Abstract
Introduction
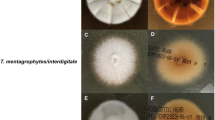
The global spread of Trichophyton indotineae presents a pressing challenge in dermatophytosis management. This systematic review explores the current landscape of T. indotineae infections, emphasizing resistance patterns, susceptibility testing, mutational analysis, and management strategies.
Methods
A literature search was conducted in November 2023 using Embase, PubMed, Scopus, and Web of Science databases. Inclusion criteria covered clinical trials, observational studies, case series, or case reports with T. indotineae diagnosis through molecular methods. Reports on resistance mechanisms, antifungal susceptibility testing, and management were used for data extraction.
Results and Discussion
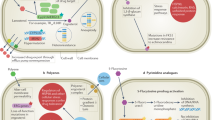
A total of 1148 articles were identified through the systematic search process, with 45 meeting the inclusion criteria. The global spread of T. indotineae is evident, with cases reported in numerous new countries in 2023. Tentative epidemiological cut-off values (ECOFFs) suggested by several groups provide insights into the likelihood of clinical resistance. The presence of specific mutations, particularly Phe397Leu, correlate with higher minimum inhibitory concentrations (MICs), indicating potential clinical resistance. Azole resistance has also been reported and investigated in T. indotineae, and is a growing concern. Nevertheless, itraconazole continues to be an alternative therapy. Recommendations for management include oral or combination therapies and individualized approaches based on mutational analysis and susceptibility testing.
Conclusion
Trichophyton indotineae poses a complex clinical scenario, necessitating enhanced surveillance, improved diagnostics, and cautious antifungal use. The absence of established clinical breakpoints for dermatophytes underscores the need for further research in this challenging field.



Similar content being viewed by others
Data Availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
Nenoff P, Uhrlaß S, Verma SB, Panda S. Trichophyton mentagrophytes ITS genotype VIII and Trichophyton indotineae: A terminological maze, or is it? Indian J Dermatol Venereol Leprol. 2022;88:586–9.
Uhrlaß S, Verma SB, Gräser Y, Rezaei-Matehkolaei A, Hatami M, Schaller M, et al. Trichophyton indotineae—an emerging pathogen causing recalcitrant dermatophytoses in India and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8:757.
Sacheli R, Hayette MP. Antifungal resistance in dermatophytes: genetic considerations, clinical presentations and alternative therapies. J Fungi (Basel). 2021;7:983.
Jabet A, Normand AC, Brun S, Dannaoui E, Bachmeyer C, Piarroux R, et al. Trichophyton indotineae, from epidemiology to therapeutic. J Mycol Med. 2023;33:101383.
Gupta AK, Polla Ravi S, Wang T, Cooper EA, Lincoln SA, Foreman HC, et al. Antifungal resistance, susceptibility testing and treatment of recalcitrant dermatophytosis caused by Trichophyton indotineae: a North American perspective on management. Am J Clin Dermatol. 2023;24:927–38.
Chowdhary A, Singh A, Kaur A, Khurana A. The emergence and worldwide spread of the species Trichophyton indotineae causing difficult-to-treat dermatophytosis: a new challenge in the management of dermatophytosis. PLoS Pathog. 2022;18:e1010795.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;2021:n71.
OCEBM Levels of Evidence Working Group. The Oxford 2011 Levels of Evidence. 2011 [cited 2023 Dec 27]. Available from: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence.
Jia S, Long X, Hu W, Zhu J, Jiang Y, Ahmed S, et al. The epidemic of the multiresistant dermatophyte Trichophyton indotineae has reached China. Front Immunol. 2023;13:1113065.
Messina F, Santiso G, Romero M, Bonifaz A, Fernandez M, Marin E. First case report of tinea corporis caused by Trichophyton indotineae in Latin America. Med Mycol Case Rep. 2023;41:48–51.
Kong X, Song G, Mei H, Zheng H, Tang C, de Hoog S, et al. The domestic isolation of terbinafine- and itraconazole-resistant Trichophyton indotineae in Chinese Mainland. Mycopathologia. 2023;188:383–93.
Bortoluzzi P, Prigitano A, Sechi A, Boneschi V, Germiniasi F, Esposto MC, et al. Report of terbinafine resistant Trichophyton spp. in Italy: Clinical presentations, molecular identification, antifungal susceptibility testing and mutations in the squalene epoxidase gene. Mycoses. 2023;66:680–7.
Dashti Y, Alobaid K, Al-Rashidi S, Dashti M, AbdulMoneim MH, Al-Enezi M, et al. Autochthonous case of Trichophyton indotineae in Kuwait. J Med Mycol. 2023;33:101432.
