Abstract
Chemical reactions and diffusion can produce a wide variety of static or transient spatial patterns in the concentrations of chemical species. Little is known, however, about what dynamical patterns of concentrations can be reliably programmed into such reaction–diffusion systems. Here we show that given simple, periodic inputs, chemical reactions and diffusion can reliably emulate the dynamics of a deterministic cellular automaton, and can therefore be programmed to produce a wide range of complex, discrete dynamics. We describe a modular reaction–diffusion program that orchestrates each of the fundamental operations of a cellular automaton: storage of cell state, communication between neighboring cells, and calculation of cells’ subsequent states. Starting from a pattern that encodes an automaton’s initial state, the concentration of a “state” species evolves in space and time according to the automaton’s specified rules. To show that the reaction–diffusion program we describe produces the target dynamics, we simulate the reaction–diffusion network for two simple one-dimensional cellular automata using coupled partial differential equations. Reaction–diffusion based cellular automata could potentially be built in vitro using networks of DNA molecules that interact via branch migration processes and could in principle perform universal computation, storing their state as a pattern of molecular concentrations, or deliver spatiotemporal instructions encoded in concentrations to direct the behavior of intelligent materials.









Similar content being viewed by others
Notes
In practice, catalysts have a fixed turnover number. Reactions that cyclically produce and degrade catalysts could enable the periodic replacement of key catalysts (and other catalysts in the system) that are no longer functional.
References
Allen PB, Chen X, Ellington AD (2012) Spatial control of DNA reaction networks by DNA sequence. Molecules 17:13390–13402
Baker MD, Wolanin PM, Stock JB (2006) Signal transduction in bacterial chemotaxis. Bioessays 28(1):9–22
Bánsági T, Vanag VK, Epstein IR (2011) Tomography of reaction–diffusion microemulsions reveals three-dimensional Turing patterns. Science 331(6022):1309–1312
Chen Y, Dalchau N, Srinivas N, Phillips A, Cardelli L, Soloveichik D, Seelig G (2013) Programmable chemical controllers made from DNA. Nat Nanotechnol 8(10):755–762
Chirieleison SM, Allen PB, Simpson ZB, Ellington AD, Chen X (2013) Pattern transformation with DNA circuits. Nat Chem 5:1000–1005
Codd EF (1968) Cellular automata. Academic Press Inc, San Diego
Codon A, Kirkpatrick B, Maňuch J (2012) Reachability bounds for chemical reaction networks and strand displacement systems. DNA Computing and Molecular Programming. Springer, Heidelberg, Berlin
Cook M (2004) Universality in elementary cellular automata. Complex Syst 15(1):1–40
Dalchau N, Seelig G, Phillips A (2014) Computational design of reaction–diffusion patterns using DNA-based chemical reaction networks. DNA computing and molecular programming. Springer, Heidelberg, Berlin
Danino T, Mondragn-Palomino O, Tsimring L, Hasty J (2010) A synchronized quorum of genetic clocks. Nature 463(7279):326–330
Doty D (2014) Timing in chemical reaction networks. In: Proceedings of the 25th ACM-SIAM symposium on discrete algorithms, pp 772–784
Du Y, Lo E, Ali S, Khademhosseini A (2008) Directed assembly of cell-laden microgels for fabrication of 3D tissue constructs. In; Proceedings of the National Academy of Sciences 105(28):9522–9527
Fujibayashi K, Hariadi R, Park SH, Winfree E, Murata S (2007) Toward reliable algorithmic self-assembly of DNA tiles: a fixed-width cellular automaton pattern. Nano Lett 8(7):1791–1797
Gács P (2001) Reliable cellular automata with self-organization. J Stat Phys 103(1/2):45–267
Gács P, Reif J (1988) A simple three-dimensional real-time reliable cellular array. J Comput Syst Sci 36(2):125–147
Lakin M, Phillips A, Stefanovic D (2013) Modular verification of DNA strand displacement networks via serializability analysis. DNA computing and molecular programming. Springer, Heidelberg, Berlin
Greenfield D, McEvoy AL, Shroff H, Crooks GE, Wingreen NS, Betzig E, Liphardt J (2009) Self-organization of the Escherichia coli chemotaxis network imaged with super-resolution light microscopy. PLoS Biol. 7(6)
Langton CG (1984) Self-reproduction in cellular automata. Phys D 10(1):135–144
Lindenmayer A (1968) Mathematical models for cellular interactions in development I. filaments with one-sided inputs. J Theor Biol 18(3):280–299
Lukacs G, Haggie P, Seksek O, Lechardeur D, Verkman NFA (2000) Size-dependent DNA mobility in cytoplasm and nucleus. J Biol Chem 275(1625)
Montagne K, Plasson R, Sakai Y, Fujii T, Rondelez Y (2011) Programming an in vitro DNA oscillator using a molecular networking strategy. Mol Sys Biol 7(1)
Murray JD (2003) Mathematical biology II: spatial models and biomedical applications, 3rd edn. Springer, New York
Neary T, Woods D (2006) P-completeness of cellular automaton rule 110. LNCS 4051(132–143)
Nehaniv CL (2004) Asynchronous automata networks can emulate any synchronous automata network. Int J Algebra Comput 14(05):719–739
von Neumann J, Burks AW (1966) The theory of self-reproducing automata. University of Illinois Press, Urbana
Qian L, Soloveichik D, Winfree E (2011) Efficient turing-universal computation with DNA polymers. DNA computing and molecular programming pp 123–140
