Abstract
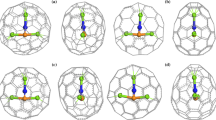
Among all the 4478 classical isomers of C66, C66(C s :0060) with the lowest number of pentagon–pentagon fusions was predicted to be the most stable isomer, followed by isomers C66(C 2v :0011) and C66(C 2:0083). The infrared spectra and aromaticity of the most stable isomers were predicted. The relative stabilities of C66 isomers change with charges or doping of metals. The structures and relative stabilities of the most stable metallofullerenes were delineated and compared with experiment. Sc2@C66(C 2:0083) was predicted to be the most stable metallofullerene, although Sc2@C66(C 2v :0011) was observed. Charge-transfer from Sc2 to the fused pentagons and the bonding between these two moieties significantly decrease the strain energies caused by the pair of fused pentagons thereby stabilizing the fullerene cage.







Similar content being viewed by others
References
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652. doi:10.1063/1.464913
Boese AD, Handy NC (2001) A new parametrization of exchange–correlation generalized gradient approximation functional. J Chem Phys 114:5497–5503. doi:10.1063/1.1347371
Boltalina OV, Ioffe IN, Sorokin ID, Sidorov LN (1997) Electron affinity of some endohedral lanthanide fullerenes. J Phys Chem A 101:9561–9563. doi:10.1021/jp972643f
Böttcher A, Weis P, Jester SS, Löffler D, Bihlmeier A, Klopper W, Kappes MM (2005) Solid C58 films. Phys Chem Chem Phys 7:2816–2820. doi:10.1039/b505703e
Brinkmann G, Dress AWM (1997) A constructive enumeration of fullerenes. J Algorithms 23:345–348. doi:10.1006/jagm.1996.0806
Campbell EEB, Fowler PW, Mitchell D, Zerbetto F (1996) Increasing cost of pentagon adjacency for larger fullerenes. Chem Phys Lett 250:544–548. doi:10.1016/0009-2614(96)00055-3
Carpenter JE, Weinhold F (1988) Analysis of the geometry of the hydroxymethyl radical by the different hybrids for different spins natural bond orbital procedure. J Mol Struct (Theochem) 169:41–62. doi:10.1016/0166-1280(88)80248-3
Chen DL, Tian WQ, Feng JK, Sun CC (2007a) Structures, stabilities, and electronic and optical properties of C52 fullerene, ions, and metallofullerenes. J Chem Phys 126:074313-1–074313-7
Chen DL, Tian WQ, Feng JK, Sun CC (2007b) Structures, stabilities, and electronic and optical properties of C58 fullerene isomers, ions, and metallofullerenes. ChemPhysChem 8:1029–1036. doi:10.1002/cphc.200600785
Chen DL, Tian WQ, Feng JK, Sun CC (2007c) Structures and electronic properties of C56Cl8 and C56Cl10 fullerene compounds. ChemPhysChem 8:2386–2390. doi:10.1002/cphc.200700405
Chen DL, Tian WQ, Feng JK, Sun CC (2008a) Theoretical investigation of C56 fullerene isomers and related compounds. J Chem Phys 128:044318-1–044318-7
Chen DL, Tian WQ, Feng JK, Sun CC (2008b) C68 fullerene isomers, anions, and their metallofullerenes charge-stabilizing different isomers. ChemPhysChem 9:454–461. doi:10.1002/cphc.200700616
Cui YH, Chen DL, Tian WQ, Feng JK (2007) Structures, stabilities, and electronic and optical properties of C62 fullerene isomers. J Phys Chem A 111:7933–7939. doi:10.1021/jp072768c
Dewar MJS, Thiel W (1977) Ground states of molecules. 38. The MNDO method. Approximations and parameters. J Am Chem Soc 99:4899–4907. doi:10.1021/ja00457a004
Eklund PC, Rao AM (1999) Fullerene polymers and fullerene-polymer composites. Springer-Verlag, Berlin, pp 1–58
Fowler PW, Manolopoulos DE (1995) An atlas of fullerenes, vol 30 of International Series of Monographs in Chemistry. Oxford University Press, Oxford
Fowler PW, Zerbetto F (1995) Charging and equilibration of fullerene isomers. Chem Phys Lett 243:36–41. doi:10.1016/0009-2614(95)00849-Y
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR et al (2004) Gaussian 03, revision C.02. Gaussian, Inc, Wallingford CT
Gao X, Zhao Y (2007) The way of stabilizing non-IPR fullerenes and structural elucidation of C54Cl8. J Comput Chem 28:795–801. doi:10.1002/jcc.20602
Guha S, Nakamoto K (2005) Electronic structures and spectral properties of endohedral fullerenes. Coord Chem Rev 249:1111–1132. doi:10.1016/j.ccr.2004.11.017
Heflin JR, Marciu D, Figura C, Wang S, Burbank P, Stevenson S et al (1998) Enhanced nonlinear optical response of an endohedral metallofullerene through metal-to-cage charge transfer. Appl Phys Lett 72:2788–2790. doi:10.1063/1.121456
