Abstract
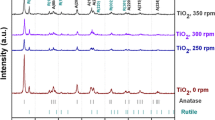
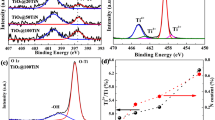
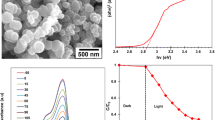
TiN/TiO2 nanoparticle photocatalyst was prepared by ball milling of TiO2 in H2O solution doped with TiN. The photocatalyst was characterized by UV–Vis diffuse reflection spectroscopy, X-ray powder diffraction (XRD), and X-ray photoelectron spectroscopy (XPS). Based on the results of the characterization, the mechanism of the increase in photocatalytic activity was investigated. The results show that when the amount of doped TiN is 0.15 wt%, the photocatalytic activity of the TiN/TiO2 is at its peak. Compared with TiO2, the photoabsorption wavelength range of the TiN/TiO2 photocatalyst red-shifts about 30 nm, and the photoabsorption intensity increases as well. The photocatalytic activities of the photocatalyst are higher than that of TiO2 under UV and visible light irradiation. The increase of surface Ti3+ reactive center and the extension of the photoabsorption wavelength are the main factors for the increase in the photocatalytic activity of the TiN/TiO2. Doped TiN neither changes the TiO2 crystal phase nor creates new crystal phase by ball milling.






Similar content being viewed by others
References
Anpo M, Takeuchi M, Ikeue K, Dohshi S (2000) Design and development of titanium oxide photocatalysts operating under visible and UV light irradiation: the applications of metal ion-implantation techniques to semiconducting TiO2 and Ti/zeolite catalysts. Curr Opin Solid State Mater Sci 6:381–388
Asahi R, Morikawa T, Ohwaki T, Aoki K, Taga Y (2001) Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 293:269–271
Cesar L, Kay A, Martinez JAG, Gratzel MJ (2006) Translucent thin film Fe2O3 photoanodes for efficient water splitting by sunlight: nanostructure-directing effect of Si-doping. J Am Chem Soc 128:4582–4583
Chen X, Burda C (2004) Photoelectron spectroscopic investigation of nitrogen-doped titania nanoparticles. J Phys Chem B 108:15446–15449
Chen SF, Cao GY (2005a) The preparation of nitrogen-doped photocatalyst TiO2−xNx by ball milling. Chem Phys Lett 413:404–409
Chen SF, Cao GY (2005b) The preparation of coupled WO3/TiO2 photocatalyst by ball milling. Powder Technol 160:198–202
Chen SF, Cheng XL, Tao YW, Zhao MY (1998) Photocatalytic degradation of trace gaseous acetone and acetaldehyde using TiO2 supported on fiberglass cloth. J Chem Technol Biotechnol 73:264–268
Chen SF, Liang X, Tao YW, Zhao MY (1999) Photocatalytic degradation of organophorous pesticide using TiO2 supported on hollow glass microbeads. Photogr Sci Photochem (Chin) 17:85–91
Chen YS, Crittenden JC, Hackney S, Sutter L, Hand DW (2005) Preparation of a novel TiO2-based p-n junction nanotube photocatalyst. Environ Sci Technol 39:1201–1208
Hu C, Lan Y, Qu J, Hu X, Wang A (2006) Ag/AgBr/TiO2 visible light photocatalyst for destruction of azodyes and bacteria. J Phys Chem B 110:4066–4072
Ihara T, Miyoshi M, Iriyama Y (2003) Visible-light-active titanium oxide photocatalyst realized by an oxygen-deficient structure and by nitrogen doping. Appl Catal B: Environ 42:403–409
Jung KY, Park BP, Ihm SK (2004) Local structure and photocatalytic activity of B2O3-SiO2/TiO2 ternary mixed oxides prepared by sol-gel method. Appl Catal B: Environ 51:239–245
Khah SUM, Al-shahry M, Lngler WB (2002) Efficient photochemical water splitting by a chemically modified n-TiO2. Science 297:2243–2245
Li FB, Li XZ, Hou MF (2004) Photocatalytic degradation of 2-mercaptobenzothiazole in aqueous La3+-TiO2 suspension for odor control. Appl Catal B: Environ 48:185–194
Li D, Haneda H, Labhsetwar NK, Hishita S, Ohashi N (2005a) Visible-light-driven photocatalysis on fluorine-doped TIO2 powders by the creation of surface oxygen vacancies. Chem Phys Lett 401:579–584
Li D, Haneda H, Hishita S, Ohashi N (2005b) Visible-light-driven nitrogen-doped TiO2 photocatalysts: effect of nitrogen precursors on their photocatalysis for decomposition of gas-phase organic pollutants. Mat Sci Eng 117:67–75
Liu SX, Qu ZP, Han XW, Sun CL (2004) A mechanism for enhanced photocatalytic activity of silver-loaded titanium dioxide. Catal Today 93:877–884
Sathish M, Viswanathan B, Viswanath RP, Gopinath CS (2005) Synthesis, characterization, electronic structure, and photocatalytic activity of nitrogen-doped TiO2 nanocatalyst. Chem Mater 17:6349–6353
Wang JS, Yin S, Komatsu M, Zhang QW, Saito F, Sato T (2004) Preparation and characterization of nitrogen doped SrTiO3 photocatalyst. J Photochem Photobiol A: Chem 165:149–156
Wang XX, Lian WH, Fu XZ, Basset J, Lefebvre F (2006) Structure, preparation and photocatalytic activity of titanium oxides on MCM-41 surface. J Catal 238:13–20
Yin S, Yamaki H, Komatsu M (2005) Synthesis of visible-light reactive TiO2−xNx photocatalyst by mechanochemical doping. Solid State Sci 12:1479–1485
Yu JG, Wang GH, Cheng B, Zhou MH (2007) Effects of hydrothermal temperature and time on the photocatalytic activity and microstructures of bimodal mesoporous TiO2 powders. Appl Catal B: Environ 69:171–180
Acknowledgments
This work was supported by the Natural Science Foundation of China (No. 20673042), the Natural Science Foundation of Anhui Province (Contract No. 070415211), the Key Project of Science and Technology Research of Ministry of Education of China (208062), and the Natural Science Foundation of Anhui Provincial Education Committee (KJ2007A015).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, S., Zhang, S., Zhao, W. et al. Study on the photocatalytic activity of TiN/TiO2 nanoparticle formed by ball milling. J Nanopart Res 11, 931–938 (2009). https://doi.org/10.1007/s11051-008-9475-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-008-9475-z