Villa-Gonzalez JM, Pascual Ares M, López-Soria LM, Gonzalez-Hermosa MR, Gardeazabal García J, Lasa EO. Extensive tinea corporis caused by Trichophyton indotineae: report of a case in Spain. J Eur Acad Dermatol Venereol. 2023;38:e22–3.
Durdu M, Kandemir H, Karakoyun AS, Ilkit M, Tang C, de Hoog S. First Terbinafine-resistant Trichophyton indotineae isolates with Phe397Leu and/or Thr414His mutations in Turkey. Mycopathologia. 2023;188:2.
Cañete-Gibas CF, Mele J, Patterson HP, Sanders CJ, Ferrer D, Garcia V, et al. Terbinafine-resistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol. 2023;61:e0056223.
Caplan AS, Chaturvedi S, Zhu Y, Todd GC, Yin L, Lopez A, et al. Notes from the field: first reported U.S. cases of Tinea caused by Trichophyton indotineae—New York City, December 2021–March 2023. Morb Mortal Wkly Rep. 2023;72:536.
Gupta AK, Venkataraman M, Hall DC, Cooper EA, Summerbell RC. The emergence of Trichophyton indotineae: implications for clinical practice. Int J Dermatol. 2023;62:857–61.
Caplan AS, Zakhem GA, Pomeranz MK. Trichophyton mentagrophytes internal transcribed spacer genotype VIII. JAMA Dermatol. 2023;159:1130.
Verma SB, Panda S, Nenoff P, Singal A, Rudramuruthy SM, Uhrlass S, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: I. Epidemiology, risk factors and clinical features. Indian J Dermatol Venereol Leprol. 2021;87:154–75.
Mukherjee PK, Leidich SD, Isham N, Leitner I, Ryder NS, Ghannoum MA. Clinical Trichophyton rubrum strain exhibiting primary resistance to terbinafine. Antimicrob Agents Chemother. 2003;47:82–6.
Iyer KR, Revie NM, Fu C, Robbins N, Cowen LE. Treatment strategies for cryptococcal infection: challenges, advances and future outlook. Nat Rev Microbiol. 2021;19(7):454–66.
Fattahi A, Shirvani F, Ayatollahi A, Rezaei-Matehkolaei A, Badali H, Lotfali E, et al. Multidrug-resistant Trichophyton mentagrophytes genotype VIII in an Iranian family with generalized dermatophytosis: report of four cases and review of literature. Int J Dermatol. 2021;60:686–92.
Gueneau R, Joannard B, Haddad N, Alby F, Jullien V, Schlatter J, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, successfully treated with topical voriconazole. Int J Antimicrob Agents. 2022;60:106677.
Nenoff P, Verma SB, Ebert A, Süß A, Fischer E, Auerswald E, et al. Spread of terbinafine-resistant Trichophyton mentagrophytes type VIII (India) in Germany—“The tip of the iceberg?” J Fungi. 2020;6:1–20.
Dellière S, Joannard B, Benderdouche M, Mingui A, Gits-Muselli M, Hamane S, et al. Emergence of difficult-to-treat tinea corporis caused by Trichophyton mentagrophytes complex isolates, Paris, France. Emerg Infect Dis. 2022;28:224–8.
Dogra S, Shaw D, Rudramurthy SM. Antifungal drug susceptibility testing of dermatophytes: laboratory findings to clinical implications. Indian Dermatol Online J. 2019;10:225.
Astvad KMT, Hare RK, Jørgensen KM, Saunte DML, Thomsen PK, Arendrup MC. Increasing terbinafine resistance in Danish Trichophyton isolates 2019–2020. J Fungi. 2022;8:150.
Arendrup MC, Jørgensen KM, Guinea J, Lagrou K, Chryssanthou E, Hayette MP, et al. Multicentre validation of a EUCAST method for the antifungal susceptibility testing of microconidia-forming dermatophytes. J Antimicrob Chemother. 2020;75:1807–19.
Russo G, Toutous Trellu L, Fontao L, Ninet B. Towards an early clinical and biological resistance detection in dermatophytosis: about 2 cases of Trichophyton indotineae. J Fungi. 2023;9:733.
Shen JJ, Arendrup MC, Verma S, Saunte DML. The emerging terbinafine-resistant trichophyton epidemic: What is the role of antifungal susceptibility testing? Dermatology. 2022;238:60–79.
Arendrup MC, Jørgensen KM, Guinea J, Lagrou K, Chryssanthou E, Hayette MP, et al. Comment on: Multicentre validation of a EUCAST method for the antifungal susceptibility testing of microconidia-forming dermatophytes. J Antimicrob Chemother. 2022;77:1212–3.