Huang JY (2007) Real time microscopy, kinetics, and mechanism of giant fullerene evaporation. Phys Rev Lett 99:175503-1–175503-4
Krätschmer W, Lamb LD, Fostiropoulos K, Huffman DR (1990) Solid C: a new form of carbon. Nature 347:354–358. doi:10.1038/347354a0
Kroto HW (1987) The stability of the fullerenes C n with n = 24, 28, 32, 36, 50, 60 and 70. Nature 329:529–531. doi:10.1038/329529a0
Kroto HW, Heath JR, O’brien SC, Curl RF, Smalley RE (1985) C60: Buckminsterfullerene. Nature 318:162–163. doi:10.1038/318162a0
Lee C, Yang W, Parr RG (1988) Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789. doi:10.1103/PhysRevB.37.785
Löffler D, Jester SS, Weis P, Böttcher A, Klopper W, Kappes MM (2006) Cn films (n = 50, 52, 54, 56, and 58) on graphite: cage size dependent electronic properties. J Chem Phys 124:054705-1–054705-9
McWeeny R (1962) Perturbation theory for the Fock-Dirac density matrix. Phys Rev 126:1028–1034. doi:10.1103/PhysRev.126.1028
Miehlich B, Savin A, Stoll H, Preuss H (1989) Results obtained with the correlation energy density functionals of Becke and Lee-Yang and Parr. Chem Phys Lett 157:200–206. doi:10.1016/0009-2614(89)87234-3
Milenic DE, Brady ED, Brechbiel MW (2004) Antibody-targeted radiation cancer therapy. Nat Rev Drug Discov 3:488–498. doi:10.1038/nrd1413
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Rev 88:889–926. doi:10.1021/cr00088a005
Schleyer PvR, Manoharan M, Wang ZX, Kiran B, Jiao H, Puchta R et al (2001) Dissected nucleus-independent chemical shift analysis of π-Aromaticity and Antiaromaticity. Org Lett 3:2465–2468. doi:10.1021/ol016217v
Schmalz TG, Seitz WA, Klein DJ, Hite GE (1988) Elemental carbon cages. J Am Chem Soc 110:1113–1127. doi:10.1021/ja00212a020
Shao N, Gao Y, Zeng XC (2007) Search for lowest-energy fullerenes: C38 to C80 and C112 to C120. J Phys Chem C 111:17671–17677. doi:10.1021/jp0701082
Shi ZQ, Wu X, Wang CR, Lu X, Shinohara H (2006) Isolation and characterization of Sc2C2@C68: a metal-carbide endofullerene with a non-IPR carbon cage. Angew Chem Int Ed 45:2107–2111. doi:10.1002/anie.200503705
Shinar J, Vardeny ZV, Kafafi ZH (2000) Optical and electronic properties of fullerenes and fullerene-based materials. Marcel Dekker, Inc, New York and Basle, pp 333–365
Slanina Z (1987) Equilibrium isomeric mixtures: potential energy bypersurfaces as the origin of the overall thermodynamics and kinetics. Int Rev Phys Chem 6:251–267. doi:10.1080/01442358709353407
Stevenson S, Fowler PW, Heine T, Duchamp JC, Rice G, Glass T et al (2000) Materials science: a stable non-classical metallofullerene family. Nature 408:427–428. doi:10.1038/35044199
Stewart JJP (1989) Optimization of parameters for semiempirical methods I. Methods J Comput Chem 10:209–220. doi:10.1002/jcc.540100208
Tian WQ, Feng JK, Wang YA, Aoki Y (2006) Search for suitable approximation methods for fullerene structure and relative stability studies: case study with C50. J Chem Phys 125:094105-1–094105-10
Vosko SH, Wilk L, Nusair M (1980) Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis. Can J Phys 58:1200–1211
Wang CR, Kai T, Tomiyama T, Yoshida T, Kobayashi Y, Nishibori E et al (2000) Materials science: C66 fullerene encaging a scandium dimer. Nature 408:426–427. doi:10.1038/35044195
Wolinski K, Hinton J, Pulay P (1990) Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J Am Chem Soc 112:8251–8260. doi:10.1021/ja00179a005
Xie SY, Gao F, Lu X, Huang RB, Wang CR, Zhang X et al (2004) Capturing the labile fullerene[50] as C50Cl10. Science 304:699. doi:10.1126/science.1095567
Yan QB, Zheng QR, Su G (2007) Theoretical study on the structures, properties and spectroscopies of fullerene derivatives C66X4 (X=H, F, Cl). Carbon 45:1821–1827. doi:10.1016/j.carbon.2007.04.036
Acknowledgments
Wei Quan Tian acknowledges the start-up fund received from Jilin University. This study is supported by the National Nature Science Foundation of China (20473031), and the State Key Laboratory of Supramolecular Structure and Materials, Jilin University, China.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cui, YH., Tian, W.Q., Feng, JK. et al. Structures, stabilities, aromaticity, and electronic properties of C66 fullerene isomers, anions (C66 2−, C66 4−, C66 6−), and metallofullerenes (Sc2@C66). J Nanopart Res 12, 429–438 (2010). https://doi.org/10.1007/s11051-009-9651-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9651-9