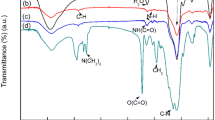
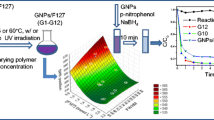
Abstract
Gold nanoparticles are immobilized in the hydrophilic coronas of spherical micelle carriers for high catalytic activity. The micelle is formed by self-assembly of block copolymer, polystyrene-b-poly (acrylic acid), in basic aqueous solution (pH 10) and has a polystyrene core and a poly (acrylic acid) corona. The gold nanoparticles are anchored into the poly (acrylic acid) corona by in situ reduction of the mixture of HAuCl4 and micelle with NaBH4. The sizes of the gold nanoparticles can be adjusted by changing the content of the HAuCl4. In the process of catalyzing p-nitrophenol to p-aminophenol, the reaction shows one-order kinetics, furthermore, the reaction rate increases with the concentration of composites as well as reaction temperature. Comparing the composites with polystyrene as core and poly (4-vinylpyridine)/Au as corona, the catalytic activity of the present composites is higher, which is ascribed to their hydrophilic corona structure.








Similar content being viewed by others
References
Anandan S, Grieser F, Ashokkumar M (2008) Sonochemical synthesis of Au–Ag core–shell bimetallic nanoparticles. J Phys Chem C 112(39):15102–15105. doi:10.1021/jp806960r
Bera P, Hedge MS (2002) Characterization and catalytic properties of combustion synthesized Au/CeO2 catalyst. Catal Lett 79(1–4):75–81. doi:10.1023/A:1015352223861
Bond GC, Thompson DT (2000) Gold-catalysed oxidation of carbon monoxide. Gold Bull 33(2):41–51
Byrd H, Clearfield A, Poojary D, Reis KP, Thompson ME (1996) Crystal structure of a porous zirconium phosphate/phosphonate compound and photocatalytic hydrogen production from related materials. Chem Mater 8(9):2239–2249. doi:10.1021/cm960030u
Campell CT, Parker SC, Starr DE (2002) The effect of size-dependent nanoparticle energetics on catalyst sintering. Science 298(5594):811–814. doi:10.1126/science.1075094
Chen S, Yang Y (2002) Magnetoelectrochemistry of gold nanoparticle quantized capacitance charging. J Am Chem Soc 124(19):5280–5281. doi:10.1021/ja025897
Chen X, Liu Y, An Yl, Lü J, Li Jb, Xiong Da, Shi Lq (2007) Novel structured composites formed from gold nanoparticles and diblock copolymers. Macromol Rapid Commun 28(12):1350–1355. doi:10.1002/marc.200700166
Chen X, Zhao Dy, An Yl, Zhang Y, Cheng J, Wang Bl, Shi Lq (2008a) Formation and catalytic activity of spherical composites with surfaces coated with gold nanoparticles. J Colloid Interface Sci 322(2):414–420. doi:10.1016/j.jcis.2008.03.029
Chen X, An Yl, Zhao, Zhang Y, Cheng J, Shi Lq (2008b) Core−shell−corona au−micelle composites with a tunable smart hybrid shell. Langmuir 24(15):8198–8204. doi:10.1021/la800244g
Daniel MC, Astruc D (2004a) Gold nanoparticles: assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem Rev 104(1):293–346. doi:10.1021/cr030698
Dokoutchaev A, James JT, Koene SC, Pathak S, Prakash GKS, Thompson ME (1999) Colloidal metal deposition onto functionalized polystyrene microspheres. Chem Mater 11(9):2389–2399. doi:10.1021/cm9900352
Ershov BG, Henglein A (1998) Time-resolved investigation of early processes in the reduction of Ag + on polyacrylate in aqueous solution. J Phys Chem B 102(52):10667–10671. doi:10.1021/jp981907a
Haiss W, Thanh NTK, Aveyard J, Fernig DG (2007) Determination of size and concentration of gold nanoparticles from UV–Vis spectra. Anal Chem 79(11):4215–4221. doi:10.1021/ac800834n
Han Y, Jiang J, Lee SS, Ying JY (2008) Reverse microemulsion-mediated synthesis of silica-coated gold and silver nanoparticles. Langmuir 24(11):5842–5848. doi:10.1021/la703440p
Haruta M, Daté M (2001) Advances in the catalysis of Au nanoparticles. Appl Catal A 222(1–2):427–437. doi:10.1016/S0926-860X(01)00847-X
Haruta M, Kobayashi T, Yamada N (1987) Novel gold catalysts for the oxidation of carbon monoxide at a temperature far below 0 °C. Chem Lett 16(2):405–408. doi:10.1246/cl.1987.405
Hussain I, Brust M, Papworth AJ, Cooper AI (2003) Preparation of acrylate-stabilized gold and silver hydrosols and gold−polymer composite films. Langmuir 19(11):4831–4839. doi:10.1021/la020710d
Johnson BFG (1999) From clusters to nanoparticles and catalysis. Coord Chem Rev 190–192:1269–1285. doi:10.1016/S0010-8545(99)00202-7