Qian L, Winfree E (2011) Scaling up digital circuit computation with DNA strand displacement. Science 332(6034):1196–1201
Qian L, Winfree E (2011) A simple DNA gate motif for synthesizing large-scale circuits. J R Soc Interface 8(62):1281–1297
Qian L, Winfree E (2014) Parallel and scalable computation and spatial dynamics with DNA-based chemical reaction networks on a surface. DNA computing and molecular programming. Springer, Heidelberg, Berlin
Rothemund PWK, Papadakis N, Winfree E (2004) Algorithmic self-assembly of DNA Sierpinski triangles. PLoS Biol 2(12):e424
Ruiza SA, Chen CS (2007) Microcontact printing: a tool to pattern. Soft Matter 3:168–177
Sayama H (1999) A new structurally dissolvable self-reproducing loop evolving in a simple cellular automata space. Artif Life 5(4):343–365
Scalise D, Schulman R (2014) Designing modular reaction–diffusion programs for complex pattern formation. Technology 2(01):55–66
Seelig G, Soloveichik D, Zhang DY, Winfree E (2006) Enzyme-free nucleic acid logic circuits. Science 314:1585–1588
Smith DE, Perkins TT, Chu S (1996) Dynamical scaling of DNA diffusion coefficients. Macromolecules 29(4):1372–1373
Soloveichik D, Cook M, Winfree E, Bruck J (2008) Computation with finite stochastic chemical reaction networks. Nat Comput 7(4):615–633
Soloveichik D, Seelig G, Winfree E (2010) DNA as a universal substrate for chemical kinetics. In: Proceedings of the National Academy of Sciences 107(12):5393–5398
Steinbock O, Kettunen P, Showalter K (1996) Chemical wave logic gates. J Phys Chem 100(49):18970–18975
Stellwagen E, Lu Y, Stellwagen N (2003) Unified description of electrophoresis and diffusion for DNA and other polyions. Biochemistry 42:11745
Tomita K, Kurokawa H, Murata S (2002) Graph automata: natural expression of self-reproduction. Phys D: Nonlin Phenom 171(4):197–210
Tóth Ágota, Showalter K (1995) Logic gates in excitable media. J Chem Phys 103(6):2058–2066
Turing AM (1952) The chemical basis of morphogenesis. Phil T R Soc B 237:37–72
Wu A, Rosenfeld A (1979) Cellular graph automata. I. basic concepts, graph property measurement, closure properties. Inf Control 42(3):305–329
Zhang DY, Winfree E (2009) Control of DNA strand displacement kinetics using toehold exchange. J Am Chem Soc 131(47):17303–17314
Acknowledgments
The authors would like to thank Paul Rothemund, Damien Woods, Josh Fern, John Zenk, and the anonymous referees for insightful reading and comments. This work was supported by NSF-CCF-1161941 and a grant to the Turing Centenary Project by the John Templeton Foundation.
Author information
Authors and Affiliations
Corresponding author
Appendices
Rule 110 chemical reactions (synonymous with Sect. 8)
This section describes the complete set of abstract chemical reactions that govern our Rule 110 chemical automaton. These chemical reactions can be converted into a set of coupled partial differential equations and solved to observe how the system behaves over time (see Section 8). Figure 10 shows a detail of the circuit from Fig. 7 for a Rule 110 automaton, with each chemical species labeled.
-
1.
Communication stage
-
a.
Broadcasting:
$$SrcA+keyA+Last\mathop {\rightarrow {}}\limits ^{k_{B}}SrcA+SigA+keyA+Last$$(7.1)$$SigA\mathop {\rightarrow {}}\limits ^{k_{Bd}}waste $$(7.2)$$SrcB+keyB+Last\mathop {\rightarrow {}}\limits ^{k_{B}}SrcB+SigB+keyB+Last $$(7.3)$$SigB\mathop {\rightarrow {}}\limits ^{k_{Bd}}waste $$(7.4)$$SrcC+keyC+Last\mathop {\rightarrow {}}\limits ^{k_{B}}SrcC+SigC+keyC+Last $$(7.5)$$SigC\mathop {\rightarrow {}}\limits ^{k_{Bd}}waste $$(7.6)$$SrcD+keyD+Last\mathop {\rightarrow {}}\limits ^{k_{B}}SrcD+SigD+keyD+Last $$(7.7)$$SigD\mathop {\rightarrow {}}\limits ^{k_{Bd}}waste $$(7.8)
-
a.
Receiving and processing left-hand signal:
$$SigD+keyA\mathop {\rightarrow {}}\limits ^{k_{x}}SigD+Lraw+keyA $$(7.9)$$SigA+keyB\mathop {\rightarrow {}}\limits ^{k_{x}}SigA+Lraw+keyB $$(7.10)$$SigB+keyC\mathop {\rightarrow {}}\limits ^{k_{x}}SigB+Lraw+keyC $$(7.11)$$SigC+keyD\mathop {\rightarrow {}}\limits ^{k_{x}}SigC+Lraw+keyD $$(7.12)$$Lraw\mathop {\longrightarrow {}}\limits ^{4*k_{x}}waste $$(7.13)$$source\mathop {\longrightarrow {}}\limits ^{c_{recTh}*k_{p}}ThL\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.14)$$source\mathop {\rightarrow {}}\limits ^{k_{p}}AmpL\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.15)$$Lraw+ThL\mathop {\rightarrow {}}\limits ^{k_{T}}waste $$(7.16)$$Lraw+AmpL\mathop {\rightarrow {}}\limits ^{k_{L}}Lraw+Lft $$(7.17)$$Lft\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.18)
-
a.
Receiving and processing right-hand signal:
$$SigB+keyA\mathop {\rightarrow {}}\limits ^{k_{x}}SigB+Rraw+keyA $$(7.19)$$SigC+keyB\mathop {\rightarrow {}}\limits ^{k_{x}}SigC+Rraw+keyB $$(7.20)$$SigD+keyC\mathop {\rightarrow {}}\limits ^{k_{x}}SigD+Rraw+keyC $$(7.21)$$SigA+keyD\mathop {\rightarrow {}}\limits ^{k_{x}}SigA+Rraw+keyD $$(7.22)$$Rraw\mathop {\longrightarrow {}}\limits ^{4*k_{x}}waste $$(7.23)$$source\mathop {\longrightarrow {}}\limits ^{c_{recTh}*k_{p}}Thr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.24)$$source\mathop {\longrightarrow {}}\limits ^{k_{p}}Ampr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.25)$$Rraw+Thr\mathop {\longrightarrow {}}\limits ^{k_{T}}waste $$(7.26)$$Rraw+Ampr\mathop {\longrightarrow {}}\limits ^{k_{L}}Rraw+Rght $$(7.27)$$Rght\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.28)
-
a.
-
2.
Calculation Stage
-
a.
