Abstract
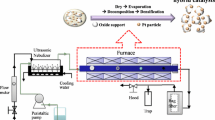
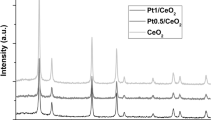
Ce–Ti–O supports with different Ce/Ti molar ratios were synthesized by the solvothermal method using hexadecyltrimethylammonium bromide. Pt nanoparticles were then supported by photochemical deposition. The shape, size, and structure of these materials were analyzed by high-resolution transmission electron microscopy. The single CeO2 support was also prepared, consisting of agglomerated cubic particles ranging from ~3 to 8 nm. When titania was combined with ceria, a nanostructured architecture was produced, evidencing the strong influence of Ti in the support structure. Photodeposition of Pt nanoparticles is more efficient on Ce–Ti–O supports than in pristine CeO2. Crystalline Pt nanoparticles (mainly of ~2 to 4 nm) were detected. The catalytic properties of the materials were tested in the selective hydrogenation of cinnamaldehyde to cinnamyl alcohol. It was observed that Pt supported on Ce–Ti–O is more active and selective than Pt on CeO2 or TiO2 separately. The catalyst with 40 mol% Ce leads to total conversion of cinnamaldehyde in a few minutes; however, higher selectivity toward the desired product (cinnamyl alcohol) was obtained with higher amounts of Ce (50 mol%).










Similar content being viewed by others
References
Abid M, Paul-Boncour V, Touroude R (2006) Pt/CeO2 catalysts in crotonaldehyde hydrogenation: selectivity, metal particle size and SMSI states. Appl Catal Gen 297:48–59. doi:10.1016/j.apcata.2005.08.048
Bell AT (2003) The impact of nanoscience on heterogeneous catalysis. Science 299:1688–1691. doi:10.1126/science.1083671
Bernal S, Calvino JJ, Cauqui MA, Gattica JM, Lopez Cartes C, Perez Omil JA, Pintado JM (2003) Some contributions of electron microscopy to the characterisation of the strong metal–support interaction effect. Catal Today 77:385–406. doi:10.1016/S0920-5861(02)00382-6
Burda C, Chen X, Narayanan R, El-Sayed MA (2005) Chemistry and properties of nanocrystals of different shapes. Chem Rev 105:1025–1102. doi:10.1021/cr030063a
Concepción P, Corma A, Silvestre-Albero J, Franco V, Chane-Ching Y (2004) Chemoselective hydrogenation catalysts: Pt on mesostructured CeO2 nanoparticles embedded within ultrathin Layers of SiO2 binder. J Am Chem Soc 126:5523–5532. doi:10.1021/ja031768x
Crisafulli C, Scire S, Giuffrida S, Ventimiglia G, Lo Nigro R (2006) An investigation on the use of liquid phase photo-deposition for the preparation of supported Pt catalysts. Appl Catal Gen 306:51–57. doi:10.1016/j.apcata.2006.03.035
da Silva AB, Jordão E, Mendes MJ, Fouilloux P (1997) Effect of metal–support interaction during selective hydrogenation of cinnamaldehyde to cinnamyl alcohol on platinum based bimetallic catalysts. Appl Catal Gen 148:253–264. doi:10.1016/S0926-860X(96)00102-0
Englisch M, Jentys A, Lercher JA (1997) Structure sensitivity of the hydrogenation of crotonaldehyde over Pt/SiO2 and Pt/TiO2. J Catal 166:25–35. doi:10.1006/jcat.1997.1494
Fang J, Bi X, Si D, Jiang Z, Huang W (2007) Spectroscopy studies of interfacial structures of CeO2–TiO2 mixed oxides. Appl Surf Sci 253:8952–8961. doi:10.1016/j.apsusc.2007.05.013
Gallezot P, Richard D (1998) Selective hydrogenation of α, β-unsaturated aldehydes. Catal Rev Sci Eng 40:81–126. doi:10.1080/01614949808007106
Ganduglia-Pirovano MV, Hofmann A, Sauer J (2007) Oxygen vacancies in transition metal rare earth oxides: current state of understanding and remaining challenges. Surf Sci Rep 62:219–270. doi:10.1016/j.surfrep.2007.03.002
Giuffrida S, Condorelli GG, Costanzo LL, Ventimiglia G, Lo Nigro R, Favazza M, Votrico E, Bongiorno C, Fragala IL (2007) Nickel nanostructured materials from liquid phase photodeposition. J Nanopart Res 9:611–619. doi:10.1007/s11051-006-9089-2
Kobayashi T, Iwamoto S, Inoue M (2006) Properties of the ceria particles prepared by the solvothermal oxidation of cerium metal. J Alloy Compd 408–412:1149–1152. doi:10.1016/j.jallcom.2004.12.188
Laha SC, Ryoo R (2003) Synthesis of thermally stable mesoporous cerium oxide with nanocrystalline frameworks using mesoporous silica templates. ChemComm 17:2138–2139. doi:10.1039/b305524h
