Abstract
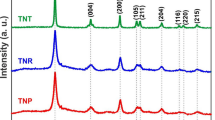
This article reports on the performances of dye-sensitized solar cells based on three different working electrode structures, i.e., (i) sintered TiO2 nanoparticles (20–40 nm diameters), (ii) ordered arrays of TiO2 nanotubules (150 nm external diameters and 80 nm internal diameters), and (iii) ordered arrays of TiO2 nanorods (150 nm diameters). Even though the highest short-circuit current density was achieved with systems based on TiO2 nanotubules, the most efficient cells were those based on ordered arrays of TiO2 nanorods. This is probably due to higher open-circuit photovoltage values attained with TiO2 nanorods than with TiO2 nanotubules. The nanorods are thicker than the nanotubules and therefore the injected electrons, stored in the trap states of the inner TiO2 molecules, are shielded from recombination with holes in the redox electrolyte at open circuit. The high short-circuit photocurrent densities seen in the ordered TiO2 systems can be explained by the fact that, as opposed to the sintered nanoparticles, the parallel and vertical orientation of the ordered nanostructures provide well-defined electrons percolation paths thus significantly reduce the diffusion distance and time constant.






Similar content being viewed by others
References
Adachi M, Murata Y, Okada I, Yoshikawa S (2003) Formation of titania nanotubes and applications for dye-sensitized solar cells. J Electrochem Soc 150:G488–G493. doi:10.1149/1.1589763
de Jong PE, Vanmaekelbergh D (1997) Investigation of the electronic transport properties of nanocrystalline particulate TiO2 electrodes by intensity-modulated photocurrent spectroscopy. J Phys Chem B 101:2716–2722. doi:10.1021/jp962226n
Ferrere S, Gregg BA (1998) Photosensitization of TiO2 by [FeII(2,2′-bipyridine-4,4′-dicarboxylic acid)2(CN)2]: band selective electron injection from ultra-short-lived excited states. J Am Chem Soc 120:843–844. doi:10.1021/ja973504e
Grätzel M (2004) Conversion of sunlight to electric power by nanocrystalline dye-sensitized solar cells. J Photochem Photobiol A Chem 164:3–14. doi:10.1016/j.jphotochem.2004.08.014
Grätzel M (2005) Solar energy conversion by dye-sensitized photovoltaic cells. Inorg Chem 44:6841–6851. doi:10.1021/ic0508371
Grunwald R, Trbutsch H (1997) Mechanisms of instability in Ru-based dye sensitization solar cells. J Phys Chem B 101:2564–2575. doi:10.1021/jp9624919
Hou Y, Xie P, Zhang B, Cao Y, Xiao X, Wang W (1999) Influence of the attaching group and substituted position in the photosensitization behavior of ruthenium polypyridyl complexes. Inorg Chem 38(26):6320–6322. doi:10.1021/ic990001w
Hulteen JC, Martin CR (1997) A general template-based method for the preparation of nanomaterials. J Mater Chem 7:1075. doi:10.1039/a700027h
Kay A, Grätzel M (1996) Low cost photovoltaic modules based on dye sensitized nanocrystalline titanium dioxide and carbon powder. Sol Energy Mater Sol Cells 44:99–117. doi:10.1016/0927-0248(96)00063-3
Kumara GRA, Kaneko S, Okuya M, Tennakone K (2002) Fabrication of dye-sensitized solar cells using triethylamine hydrothiocyanate as a cul crystal growth inhibitor. Langmuir 18(26):10493–10495. doi:10.1021/la020421p
Mor GK, Shankar K, Paulose M, Varghese OK, Grimes CA (2006) Use of highly-ordered TiO2 nanotube arrays in dye-sensitized solar cells. Nano Lett 6(2):215–218. doi:10.1021/nl052099j
Nazeeruddin MK, Péchy P, Renouard T, Zakeeruddin SM, Humphrey-Baker R, Comte P, Liska P, Cevey L, Costa E, Shklover V, Spiccia L, Deacon GB, Bignozzi CA, Grätzel M (2001) Engineering of efficient panchromatic sensitizers for nanocrystalline TiO2-based solar cells. J Am Chem Soc 123:1613–1624. doi:10.1021/ja003299u
Nusbaumer H, Moser J, Zakeeruddin SM, Nazeeruddin MK, Grätzel M (2001) CoII(dbbip)22+complex rivals tri-iodide/iodide redox mediator in dye-sensitized photovoltaic cells. J Phys Chem B 105:10461–10464. doi:10.1021/jp012075a
O’Regan B, Grätzel M (1991) A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353:737–740. doi:10.1038/353737a0
O’Regan B, Lenzmann F, Muis R, Wienke J (2002) A solid-state dye-sensitized solar cell fabricated with pressure-treated P25-TiO2 and CuSCN: analysis of pore filling and I–V characteristics. Chem Mater 14(12):5023–5029. doi:10.1021/cm020572d
Oskam G, Bergeron BV, Meyer GJ, Searson PC (2001) Pseudohalogens for dye-sensitized TiO2 photoelectrochemical cells. J Phys Chem B 105:6867–6873. doi:10.1021/jp004411d
Park NG, Schlichthrol G, van de Lagemaat J, Cheong HM, Mascarenhas A, Frank AJ (1999) Dye-sensitized TiO2 solar cells: structural and photoelectrochemical characterization of nanocrystalline electrodes formed from hydrolysis of TiCl4. J Phys Chem B 103:3308–3314. doi:10.1021/jp984529i
Suzuki K, Yamaguchi M, Kumagai M, Yanagida S (2003) Application of carbon nanotubes to counter electrodes of dye-sensitized solar cells. Chem Lett 32:28–29. doi:10.1246/cl.2003.28
Tenne R, Rao CNR (2004) Inorganic nanotubes. Phil Trans R Soc Lond A Math Phys Eng Sci 362:2099–2125
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bwana, N.N. Comparison of the performances of dye-sensitized solar cells based on different TiO2 electrode nanostructures. J Nanopart Res 11, 1917–1923 (2009). https://doi.org/10.1007/s11051-008-9545-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-008-9545-2