Copy left, right and previous time step (pr) signals (multiple gates operate on each):
$$Last\mathop {\longrightarrow {}}\limits ^{k_{x}}Last+BrPr $$(7.29)$$BrPr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.30)$$Rght\mathop {\longrightarrow {}}\limits ^{k_{x}}Rght+RBr $$(7.31)$$RBr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.32)$$Last\mathop {\longrightarrow {}}\limits ^{k_{x}}Last+OnPr $$(7.33)$$OnPr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.34)$$Rght\mathop {\longrightarrow {}}\limits ^{k_{x}}Rght+RCr $$(7.35)$$RCr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.36)$$Lft\mathop {\longrightarrow {}}\limits ^{k_{x}}Lft+LCr $$(7.37)$$LCr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.38)$$source\mathop {\longrightarrow {}}\limits ^{k_{p}}OffPr $$(7.39)$$OffPr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.40)$$BrPr+OffPr\mathop {\longrightarrow {}}\limits ^{k_{T}}waste $$(7.41)
-
a.
Boolean logic for Br (birth) condition: off to on transition:
$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}SgBr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.42)$$OffPr+SgBr\mathop {\longrightarrow {}}\limits ^{k_{L}}SumBr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.43)$$RBr+SgBr\mathop {\longrightarrow {}}\limits ^{k_{L}}SumBr $$(7.44)$$source\mathop {\longrightarrow {}}\limits ^{1.35*k_{p}}ThBr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.45)$$source\mathop {\longrightarrow {}}\limits ^{k_{p}}AmpBr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.46)$$SumBr+ThBr\mathop {\longrightarrow {}}\limits ^{k_{T}}waste $$(7.47)$$SumBr+AmpBr\mathop {\longrightarrow {}}\limits ^{k_{L}}SumBr+Br $$(7.48)$$Br\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.49)
-
a.
Boolean logic for no death condition (on and at least one neighbor off): stay on:
$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}SgnbOff\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.50)$$RCr+SgnbOff\mathop {\longrightarrow {}}\limits ^{k_{L}}SumnbOff\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.51)$$LCr+SgnbOff\mathop {\longrightarrow {}}\limits ^{k_{L}}SumnbOff $$(7.52)$$source\mathop {\longrightarrow {}}\limits ^{1.35*k_{p}}ThnbOff\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.53)$$source\mathop {\longrightarrow {}}\limits ^{k_{p}}AmpnbOff\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.54)$$SumnbOff+ThnbOff\mathop {\longrightarrow {}}\limits ^{k_{T}}waste $$(7.55)$$ThnbOff+AmpnbOff\mathop {\longrightarrow {}}\limits ^{k_{L}}ThnbOff+nbrOff $$(7.56)$$nbrOff\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.57)$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}SgCr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.58)$$OnPr+SgCr\mathop {\longrightarrow {}}\limits ^{k_{L}}SumCr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.59)$$nbrOff+SgCr\mathop {\longrightarrow {}}\limits ^{k_{L}}SumCr $$(7.60)$$source\mathop {\longrightarrow {}}\limits ^{1.35*k_{p}}ThCr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.61)$$source\mathop {\longrightarrow {}}\limits ^{k_{p}}AmpCr\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.62)$$SumCr+ThCr\mathop {\longrightarrow {}}\limits ^{k_{T}}waste $$(7.63)$$SumCr+AmpCr\mathop {\longrightarrow {}}\limits ^{k_{L}}SumCr+Live $$(7.64)$$Live\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.65)
-
a.
Boolean logic to determine if next state is on:
$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}SgNx\mathop {\longrightarrow {}}\limits ^{k_{x}}waste $$(7.66)$$Live+SgNx\mathop {\rightarrow {}}\limits ^{k_{L}}SumNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.67)$$Br+SgNx\mathop {\rightarrow {}}\limits ^{k_{L}}SumNx $$(7.68)$$source\mathop {\longrightarrow {}}\limits ^{0.65*k_{p}}ThNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.69)$$source\mathop {\rightarrow {}}\limits ^{k_{p}}AmpNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.70)$$SumNx+ThNx\mathop {\rightarrow {}}\limits ^{k_{T}}waste $$(7.71)$$SumNx+AmpNx\mathop {\rightarrow {}}\limits ^{k_{L}}SumNx+Nx $$(7.72)$$Nx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.73)$$Nx\mathop {\rightarrow {}}\limits ^{k_{x}}Nx+OnNx $$(7.74)$$OnNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.75)
-
a.
Boolean logic to determine if next state is off:
$$Nx\mathop {\rightarrow {}}\limits ^{k_{x}}Nx+NtNx $$(7.76)$$NtNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.77)$$source\mathop {\rightarrow {}}\limits ^{k_{p}}OffNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.78)$$NtNx+OffNx\mathop {\rightarrow {}}\limits ^{k_{T}}waste $$(7.79)
-
a.
Clocked synchronization gates:
$$clk\mathop {\rightarrow {}}\limits ^{k_{x}}clk+clkOn $$(7.80)$$clkOn\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.81)$$clk\mathop {\rightarrow {}}\limits ^{k_{x}}clk+clkOff $$(7.82)$$clkOff\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.83)$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}SgNxOn\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.84)$$OnNx+SgNxOn\mathop {\rightarrow {}}\limits ^{k_{L}}OnNxSum\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.85)$$clkOn+SgNxOn\mathop {\rightarrow {}}\limits ^{k_{L}}OnNxSum $$(7.86)$$source\mathop {\longrightarrow {}}\limits ^{1.35*k_{p}}OnNxTh\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.87)$$source\mathop {\rightarrow {}}\limits ^{k_{p}}OnNxAmp\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.88)$$OnNxSum+OnNxTh\mathop {\rightarrow {}}\limits ^{k_{T}}waste $$(7.89)$$OnNxSum+OnNxAmp\mathop {\rightarrow {}}\limits ^{k_{L}}OnNxSum+SetBfr $$(7.90)$$SetBfr\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.91)$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}SgOffNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.92)$$OffNx+SgOffNx\mathop {\rightarrow {}}\limits ^{k_{L}}SumOffNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.93)$$clkOff+SgOffNx\mathop {\rightarrow {}}\limits ^{k_{L}}SumOffNx $$(7.94)$$source\mathop {\longrightarrow {}}\limits ^{1.35*k_{p}}ThOffNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.95)$$source\mathop {\rightarrow {}}\limits ^{k_{p}}AmpOffNx\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.96)$$SumOffNx+ThOffNx\mathop {\rightarrow {}}\limits ^{k_{T}}waste $$(7.97)$$SumOffNx+AmpOffNx\mathop {\rightarrow {}}\limits ^{k_{L}}SumOffNx+ResBfr $$(7.98)$$ResBfr\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.99)
-
a.