Lee J-S, Choi S-C (2004) Crystallization behaviour of nano-ceria powders by hydrothermal synthesis using a mixture of H2O2 and NH4OH. Mater Lett 58:390–393. doi:10.1016/S0167-577X(03)00508-1
Luisetto I, Pepe F, Bemporad E (2008) Preparation and characterization of nano cobalt oxide. J Nanopart Res 10:59–67. doi:10.1007/s11051-008-9365-4
Martins TS, Hewer TLR, Freire RS (2007) Cerium: catalytic properties and environmental applications. Quim Nova 30:2001–2006
Mohr C, Claus P (2001) Hydrogenation properties of supported nanosized gold particles. Sci Prog 84:311–334. doi:10.3184/003685001783238925
Nakagawa K, Murata Y, Kishida M, Adachi M, Hiro M, Susa K (2007) Formation and reaction activity of CeO2 nanoparticles of cubic structure and various shaped CeO2–TiO2 composite nanostructures. Mater Chem Phys 104:30–39. doi:10.1016/j.matchemphys.2007.02.047
Patil S, Kuiry SC, Seal S, Vanfleet R (2002) Synthesis of nanocrystalline ceria particles for high temperature oxidation resistant coating. J Nanopart Res 4:433–438. doi:10.1023/A:1021696107498
Rawle AF (2007) Micron sized nano-materials. Powder Technol 174:6–9. doi:10.1016/j.powtec.2006.10.012
Silva AMT, Marques RN, Quinta-Ferreira RM (2004a) Catalysts based in cerium oxide for wet oxidation of acrylic acid in the prevention of environmental risks. Appl Catal Environ 47:269–279. doi:10.1016/j.apcatb.2003.09.019
Silva AMT, Oliveira ACM, Quinta-Ferreira RM (2004b) Catalytic wet oxidation of ethylene glycol: kinetics of reaction on a Mn–Ce–O catalyst. Chem Eng Sci 59:5291–5299. doi:10.1016/j.ces.2004.08.024
Silvestre-Albero J, Rodríguez-Reinoso F, Sepúlveda-Escribano A (2002) Improved metal–support interaction in Pt/CeO2–SiO2 catalysts after zinc addition. J Catal 210:127–136. doi:10.1006/jcat.2002.3670
Tang C, Bando Y, Liu B, Golberg D (2005) Cerium oxide nanotubes prepared from cerium hydroxide nanotubes. Adv Mater 17:3005–3009. doi:10.1002/adma.200501557
Tauster SJ (1987) Strong metal–support interactions. Acc Chem Res 20:389–394. doi:10.1021/ar00143a001
Tauster SJ, Fung SC, Garten RL (1978) Strong metal–support interactions. Group 8 noble metals supported on TiO2. J Am Chem Soc 100:170–175. doi:10.1021/ja00469a029
Tok AIY, Boey FYC, Dong Z, Sun XL (2007) Hydrothermal synthesis of CeO2 nano-particles. J Mater Process Technol 190:217–222. doi:10.1016/j.jmatprotec.2007.02.042
Vannice MA, Sen B (1989) Metal–support effects on the intramolecular selectivity of crotonaldehyde hydrogenation over platinum. J Catal 115:65–78. doi:10.1016/0021-9517(89)90007-9
Verdon E, Devalette M, Demazeau G (1995) Solvothermal synthesis of cerium dioxide microcrystallites: effect of the solvent. Mater Lett 25:127–131. doi:10.1016/0167-577X(95)00161-1
Zhang F, Chen J, Zhang X, Gao W, Jin R, Guan N, Li Y (2004) Synthesis of titania-supported platinum catalysts: the effect of pH on morphology control and valence state during photodeposition. Langmuir 20:9329–9334. doi:10.1021/la049394o
Zheng X, Wang S, Wang X, Wang S, Wang X, Wu S (2005) Synthesis, characterization and catalytic property of ceria spherical nanocrystals. Mater Lett 59:2769–2773. doi:10.1016/j.matlet.2005.04.025
Acknowledgments
The authors acknowledge Fundação para a Ciência e Tecnologia (FCT/DREBM), Portugal, and the Ministry of Higher Education, Science and Technology, Slovenia, for financial support from the Portugal-Slovenia Cooperation in Science and Technology (2008–2009) project “Synthesis and Characterization of Nanostructured Catalytic Materials”. AMTS acknowledges the financial support from POCI/N010/2006. GD acknowledges the financial support from the Slovenian Research Agency. BFM gratefully acknowledges FCT for the PhD grant SFRH/BD/16565/2004. This research was partially supported by project POCTI/58252/EQU/2004 approved by FCT/POCTI and co-sponsored by FEDER.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Silva, A.M.T., Machado, B.F., Gomes, H.T. et al. Pt nanoparticles supported over Ce–Ti–O: the solvothermal and photochemical approaches for the preparation of catalytic materials. J Nanopart Res 12, 121–133 (2010). https://doi.org/10.1007/s11051-009-9584-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9584-3