature, pH and reflux time on the phase formation and morphology of (Ba x Sr1−x TiO3) powders is assessed. Amorphous powders are obtained at temperatures lower than 90 °C and crystalline, stoichiometric and monophasic Ba x Sr1−x TiO3 (x = 0, 0.5, 0.8 and 1), powders are synthesized at 95 °C. Under identical preparation conditions, the powder particle size depends on the Ba/Sr ratio. High values of pH decrease the particle size. For reflux times lower than 2 h the particle size decreases with increasing of reflux time, reflecting the dominant role of the nucleation step. For reflux times higher than 2 h the growth step becomes dominant and the particle size increases with the increase of reflux time.














Similar content being viewed by others
References
Arya PR, Jha P, Ganguli AK (2003) Synthesis, characterization and dielectric properties of nanometer-sized barium strontium titanates prepared by the polymeric citrate precursor method. J Mater Chem 13:415–423
Battisha IK, Speghini A, Polizzi S, Agnoli F, Bettinelli M (2002) Molten chloride synthesis, structural characterisation and luminescence spectroscopy of ultrafine Eu3+-doped BaTiO3 and SrTiO3. Mater Lett 57:183–187
Beck C, Härtl W, Hempelmann R (1998) Size-controlled synthesis of nanocrystalline BaTiO3 by a sol-gel type hydrolysis in microemulsion-provided nanoreactors. J Mater Res 13(11):3174–3180
Byrappa K, Yoshimura M (2001) Handbook of hydrothermal technology: a technology for crystal growth and materials processing. Noyes Publications, New Jersey
Carvalho JC, Paiva-Santos CO, Zaghete MA, Oliveira CF, Varela JA, Longo E (1996) Phase analysis of seeded and doped PbMg1/3Nb2/3O3 prepared by organic solution of citrates. J Mater Res 11:1795–1799
Denton AR, Ashcroft NW (1991) Vegard’s law. Phys Rev A 43(6):3161–3164
European Patent Number 0150135A1 (1985) Dielectric barium strontium-titanate fine powder
German RM (1996) A measure of the number of particles in agglomerates. Int J Powder Metall 32(4):365–373
Gersten BL (1999) Influence of the A and B site cation species in the kinetics of hydrothermal synthesis of ABO3 perovskite-type materials. PhD dissertation, Rutgers University, Piscataway
Hennings D (1989) Review of chemical preparation routes for barium titanate. Br Ceram Proc 41:1–10
Jie G (2007) Sol-gel (BaxSr1-x)TiO3 thin films for microelectronic applications. Master dissertation, University of Aveiro
Kiss K, Magder J, Vukasovich MS, Lockhart RJ (1966) Ferroelectrics of ultrafine particle size: I—synthesis of titanate powders of ultrafine particle size. J Am Ceram Soc 49(6):291–295
Lencka MM, Riman RE (1993) Thermodynamic modelling of hydrothermal synthesis of ceramic powders. Chem Mater 5:61–70
Lencka MM, Riman RE (1994) Hydrothermal synthesis of perovskite materials: thermodynamic modelling and experimental verification. Ferroelectrics 151:159–164
Lenggoro IW, Panatarani C, Okuyama K (2004) One-step synthesis and photoluminescence of doped strontium titanate particles with controlled morphology. Mater Sci Eng B 113:60–66
Li W-J, Shi E-W, Fukuda T (2003) Particle size of powders under hydrothermal conditions. Cryst Res Technol 38(10):847–858
Li Z, Wu A, Vilarinho PM (2004) Perovskite phase stabilization of Pb(Zn1/3Ta2/3)O3 ceramics induced by PbTiO3 seeds. Chem Mater 16:717–723
Mao Y, Banerjee S, Wong SS (2003) Large-scale synthesis of single-crystalline perovskite nanostructures. J Am Chem Soc 125:15718–15719
Moriarty P (2001) Nanostructured materials. Rep Progr Phys 64:297–381
Niederberger M, Garnweitner G, Pinna N, Antonietti M (2004) Nonaqueous and halide-free route to crystalline BaTiO3, SrTiO3, and (Ba, Sr)TiO3 nanoparticles via a mechanism involving C–C bond formation. J Am Chem Soc 126:9120–9126
Qi JQ, Wan YW, Chen P, Li L, Chan WHL (2005) Direct large-scale synthesis of perovskite barium strontium titanate nano-particles from solutions. J Solid State Chem 178:279–284
Rout SK, Panigrahi S (2006) Mechanism of phase formation of BaTiO3-SrTiO3 solid solution through solid–oxide reaction. Indian J Pure Appl Phys 44:606–611
Su B, Button TW, Ponton CB (2004) Control of the particle size and morphology of hydrothermally synthesised lead zirconate titanate powders. J Mater Sci 39:6439–6447
Testino A, Buscaglia V, Buscaglia MT, Viviani M, Nanni P (2005) Kinetic modeling of aqueous and hydrothermal synthesis of barium titanate (BaTiO3). Chem Mater 17:5346–5356
Viviani M, Buscaglia MT, Nanni P, Parodi R, Gemme G, Dacca A (1999) XPS investigation of surface properties of Ba(1−x)Sr x TiO3 powders prepared by low temperature aqueous synthesis. J Eur Ceram Soc 19:1047–1051
Wang XY, Lee BI, Hu M, Payzant EA, Blom DA (2006) Nanocrystalline BaTiO3 powder via a sol process ambient conditions. J Eur Ceram Soc 26:2319–2326
Wendelbo R, Akporiaye DE, Karlsson A, Plassen M, Olafsen A (2006) Combinatorial hydrothermal synthesis and characterisation of perovskites. J Eur Ceram Soc 26:849–859
Wu A, Vilarinho PM, Reaney I, Salvado IMM (2003) Early stages of crystallization of sol-gel derived lead zirconate titanate thin films. Chem Mater 15(5):1147–1155
Xu H, Gao L (2004) Hydrothermal synthesis of high-purity BaTiO3 powders: control of powder phase and size, sintering density, and dielectric properties. Mater Lett 58:1582–1586
Acknowledgements
Financial support from FCT (POCTI/CTM/47285/2002), FEDER and the European Network of Excellence, FAME under the contract FP6-500159-1 is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, A., Vilarinho, P.M. & González, M. Synthesis and characterization of barium strontium titanate nano powders by low temperature ambient pressure sol process. J Nanopart Res 12, 2221–2231 (2010). https://doi.org/10.1007/s11051-009-9788-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9788-6