-
3.
Storage Stage
-
a.
Copies of Set/Res signals:
$$SetBfr\mathop {\rightarrow {}}\limits ^{k_{x}}SetBfr+Set $$(7.100)$$Set\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.101)$$ResBfr\mathop {\rightarrow {}}\limits ^{k_{x}}ResBfr+Res $$(7.102)$$Res\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.103)
-
a.
Flip-flop module:
$$ffBfrd\mathop {\rightarrow {}}\limits ^{k_{x}}ffBfrd+ffFback $$(7.104)$$ffFback\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.105)$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}N1Sg\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.106)$$Set+N1Sg\mathop {\rightarrow {}}\limits ^{k_{L}}N1Sum\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.107)$$ffFback+N1Sg\mathop {\rightarrow {}}\limits ^{k_{L}}N1Sum $$(7.108)$$source\mathop {\longrightarrow {}}\limits ^{0.65*k_{p}}N1Th\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.109)$$source\mathop {\rightarrow {}}\limits ^{k_{p}}N1\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.110)$$N1Sum+N1Th\mathop {\rightarrow {}}\limits ^{k_{T}}waste $$(7.111)$$N1Th+N1\mathop {\rightarrow {}}\limits ^{k_{L}}N1Th+ffFbackNot $$(7.112)$$ffFbackNot\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.113)$$source\mathop {\longrightarrow {}}\limits ^{2*k_{p}}N2Sg\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.114)$$Res+N2Sg\mathop {\rightarrow {}}\limits ^{k_{L}}N2Sum\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.115)$$ffFbackNot+N2Sg\mathop {\rightarrow {}}\limits ^{k_{L}}N2Sum $$(7.116)$$source\mathop {\longrightarrow {}}\limits ^{0.65*k_{p}}N2Th\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.117)$$source\mathop {\rightarrow {}}\limits ^{k_{p}}N2\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.118)$$N2Sum+N2Th\mathop {\rightarrow {}}\limits ^{k_{T}}waste $$(7.119)$$N2Th+N2\mathop {\rightarrow {}}\limits ^{k_{L}}N2Th+ffBfrd $$(7.120)$$ffBfrd\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.121)$$ffBfrd\mathop {\rightarrow {}}\limits ^{k_{x}}ffBfrd+Last $$(7.122)$$Last\mathop {\rightarrow {}}\limits ^{k_{x}}waste $$(7.123)
-
a.
Rule 110 partial differential equations
“I hope to say something about a ‘continuous’ rather than ‘crystalline’ model [of automata]. There, as far as I can now see, a system of nonlinear partial differential equations, essentially of the diffusion type, will be used.” John von Neumann (Oct. 28th, 1952) discussing unfinished Theory of Automata.
von Neumann papers, Library of Congress, Box 28 “Theory of Automata”.
This section describes the set of coupled partial differential equations that govern our Rule 110 chemical automaton, derived from the reactions in this section. These equations use standard mass-action equations and the diffusion equation. Figure 9b contains a plot of the solution to these equations. One equation is devoted to each of the 76 species in our network. Figure 10 shows a detail of the circuit from Fig. 7 for a Rule 110 automaton, with each species labeled.
Unless otherwise specified, all species start with zero initial concentration. Absorbing boundary conditions apply to all species whose concentrations change over time. Our reaction rate constants and diffusion coefficients are selected to be realistically attainable values for DNA-based reaction–diffusion networks, on the same order of magnitude as experimentally derived data in the literature (Zhang and Winfree 2009; Lukacs et al. 2000; Stellwagen et al. 2003). The Mathematica code we used to numerically solve these equations is available upon request. Constants:
-
1.
External signals:
-
a.
Keys (in \(\upmu \)M):
$$\begin{aligned} Key_A(t,x)=\,\left\{ \begin{array}{ll}1/(1 + Exp[-25*(Mod(x,16) - 1)]) &{} : Mod(x,16) \le 2\\ 1 - 1/(1 + Exp[-25*(Mod(x,16) - 3)]) &{} : otherwise.\end{array}\right. \end{aligned}$$(8.1)$$\begin{aligned} Key_B(t,x)=\,\left\{ \begin{array}{ll}1/(1 + Exp[-25*(Mod(x,16) - 5)]) &{} : Mod(x,16) \le 6\\ 1 - 1/(1 + Exp[-25*(Mod(x,16) - 7)]) &{} : otherwise.\end{array}\right. \end{aligned}$$(8.2)$$\begin{aligned} Key_C(t,x)=\,\left\{ \begin{array}{ll}1/(1 + Exp[-25(Mod(x,16) - 9)]) &{} : mod(x,16) \le 10\\ 1 - 1/(1 + Exp[-25(Mod(x,16) - 11)]) &{} : otherwise.\end{array}\right. \end{aligned}$$(8.3)$$\begin{aligned} Key_D(t,x)=\,\left\{ \begin{array}{ll}1/(1 + Exp[-25(Mod(x,16) - 13)]) &{} : mod(x,16) \le 14\\ 1 - 1/(1 + Exp[-25(Mod(x,16) - 15)]) &{} : otherwise.\end{array}\right. \end{aligned}$$(8.4) -
b.
Clock:
$$\begin{aligned} clk(t,x)= & {} \left\{ \begin{array}{ll}1 \;\upmu {}\text {M} &{} : t < 4000\\ 1 \;\upmu {}\text {M} &{} : Mod(t, clkPeriod) < clkDuty\\ 0 &{} : otherwise. \end{array}\right. \end{aligned}$$(8.5)$$\begin{aligned} \frac{\partial {}clk_{On}(t,x)}{\partial {}t}=\, & {} D \nabla {}^2clk_{On}(t,x)-k_{d}*clk_{On}(t,x)+k_{d}*clk(t,x) -k_{L}*clk_{On}(t,x)*Sg_{nxOn}(t,x) \end{aligned}$$(8.6)$$\begin{aligned} \frac{\partial {}clk_{Off}(t,x)}{\partial {}t}= & {} D\nabla {}^2clk_{Off}(t,x) -k_{d}*clk_{Off}(t,x)+k_{d}*clk(t,x)-k_{L}*clk_{Off} (t,x)*Sg_{OffNx}(t,x) \end{aligned}$$(8.7)
-
a.
-
2.
Communication Stage:
-
a.
