Abstract
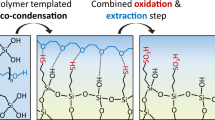
We compare the methods of continuous solvent (Soxhlet) and supercritical solvent extractions for the removal of the organic template from nanostructured silica monoliths. Our monoliths are formed by templating the L 3 liquid crystal phase of cetylpyridinium chloride in aqueous solutions with tetramethoxy silane. The monoliths that result from both Soxhlet and supercritical extraction methods are mechanically robust, optically clear, and free of cracks. The Soxhlet method compares favorably with supercritical solvent extraction in that equivalent L 3-templated silica can be synthesized without the use of specialized reactor hardware or higher temperatures and high pressures, while avoiding noxious byproducts. The comparative effectiveness of various solvents in the Soxhlet process is related to the Hildebrand solubility parameter, determined by the effective surface area of the extracted silica.
Similar content being viewed by others
References
Beck J.S., Vartuli J.C., Roth W.J., Leonowicz M.E., Kresge C.T., Schmitt K.D., Chu C.T.W., Olson D.H., Sheppard E.W., McCullen S.B., Higgins J.B. & Schlenker J.L. (1992). A new family of mesoporous molecular sieves prepared with liquid crystal templates.J. Am. Chem. Soc. 114(27):10834–10843
Bhansali S., A.-S. Malik, I. Akartuna, D.M. Dabbs, J.C. Carbeck & I.A. Aksay, 2006. The stability of L3 sponge phase in acidic solutions. Langmuir (in review).
Brennecke J.F. (1997). Molecular trees for green chemistry.Nature 389(6649):333–334
Brinker C.J., Scherer G.W. (1990) Sol-Gel Science. Academic Press, Inc., New York, pp. 444–448 and 493–505
Burke, J. 1984, In: Jensen C. ed., AIC Book and Paper Group Annual, Vol. 3, American Institute for Conservation of Historic and Artistic Works, Washington, D.C., pp. 13–58
Dabbs D.M., Aksay I.A. (2000). Self-assembled ceramics produced by complex–fluid templation. Annu. Rev. Phys. Chem. 51:601–22
Dale Spall F.K. & Laintz K.E., 1998. In: McHardy, J. & Sawan S.P. eds. Supercritical Fluid Cleaning; Fundamentals, Technology, and Applications, Noyes Publications, Westwood, NJ, pp. 162–194
Darr J.A., Poliakoff M. (1999). New directions in inorganic and metal–organic coordination chemistry in supercritical fluids.Chem. Rev. 99(2):495–541
van Grieken R., Calleja G., Stucky G.D., Melero J.A., Garcia R.A., Iglesias J. (2003). Supercritical fluid extraction of a nonionic surfactant template from SBA-15 materials and consequences on the porous structure.Langmuir 19(9):3966–3973
Kawi S., Lai M.W. (2002). Supercritical fluid extraction of surfactant from Si-MCM-41.AIChE Journal 48(7):1572–1580
Kim S.S., Zhang W.Z., Pinnavaia T.J. (1998). Ultrastable mesostructured silica vesicles.Science 282(5392):1302–1305
Kresge C.T., Leonowicz M.E., Roth W.J., Vartuli J.C., Beck J.S. (1992). Ordered mesoporous molecular sieves synthesized by a liquid–crystal templated mechanism.Nature 359(6397):710–712
Lee H., Zones S.I., Davis M.E. (2003). A combustion-free methodology for synthesizing zeolites and zeolite-like materials.Nature 425(6956):385–388
Lei N., Safinya C.R., Roux D., Liang K.S. (1997). Synchrotron x-ray-scattering studies on the sodium dodecyl sulfate–water–pentanol–dodecane L3 sponge phase.Phys. Rev.E 56(1):608–613
Malik A.-S., D.M. Dabbs, H.E. Katz & I.A. Aksay, 2006.Silica monoliths templated on L3 liquid crystal. Langmuir 22(1), 325–331
McGehee M.D., S.M. Gruner, N. Yao, C.M. Chun, A. Navrotsky, I.A. Aksay, 1994. Proc. 52nd Ann. Meeting Microscopy Soc. Am.; Bailey G.W., A.J. Garret-Reed, eds. San Francisco Press: San Francisco, CA, 1994; pp. 448–449
McGrath K.M. (1997). Formation of two lamellar phases in the dilute region of a quasiternary surfactant system.Langmuir 13(7):1987–1995
McGrath K.M., D.M. Dabbs, N. Yao, I.A. Aksay & S.M. Gruner, 1997. The formation of a silicate L3 phase with continuously adjustable pore sizes. Science, 277 (5325), 552–556; ibid., 1998, 279(5355), 1289
McGrath K.M., Dabbs D.M., Yao N., Edler K.J., Aksay I.A., Gruner S.M. (2000). Silica gels with tunable nanopores through templating of the L3 phase.Langmuir 16(2):398–406
See for example, McHardy J. & S. P. Sawan eds., 1998. Supercritical Fluid Cleaning; Fundamentals, Technology, and Applications, Noyes Publications, Westwood, NJ, 1998
Noble K., Seddon A.B., Turner M.L., Chevalier P., Ou D.L. (2003). Porous siloxane-silica hybrid materials by sol–gel processing.Journal of Sol–Gel Science and Technology 26(1–3):419–423
Patarin J. (2004). Mild methods for removing organic templates from inorganic host materials.Angew.Chemie-Intern.Ed. 43(30):3878–3880
Pinnavaia T.J. & M.F. Thorpe, eds., 1996. Access in Nanoporous Materials, Plenum, New York
Porte G. (1992). Lamellar phases and disordered phases of fluid bilayer membranes.J.Phys.-Condensed Matter 4(45):8649–8670
Prakash S.S., Brinker C.J., Hurd A.J., Rao S.M. (1995). Silica aerogel films prepared at ambient pressure by using surface derivatization to induce reversible drying shrinkage.Nature 374(6521):439–443
Skouri M., Marignan J., Appell J., Porte G. (1991). Fluid membranes in the “semirigid regime”: Scale-invariance.J.Phys.II 1(9):1121–1132
Strey R., Jahn W., Porte G., Bassereau P. (1990). Freeze fracture electron microscopy of dilute lamellar and anomalous isotropic (L3) phases.Langmuir 6(11):1635–1639
Tanev P.T., Pinnavaia T.J. (1996). Biomimetic templating of porous lamellar silicas by vesicular surfactant assemblies.Science 271(5253):1267–1269
Whitehurst, D.D. 1992. Method to recover organic templates from freshly synthesized molecular sieves. U.S. Patent#5,143,879; September 1, 1992
Acknowledgements
Support for this research was provided by support from Lucent Technologies and the DARPA-sponsored MURI program at Princeton University (Grant # DAAH04-95-1-0102). Additional support was provided by NASA under the BIMat URETI (Grant # NCC-1-02037).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dabbs, D.M., Mulders, N. & Aksay, I.A. Solvothermal removal of the organic template from L 3 (“sponge”) templated silica monoliths. J Nanopart Res 8, 603–614 (2006). https://doi.org/10.1007/s11051-005-9063-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-005-9063-4