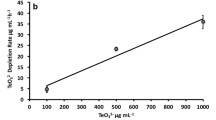
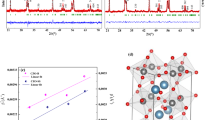
Abstract
Thermophilic bacterial strains HEN-Qn1 were incubated at 60 °C in a solution containing calcium chloride. With slow release of CO2 metabolic end products from the bacteria, CaCO3 nanomaterials were found after 12 h through a transmission electron microscope (TEM). CaCO3 nanorods were obtained extracellularly, whereas a unique morphology of nanosphere was observed intracelluarly. A single crystal was further characterized by electron pattern (ED) and X-ray powder diffraction (XRD). Moreover, a putative mechanism has been proposed based on theoretical analyses and experimental evidences. These results indicated that thermophilic bacteria had a well-controlled effect during the crystal growth of inorganic materials, which provided us a promising application of bacteria in biosynthesis of nanomaterials.






Similar content being viewed by others
References
Absar A, Debabrate R, Murali S (2004) Biogenic calcium carbonate: calcite crystals of variable morphology by the reaction of aqueous Ca2+ ions with fungi. Adv Funct Mater 14:1075–1080. doi:10.1002/adfm.200400005
Aizenberg J, Muller DA, Grazul JL, Hamman DR (2003) Direct fabrication of large micropatterned single crystals. Science 299:1205–1208. doi:10.1126/science.1079204
Bellomo EG, Wyrsta MD, Pakstis L (2004) Stimuli-responsive polypeptide vesicles by conformation-specific assembly. Nat Mater 3:244–248. doi:10.1038/nmat1093
Blakemore RP (1975) Magnetotactic bacteria. Science 190:377–379. doi:10.1126/science.170679
Chen Y, Wu QS, Ding YP (2007) Stepwise assembly of nanoparticles, -tubes, -rods, and -wires in reverse micelle systems. Eur J Inorg Chem 31:4906–4910. doi:10.1002/ejic.200700196
Colfen H, Mann S (2003) Higher-order organization by mesoscale self-assembly and transformation of hybrid nanostructures. Angew Chem Int Ed 42:2350–2365. doi:10.1002/anie.200200562
Deng ZX, Li LB, Li YD (2003) Novel inorganic-organic-layered structures: crystallographic understanding of both phase and morphology formations of one-dimensional CdE (E = S, Se, Te) nanorods in ethylenediamine. Inorg Chem 42:2331–2341. doi:10.1021/ic025846d
Li YD, Wang JW, Deng ZX (2001) Bismuth nanotubes: a rational low-temperature synthetic route. J Am Chem Soc 123:9904–9905. doi:10.1021/ja016435j
Li L, Wu QS, Ding YP (2004a) Living bio-membrane bi-template route for simultaneous synthesis of lead selenide nanorods and nanotubes. Nanotechnology 15:1877–1881. doi:10.1088/0957-4484/15/12/033
Li XY, Wu XW, Gao YY, Zhou QY (2004b) Domestic waste composting with complex thermophilic microbial inoculation. J Tongji Univ 32:367–371
Mao CB, Daniel JS, Brian DR (2004) Virus-based toolkit for the directed synthesis of magnetic and semiconducting nanowires. Science 303:213–217. doi:10.1126/science.1092740
Mehmet S, Candan T, Jen AKY, Klaus S, Baneyx F (2003) Molecular biomimetics: nanotechnology through biology. Nat Mater 2:577–585. doi:10.1038/nmat964
Rautaray D, Ahmad A, Sastry M (2003) Biosynthesis of CaCO3 crystals of complex morphology using a fungus and an actinomycete. J Am Chem Soc 125:14656–14657. doi:10.1021/ja0374877
Ronald S, Mitchell JH, Jodi SB (2004) Structural and spectral features of selenium nanoshperes produce by Se-resting bacteria. Appl Environ Microbiol 70:52–56. doi:10.1128/AEM.70.1.52-60.2004
Scott AW, Michael TK, Joseph AZ (1997) Encapsulation of bilayer vesicles by self-assembly. Nature 387:61–64. doi:10.1038/387061a0
Sean AD, Dujardin E, Mann S (2003) Biomolecular inorganic materials chemistry. Curr Opin Solid State Mater Sci 7:273–281. doi:10.1016/j.cossms.2003.09.013
Stephen M, Brigid RH, Sundara R, Birchall JD (1988) Controlled crystallization of CaCO3 under stearic acid monolayers. Nature 334:692–695. doi:10.1038/334692a0
Sun QH, Deng YL (2004) Synthesis of micrometer to nanometer CaCO3 particles via mass restriction method in an emulsion liquid membrane process. J Colloid Interface Sci 278:376–382. doi:10.1016/j.jcis.2004.06.013
Wayne S, Dietmar P, Uwe BS, Stephen M (1997) Synthesis of cadmium sulphide superlattices using self-assembled bacterial S-layers. Nature 389:585–587. doi:10.1038/39287
Xiang L, Xiang Y, Wen Y (2004) Formation of CaCO3 nanoparticles in the presence of terpineol. Mater Lett 58:959–965. doi:10.1016/j.matlet.2003.07.034
Xu X, Han JT, Cho K (2004) Formation of amorphous calcium carbonate thin films and their role in biomineralization. Chem Mater 16:1740–1746. doi:10.1021/cm035183d
Acknowledgment
This work was supported by the National Natural Science Foundation of China (No. 50772074), the Nano-Foundation of Shanghai (No. 0652nm007), and Innovation Program of Shanghai Municipal Education Commission.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, P., Wu, C., Wu, Q. et al. Biosynthesis of different morphologies of CaCO3 nanomaterial assisted by thermophilic strains HEN-Qn1. J Nanopart Res 11, 903–908 (2009). https://doi.org/10.1007/s11051-008-9476-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-008-9476-y