Broadcast Modules (in \(\mu \)M):
$$\begin{aligned} Src_A(t,x)= 1 \end{aligned}$$(8.8)$$\begin{aligned} Src_B(t,x)= 1 \end{aligned}$$(8.9)$$\begin{aligned} Src_C(t,x)= 1 \end{aligned}$$(8.10)$$\begin{aligned} Src_D(t,x)= 1 \end{aligned}$$(8.11) -
b.
Broadcast signals, note initial concentration of \(Sig_A\) triggers initial on cell:
$$\begin{aligned} \frac{\partial {}Sig_A(t,x)}{\partial {}t}=\, & {} D\nabla {}^2Sig_A(t,x) +k_{B}Src_A(t,x)Key_A(t,x)Last(t,x)-k_{Bd}Sig_A(t,x)\nonumber \\ Sig_A(t=0,x)= & {} \left\{ \begin{array}{ll}2 \;\upmu {}\text {M} &{} : x_{max}-8<x<x_{max}-0.1\\ 0 &{} \quad: otherwise.\end{array}\right. \end{aligned}$$(8.12)$$\begin{aligned} \frac{\partial {}Sig_B(t,x)}{\partial {}t}= & {} D\nabla {}^2Sig_B(t,x) +k_{B}Src_B(t,x)Key_B(t,x)Last(t,x)-k_{Bd}Sig_B(t,x) \end{aligned}$$(8.13)$$\begin{aligned} \frac{\partial {}Sig_C(t,x)}{\partial {}t}=\, & {} D\nabla {}^2Sig_C(t,x) +k_{B}Src_C(t,x)Key_C(t,x)Last(t,x)-k_{Bd}Sig_C(t,x) \end{aligned}$$(8.14)$$\begin{aligned} \frac{\partial {}Sig_D(t,x)}{\partial {}t}= & {} D\nabla {}^2Sig_D(t,x) +k_{B}Src_D(t,x)Key_D(t,x)Last(t,x)-k_{Bd}Sig_D(t,x) \end{aligned}$$(8.15) -
c.
Receiving and processing left-hand neighbor signal:
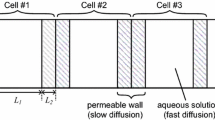
$$\begin{aligned} \frac{\partial {}L_{raw}(t,x)}{\partial {}t}= & {} D\nabla {}^2L_{raw}(t,x) -4k_{d}L_{raw}(t,x)-k_{T}L_{raw}(t,x)Th_l(t,x) +k_{x}Sig_D(t,x)Key_A(t,x)\nonumber \\&+k_{x}Sig_A(t,x)Key_B(t,x) +k_{x}Sig_B(t,x)Key_C(t,x)+k_{x}Sig_C(t,x)Key_D(t,x) \end{aligned}$$(8.16)$$\begin{aligned} \frac{\partial {}Th_l(t,x)}{\partial {}t}= & {} D\nabla {}^2Th_l(t,x)+c_{recTh}k_{p}-k_{d}Th_l(t,x)-k_{T}L_{raw}(t,x)Th_l(t,x) \end{aligned}$$(8.17)$$\begin{aligned} \frac{\partial {}Amp_l(t,x)}{\partial {}t}= & {} D\nabla {}^2Amp_l(t,x)+k_{p}-k_{d}Amp_l(t,x)-k_{L}L_{raw}(t,x)Amp_l(t,x) \end{aligned}$$(8.18)$$\begin{aligned} \frac{\partial {}Lft(t,x)}{\partial {}t}= & {} D\nabla {}^2Lft(t,x)-k_{d}Lft(t,x)+k_{L}L_{raw}(t,x)Amp_l(t,x) \end{aligned}$$(8.19) -
d.
Receiving and processing right-hand neighbor signal:
$$\begin{aligned} \frac{\partial {}R_{raw}(t,x)}{\partial {}t}= & {} D\nabla {}^2R_{raw}(t,x) -4k_{d}R_{raw}(t,x)-k_{T}R_{raw}(t,x)Th_r(t,x)+k_{x}Sig_(t,x)Key_A(t,x)\nonumber \\&+k_{x}Sig_C(t,x)Key_B(t,x) +k_{x}Sig_D(t,x)Key_C(t,x)+k_{x}Sig_A(t,x)Key_D(t,x) \end{aligned}$$(8.20)$$\begin{aligned} \frac{\partial {}Th_r(t,x)}{\partial {}t}= & {} D\nabla {}^2Th_r(t,x)+c_{recTh}k_{p}-k_{d}Th_r(t,x)-k_{T}R_{raw}(t,x)Th_r(t,x) \end{aligned}$$(8.21)$$\begin{aligned} \frac{\partial {}Amp_{rt}(t,x)}{\partial {}t}= & {} D\nabla {}^2Amp_{rt}(t,x)+k_{p}-k_{d}Amp_{rt}(t,x)-k_{L}R_{raw}(t,x)Amp_{rt}(t,x) \end{aligned}$$(8.22)$$\begin{aligned} \frac{\partial {}Rght(t,x)}{\partial {}t}= & {} D\nabla {}^2Rght(t,x)-k_{d}Rght(t,x)+k_{L}R_{raw}(t,x)Amp_{rt}(t,x) \end{aligned}$$(8.23)
-
a.
-
3.
Calculation Stage:
-
a.
Copy left, right and previous time step (pr) signals (multiple gates operate on each)
$$\frac{\partial {}Br_{pr}(t,x)}{\partial {}t}=\,D\nabla {}^2Br_{pr}(t,x)-k_{d}Br_{pr}(t,x)+k_{d}Last(t,x)-k_{T}Br_{pr}(t,x)Off_{pr}(t,x) $$(8.24)$$\frac{\partial {}R_{br}(t,x)}{\partial {}t}=\,D\nabla {}^2R_{br}(t,x)-k_{d}R_{br}(t,x)+k_{d}Rght(t,x)-k_{L}R_{br}(t,x)Sg_{br}(t,x) $$(8.25)$$\frac{\partial {}On_{pr}(t,x)}{\partial {}t}=\,D\nabla {}^2On_{pr}(t,x)-k_{d}On_{pr}(t,x)+k_{d}Last(t,x)-k_{L}On_{pr}(t,x)Sg_{cr}(t,x) $$(8.26)$$\frac{\partial {}R_{cr}(t,x)}{\partial {}t}=\,D\nabla {}^2R_{cr}(t,x)-k_{d}R_{cr}(t,x)+k_{d}Rght(t,x)-k_{L}R_{cr}(t,x)Sg_{nbOff}(t,x) $$(8.27)$$\frac{\partial {}L_{cr}(t,x)}{\partial {}t}=\,D\nabla {}^2L_{cr}(t,x)-k_{d}L_{cr}(t,x)+k_{d}Lft(t,x)-k_{L}L_{cr}(t,x)Sg_{nbOff}(t,x) $$(8.28)$$\frac{\partial {}Off_{pr}(t,x)}{\partial {}t}=\,D\nabla {}^2Off_{pr}(t,x)+k_{p}-k_{d}Off_{pr}(t,x)-_{T}Br_{pr}(t,x)Off_{pr}(t,x)-k_{L}Off_{pr}(t,x)Sg_{br}(t,x) $$(8.29) -
b.