Kralik M, Biffis A (2001) Catalysis by metal nanoparticles supported on functional organic polymers. J Mol Catal A 177(1):113–138. doi:10.1016/S1381-1169(01)00313-2
Lee D, Rubner MF, Cohen RE (2005) Formation of nanoparticle-loaded microcapsules based on hydrogen-bonded multilayers. Chem Mater 17(5):1099–1105. doi:10.1021/cm048441v
Lewis LN (1990) On the mechanism of metal colloid catalyzed hydrosilylation: proposed explanations for electronic effects and oxygen cocatalysis. J Am Chem Soc 112(16):5998–6004. doi:10.1021/ja00172a014
Liu W, Xl Yang, Xie L (2007) Size-controlled gold nanocolloids on polymer microsphere-stabilizer via interaction between functional groups and gold nanocolloids. J Colloid Interface Sci 313(2):494–502. doi:10.1016/j.jcis.2007.04.055
Lu Y, Mei Y, Ballauff M (2006) Thermosensitive core–shell particles as carrier systems for metallic nanoparticles. J Phys Chem B 110(9):3930–3937. doi:10.1021/jp057149n
Matyjaszewski K, Davis KA (2000) Atom transfer radical polymerization of tert-butyl acrylate and preparation of block copolymers. Macromolecules 33(11):4039–4047. doi:10.1021/ma991826s
Moffitt M, Vali H, Eisenberg A (1998) Spherical assemblies of semiconductor nanoparticles in water-soluble block copolymer aggregates. Chem Mater 10(4):1021–1028. doi:10.1021/cm9705451.
Nandanan E, Jana N, Ying JY (2008) Functionalization of gold nanospheres and nanorods by chitosan oligosaccharide derivatives. Adv Mater 20(11):2068–2073. doi:10.1002/adma.200702193.
Okitsu K, Bandau H, Maeda Y, Nagata Y (1996) Sonochemical preparation of ultrafine palladium particles. Chem Mater 8(2):315–317. doi:10.1021/cm950285s
Peng XG, Xiao M (2003) Photoactivated CdSe nanocrystals as nanosensors for gases. Nano Lett 3(6):819–822. doi:10.1021/nl0340935
Pierrat S, Zins I, Breivogel A, Sönnichsen C (2007) Self-assembly of small gold colloids with functionalized gold nanorods. Nano Lett 7(2):259–263. doi:10.1021/nl062131p
Schubert MM, Hacjenberg S, van Vee AC, Muhler M, Plzak V, Behm R (2001) CO oxidation over supported gold catalysts—“Inert” and “Active” support materials and their role for the oxygen supply during reaction. J Catal 197(1):113–122. doi:10.1006/jcat.2000.3069
Semagina N, Joannet E, Parra S, Sulman E, Renken A, Kiwi-Minsker L (2005) Palladium nanoparticles stabilized in block-copolymer micelles for highly selective 2-butyne-1, 4-diol partial hydrogenation. Appl Catal A 280(2):141–147. doi:10.1016/j.apcata.2004.10.049
Sun X, Jiang X, Dong S, Wang E (2003) One-step synthesis and size control of dendrimer-protected gold nanoparticles: a heat-treatment-based strategy. Macromol Rapid Commun 24(17):1024–1028. doi:10.1002/marc.200300093
Trivino GC, Klabunde KJ, Dale EB (1987) Living colloidal palladium in nonaqueous solvents. Formation, stability, and film-forming properties. Clustering of metal atoms in organic media 14. Langmuir 3(6):986–992. doi:10.1021/la00078a019
Xia J, Zhang X, Matyjaszewski K (1999) Atom transfer radical polymerization of 4-vinylpyridine. Macromolecules 32(10):3531–3533. doi:10.1021/ma9816968
You CC, Verma AV, Rotello M (2006) Engineering the nanoparticle–biomacromolecule interface. Soft Matter 2(3):190–204. doi:10.1039/b517354j
Zhang G, Wu C (2000) Self-assembled nanostructure of a novel coil–rod diblock copolymer in dilute solution. J Am Chem Soc 122(41):10201–10205. doi:10.1021/ja002163t
Zhang W, Shi L, An Y, Shen X, Guo Y, Gao L, Liu Z, He B (2003) Evaporation-induced aggregation of polystyrene-block-poly(acrylic acid) micelles to microcubic particles. Langmuir 19(15):6026–6031. doi:10.1021/la034300g
Zhang W, Shi L, An Y, Gao L, Wu K, Ma R, He B (2004) Initial copolymer concentration influence on self-assembly of PS38-b-P (AA190-co-MA20) in water. Phys Chem Chem Phys 6(1):109–115. doi:10.1039/b309906g
Acknowledgment
We thank the National Natural Science Foundation of China (No. 20474032), Program for New Century Excellent Talents in Universities, and the Outstanding Youth Fund (No. 50625310) for financial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, X., Zhao, D., An, Y. et al. Catalytic properties of gold nanoparticles immobilized on the surfaces of nanocarriers. J Nanopart Res 12, 1877–1887 (2010). https://doi.org/10.1007/s11051-009-9750-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9750-7