Boolean logic for Br (birth) condition: off to on transition
$$\frac{\partial {}Sg_{br}(t,x)}{\partial {}t}=\,D\nabla {}^2Sg_{br}(t,x)+2k_{p}-k_{d}Sg_{br}(t,x)-k_{L}Off_{pr}(t,x)Sg_{br}(t,x)-k_{L}R_{br}(t,x)Sg_{br}(t,x) $$(8.30)$$\begin{aligned}&\frac{\partial {}Sum_{br}(t,x)}{\partial {}t}=\,D\nabla {}^2Sum_{br}(t,x)-k_{d}Sum_{br}(t,x)+k_{L}Off_{pr}(t,x)Sg_{br}(t,x) +k_{L}R_{br}(t,x)Sg_{br}(t,x)-k_{T}Sum_{br}(t,x)Th_{br}(t,x) \end{aligned}$$(8.31)$$\begin{aligned}&\frac{\partial {}Th_{br}(t,x)}{\partial {}t}=\,D\nabla {}^2Th_{br}(t,x)+1.35k_{p}-k_{d}Th_{br}(t,x)-k_{T}Sum_{br}(t,x)Th_{br}(t,x) \end{aligned}$$(8.32)$$\begin{aligned}&\frac{\partial {}Amp_{br}(t,x)}{\partial {}t}=\,D\nabla {}^2Amp_{br}(t,x)+k_{p}-k_{d}Amp_{br}(t,x)-k_{L}Sum_{br}(t,x)Amp_{br}(t,x) \end{aligned}$$(8.33)$$\begin{aligned}&\frac{\partial {}Br(t,x)}{\partial {}t}=\,D\nabla {}^2Br(t,x)-k_{d}Br(t,x)+k_{L}Sum_{br}(t,x)Amp_{br}(t,x)-k_{L}Br(t,x)Sg_{nx}(t,x) \end{aligned}$$(8.34) -
c.
Boolean logic for no death condition (on and at least one neighbor off): stay on
$$\begin{aligned}&\frac{\partial {}Sg_{nbOff}(t,x)}{\partial {}t}=\,D\nabla {}^2Sg_{nbOff}(t,x)+2k_{p}-k_{d}Sg_{nbOff}(t,x)-k_{L}R_{cr}(t,x)Sg_{nbOff}(t,x)-k_{L}L_{cr}(t,x)Sg_{nbOff}(t,x) \end{aligned}$$(8.35)$$\begin{aligned}&\frac{\partial {}Sum_{nbOff}(t,x)}{\partial {}t}=\,D\nabla {}^2Sum_{nbOff}(t,x)-k_{d}Sum_{nbOff}(t,x)+k_{L}R_{cr}(t,x)Sg_{nbOff}(t,x)+k_{L}L_{cr}(t,x)Sg_{nbOff}(t,x)-k_{T}Sum_{nbOff}(t,x)Th_{nbOff}(t,x) \end{aligned}$$(8.36)$$\begin{aligned}&\frac{\partial {}Th_{nbOff}(t,x)}{\partial {}t}=\,D\nabla {}^2Th_{nbOff}(t,x)+1.35k_{p}-k_{d}Th_{nbOff}(t,x)-k_{T}Sum_{nbOff}(t,x)Th_{nbOff}(t,x) \end{aligned}$$(8.37)$$\begin{aligned}&\frac{\partial {}Amp_{nbOff}(t,x)}{\partial {}t}=\,D\nabla {}^2Amp_{nbOff}(t,x)+k_{p}-k_{d}Amp_{nbOff}(t,x)-k_{L}Th_{nbOff}(t,x)Amp_{nbOff}(t,x) \end{aligned}$$(8.38)$$\begin{aligned}&\frac{\partial {}nbrOff(t,x)}{\partial {}t}=\,D\nabla {}^2nbrOff(t,x)-k_{d}nbrOff(t,x)+k_{L}Th_{nbOff}(t,x)Amp_{nbOff}y(t,x)-k_{L}nbrOff(t,x)Sg_{cr}(t,x) \end{aligned}$$(8.39)$$\begin{aligned}&\frac{\partial {}Sg_{cr}(t,x)}{\partial {}t}=\,D\nabla {}^2Sg_{cr}(t,x)+2k_{p}-k_{d}Sg_{cr}(t,x)-k_{L}On_{pr}(t,x)Sg_{cr}(t,x)-k_{L}nbrOff(t,x)Sg_{cr}(t,x) \end{aligned}$$(8.40)$$\begin{aligned}&\frac{\partial {}Sum_{cr}(t,x)}{\partial {}t}=\,D\nabla {}^2Sum_{cr}(t,x)-k_{d}Sum_{cr}(t,x)+k_{L}On_{pr}(t,x)Sg_{cr}(t,x)+k_{L}nbrOff(t,x)Sg_{cr}(t,x)-k_{T}Sum_{cr}(t,x)Th_{cr}(t,x) \end{aligned}$$(8.41)$$\begin{aligned}&\frac{\partial {}Th_{cr}(t,x)}{\partial {}t}=\,D\nabla {}^2Th_{cr}(t,x)+1.35k_{p}-k_{d}Th_{cr}(t,x)-k_{T}Sum_{cr}(t,x)Th_{cr}(t,x) \end{aligned}$$(8.42)$$\begin{aligned}&\frac{\partial {}Amp_{cr}(t,x)}{\partial {}t}=\,D\nabla {}^2Amp_{cr}(t,x)+k_{p}-k_{d}Amp_{cr}(t,x)-k_{L}Sum_{cr}(t,x)Amp_{cr}(t,x) \end{aligned}$$(8.43)$$\begin{aligned}&\frac{\partial {}Live(t,x)}{\partial {}t}=\,D\nabla {}^2Live(t,x)-k_{d}Live(t,x)+k_{L}Sum_{cr}(t,x)Amp_{cr}(t,x)-k_{L}Live(t,x)Sg_{nx}(t,x) \end{aligned}$$(8.44) -
d.
Boolean logic to determine if next state is on
$$\begin{aligned}&\frac{\partial {}Sg_{nx}(t,x)}{\partial {}t}=\,D\nabla {}^2Sg_{nx}(t,x)+2k_{p}-k_{d}Sg_{nx}(t,x)-k_{L}Live(t,x)Sg_{nx}(t,x)-k_{L}Br(t,x)Sg_{nx}(t,x) \end{aligned}$$(8.45)$$\begin{aligned}&\frac{\partial {}Sum_{nx}(t,x)}{\partial {}t}=\,D\nabla {}^2Sum_{nx}(t,x)-k_{d}Sum_{nx}(t,x)+k_{L}Live(t,x)Sg_{nx}(t,x)+k_{L}Br(t,x)Sg_{nx}(t,x)-k_{T}Sum_{nx}(t,x)Th_{nx}(t,x) \end{aligned}$$(8.46)$$\begin{aligned}&\frac{\partial {}Th_{nx}(t,x)}{\partial {}t}=\,D\nabla {}^2Th_{nx}(t,x)+0.65k_{p}-k_{d}Th_{nx}(t,x)-k_{T}Sum_{nx}(t,x)Th_{nx}(t,x) \end{aligned}$$(8.47)$$\begin{aligned}&\frac{\partial {}Amp_{nx}(t,x)}{\partial {}t}=\,D\nabla {}^2Amp_{nx}(t,x)+k_{p}-k_{d}Amp_{nx}(t,x)-k_{L}Sum_{nx}(t,x)Amp_{nx}(t,x) \end{aligned}$$(8.48)$$\begin{aligned}&\frac{\partial {}Nx(t,x)}{\partial {}t}=\,D\nabla {}^2Nx(t,x)-k_{d}Nx(t,x)+k_{L}Sum_{nx}(t,x)Amp_{nx}(t,x) \end{aligned}$$(8.49)$$\begin{aligned}&\frac{\partial {}On_{nx}(t,x)}{\partial {}t}=\,D\nabla {}^2On_{nx}(t,x)-k_{d}On_{nx}(t,x)+k_{d}Nx(t,x)-k_{L}On_{nx}(t,x)Sg_{nxOn}(t,x) \end{aligned}$$(8.50) -
e.
Boolean logic to determine if next state is off
$$\begin{aligned}&\frac{\partial {}Nt_{nx}(t,x)}{\partial {}t}=\,D\nabla {}^2Nt_{nx}(t,x)-k_{d}Nt_{nx}(t,x)+k_{d}Nx(t,x)-k_{T}Nt_{nx}(t,x)Off_{nx}(t,x) \end{aligned}$$(8.51)$$\begin{aligned}&\frac{\partial {}Off_{nx}(t,x)}{\partial {}t}=\,D\nabla {}^2Off_{nx}(t,x)+k_{p}-k_{d}Off_{nx}(t,x)-k_{T}Nt_{nx}(t,x)Off_{nx}(t,x)-k_{L}Off_{nx}(t,x)Sg_{OffNx}(t,x) \end{aligned}$$(8.52) -
f.
Clocked synchronization gates
$$\begin{aligned}&\frac{\partial {}Sg_{nxOn}(t,x)}{\partial {}t}=\,D\nabla {}^2Sg_{nxOn}(t,x)+2k_{p}-k_{d}Sg_{nxOn}(t,x)-k_{L}On_{nx}(t,x)Sg_{nxOn}(t,x)-k_{L}clk_{On}(t,x)Sg_{nxOn}(t,x) \end{aligned}$$(8.53)$$\begin{aligned}&\frac{\partial {}On_{nx}Sum(t,x)}{\partial {}t}=\,D\nabla {}^2On_{nx}Sum(t,x)-k_{d}On_{nx}Sum(t,x)+k_{L}On_{nx}(t,x)Sg_{nxOn}(t,x)+k_{L}clk_{On}(t,x)Sg_{nxOn}(t,x)-k_{T}On_{nx}Sum(t,x)On_{nx}Th(t,x) \end{aligned}$$(8.54)$$\begin{aligned}&\frac{\partial {}On_{nx}Th(t,x)}{\partial {}t}=\,D\nabla {}^2On_{nx}Th(t,x)+1.35k_{p}-k_{d}On_{nx}Th(t,x)-k_{T}On_{nx}Sum(t,x)On_{nx}Th(t,x) \end{aligned}$$(8.55)$$\begin{aligned}&\frac{\partial {}On_{nx}Amp(t,x)}{\partial {}t}=\,D\nabla {}^2On_{nx}Amp(t,x)+k_{p}-k_{d}On_{nx}Amp(t,x)-k_{L}On_{nx}Sum(t,x)On_{nx}Amp(t,x) \end{aligned}$$(8.56)$$\begin{aligned}&\frac{\partial {}Set_{Bfr}(t,x)}{\partial {}t}=\,D\nabla {}^2Set_{Bfr}(t,x)-k_{d}Set_{Bfr}(t,x)+k_{L}On_{nx}Sum(t,x)On_{nx}Amp(t,x) \end{aligned}$$(8.57)$$\begin{aligned}&\frac{\partial {}Sg_{OffNx}(t,x)}{\partial {}t}=\,D\nabla {}^2Sg_{OffNx}(t,x)+2k_{p}-k_{d}Sg_{OffNx}(t,x)-k_{L}Off_{nx}(t,x)Sg_{OffNx}(t,x)-k_{L}clk_{Off}(t,x)Sg_{OffNx}(t,x) \end{aligned}$$(8.58)$$\begin{aligned}&\frac{\partial {}Sum_{OffNx}(t,x)}{\partial {}t}=\,D\nabla {}^2Sum_{OffNx}(t,x)-k_{d}Sum_{OffNx}(t,x)+k_{L}Off_{nx}(t,x)Sg_{OffNx}(t,x)+k_{L}clk_{Off}(t,x)Sg_{OffNx}(t,x)-k_{T}Sum_{OffNx}(t,x)Th_{OffNx}(t,x) \end{aligned}$$(8.59)$$\begin{aligned}&\frac{\partial {}Th_{OffNx}(t,x)}{\partial {}t}=\,D\nabla {}^2Th_{OffNx}(t,x)+1.35k_{p}-k_{d}Th_{OffNx}(t,x)-k_{T}Sum_{OffNx}(t,x)Th_{OffNx}(t,x) \end{aligned}$$(8.60)$$\begin{aligned}&\frac{\partial {}Amp_{OffNx}(t,x)}{\partial {}t}=\,D\nabla {}^2Amp_{OffNx}(t,x)+k_{p}-k_{d}Amp_{OffNx}(t,x)-k_{L}Sum_{OffNx}(t,x)Amp_{OffNx}(t,x) \end{aligned}$$(8.61)$$\begin{aligned}&\frac{\partial {}Res_{Bfr}(t,x)}{\partial {}t}=\,D\nabla {}^2Res_{Bfr}(t,x)-k_{d}Res_{Bfr}(t,x)+k_{L}Sum_{OffNx}(t,x)Amp_{OffNx}(t,x) \end{aligned}$$(8.62)
-
a.
-
4.
Storage stage
-
a.
Copies of Set/Res signals
$$\begin{aligned}&\frac{\partial {}Set(t,x)}{\partial {}t}=\,D\nabla {}^2Set(t,x)-k_{d}Set(t,x)+k_{d}Set_{Bfr}(t,x)-k_{L}Set(t,x)N1Sg(t,x) \end{aligned}$$(8.63)$$\begin{aligned}&\frac{\partial {}Res(t,x)}{\partial {}t}=\,D\nabla {}^2Res(t,x)-k_{d}Res(t,x)+k_{d}Res_{Bfr}(t,x)-k_{L}Res(t,x)N2Sg(t,x) \end{aligned}$$(8.64) -
b.
Flip-flop module
$$\begin{aligned}&\frac{\partial {}ffBfrd(t,x)}{\partial {}t}=\,D\nabla {}^2ffBfrd(t,x)-k_{d}ffBfrd(t,x)+k_{L}N2Th(t,x)N2(t,x) \end{aligned}$$(8.65)$$\begin{aligned}&\frac{\partial {}ffFbackNot(t,x)}{\partial {}t}=\,D\nabla {}^2ffFbackNot(t,x)-k_{d}ffFbackNot(t,x)+k_{L}N1Th(t,x)N1(t,x)-k_{L}ffFbackNot(t,x)N2Sg(t,x) \end{aligned}$$(8.66)$$\begin{aligned}&\frac{\partial {}N1Th(t,x)}{\partial {}t}=\,D\nabla {}^2N1Th(t,x)+0.65k_{p}-k_{d}N1Th(t,x)-k_{T}N1Sum(t,x)N1Th(t,x) \end{aligned}$$(8.67)$$\begin{aligned}&\frac{\partial {}N2Sum(t,x)}{\partial {}t}=\,D\nabla {}^2N2Sum(t,x)-k_{d}N2Sum(t,x)+k_{L}Res(t,x)N2Sg(t,x)+k_{L}ffFbackNot(t,x)N2Sg(t,x)-k_{T}N2Sum(t,x)N2Th(t,x) \end{aligned}$$(8.68)$$\begin{aligned}&\frac{\partial {}ffFback(t,x)}{\partial {}t}=\,D\nabla {}^2ffFback(t,x)-k_{d}ffFback(t,x)+k_{d}ffBfrd(t,x)-k_{L}ffFback(t,x)N1Sg(t,x) \end{aligned}$$(8.69)$$\begin{aligned}&\frac{\partial {}N1Sg(t,x)}{\partial {}t}=\,D\nabla {}^2N1Sg(t,x)+2k_{p}-k_{d}N1Sg(t,x)-k_{L}Set(t,x)N1Sg(t,x)-k_{L}ffFback(t,x)N1Sg(t,x) \end{aligned}$$(8.70)$$\begin{aligned}&\frac{\partial {}N1Sum(t,x)}{\partial {}t}=\,D\nabla {}^2N1Sum(t,x)-k_{d}N1Sum(t,x)+k_{L}Set(t,x)N1Sg(t,x)+k_{L}ffFback(t,x)N1Sg(t,x)-k_{T}N1Sum(t,x)N1Th(t,x) \end{aligned}$$(8.71)$$\begin{aligned}&\frac{\partial {}N1(t,x)}{\partial {}t}=\,D\nabla {}^2N1(t,x)+k_{p}-k_{d}N1(t,x)-k_{L}N1Th(t,x)N1(t,x) \end{aligned}$$(8.72)$$\begin{aligned}&\frac{\partial {}N2Sg(t,x)}{\partial {}t}=\,D\nabla {}^2N2Sg(t,x)+2k_{p}-k_{d}N2Sg(t,x)-k_{L}Res(t,x)N2Sg(t,x)-k_{L}ffFbackNot(t,x)N2Sg(t,x) \end{aligned}$$(8.73)$$\begin{aligned}&\frac{\partial {}N2Th(t,x)}{\partial {}t}=\,D\nabla {}^2N2Th(t,x)+0.65k_{p}-k_{d}N2Th(t,x)-k_{T}N2Sum(t,x)N2Th(t,x) \end{aligned}$$(8.74)$$\begin{aligned}&\frac{\partial {}N2(t,x)}{\partial {}t}=\,D\nabla {}^2N2(t,x)+k_{p}-k_{d}N2(t,x)-k_{L}N2Th(t,x)N2(t,x) \end{aligned}$$(8.75) -
c.
Stored state
$$\begin{aligned} \frac{\partial {}Last(t,x)}{\partial {}t}=\,D\nabla {}^2Last(t,x)-k_{d}Last(t,x)+k_{d}ffBfrd(t,x) \end{aligned}$$(8.76)
-
a.
Rights and permissions
About this article
Cite this article
Scalise, D., Schulman, R. Emulating cellular automata in chemical reaction–diffusion networks. Nat Comput 15, 197–214 (2016). https://doi.org/10.1007/s11047-015-9503-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11047-015-9503-8