Blanchard G, Amarov B, Fratti M, Salamin K, Bontems O, Chang YT, et al. Reliable and rapid identification of terbinafine resistance in dermatophytic nail and skin infections. J Eur Acad Dermatol Venereol. 2023;37:2080–9.
Ebert A, Monod M, Salamin K, Burmester A, Uhrlaß S, Wiegand C, et al. Alarming India-wide phenomenon of antifungal resistance in dermatophytes: a multicentre study. Mycoses. 2020;63:717–28.
Kong X, Tang C, Singh A, Ahmed SA, Al-Hatmi AMS, Chowdhary A, et al. Antifungal susceptibility and mutations in the squalene epoxidase gene in dermatophytes of the Trichophyton mentagrophytes species complex. Antimicrob Agents Chemother. 2021;65:e0005621.
Martinez-Rossi NM, Peres NTA, Bitencourt TA, Martins MP, Rossi A. State-of-the-art dermatophyte infections: epidemiology aspects, pathophysiology, and resistance mechanisms. J Fungi (Basel). 2021;7:629.
Brasch J, Gräser Y, Beck-Jendroscheck V, Voss K, Torz K, Walther G, et al. “Indian” strains of Trichophyton mentagrophytes with reduced itraconazole susceptibility in Germany. JDDG J German Soc Dermatol. 2021;19:1723–7.
Gupta AK, Talukder M, Carviel JL, Cooper EA, Piguet V. Combatting antifungal resistance: paradigm shift in the diagnosis and management of onychomycosis and dermatomycosis. J Eur Acad Dermatol Venereol. 2023;37:1706–17.
Yamada T, Yaguchi T, Tamura T, Pich C, Salamin K, Feuermann M, et al. Itraconazole resistance of Trichophyton rubrum mediated by the ABC transporter TruMDR2. Mycoses. 2021;64:936–46.
Monod M, Feuermann M, Salamin K, Fratti M, Makino M, Alshahni MM, et al. Trichophyton rubrum azole resistance mediated by a new ABC transporter, TruMDR3. Antimicrob Agents Chemother. 2019;63:e00863-e919.
Yamada T, Yaguchi T, Maeda M, Alshahni MM, Salamin K, Guenova E, et al. Gene amplification of CYP51B: a new mechanism of resistance to azole compounds in Trichophyton indotineae. Antimicrob Agents Chemother. 2022;66:e0005922.
Burmester A, Hipler UC, Elsner P, Wiegand C. Point mutations in the squalene epoxidase erg1 and sterol 14-α demethylase erg11 gene of T. indotineae isolates indicate that the resistant mutant strains evolved independently. Mycoses. 2022;65:97–102.
Shafiei M, Peyton L, Hashemzadeh M, Foroumadi A. History of the development of antifungal azoles: a review on structures, SAR, and mechanism of action. Bioorg Chem. 2020;104:104240.
Yamada T, Nojo H, Kano R. Long amplification PCR (LA-PCR) detection of azole resistant Trichophyton indotineae. Mycopathologia. 2023;188:1085–8.
Yamada T, Maeda M, Nagai H, Salamin K, Chang Y-T, Guenova E, et al. Two different types of tandem sequences mediate the overexpression of TinCYP51B in azole-resistant Trichophyton indotineae. Antimicrob Agents Chemother. 2023;67:e0093323.
Crotti S, Cruciani D, Spina S, Piscioneri V, Natalini Y, Pezzotti G, et al. A terbinafine sensitive Trichophyton indotineae strain in Italy: the first clinical case of tinea corporis and onychomycosis. J Fungi. 2023;9:865.
Sardana K, Khurana A, Panesar S, Singh A. Super bioavailable itraconazole and its place and relevance in recalcitrant dermatophytosis: revisiting skin levels of itraconazole and minimum inhibitory concentration data. Indian Dermatol Online J. 2021;12:1–1.
Thakur R, Kushwaha P, Kalsi AS. Tinea universalis due to Trichophyton indotineae in an adult male. Indian J Med Microbiol. 2023;46:100476.
Song G, Kong X, Li X, Liu W, Liang G. Prior selection of itraconazole in the treatment of recalcitrant Trichophyton indotineae infection: real-world results from retrospective analysis. Mycoses. 2023;67:e13663.
Khurana A, Agarwal A, Agrawal D, Panesar S, Ghadlinge M, Sardana K, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol. 2022;158:1269–78.
Jabet A, Brun S, Normand AC, Imbert S, Akhoundi M, Dannaoui E, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae. France Emerg Infect Dis. 2022;28:229–33.
Harada K, Maeda T, Kano R. Extensive tinea corporis caused by a virulent strain of Trichophyton interdigitale. J Dermatol. 2021;48:e190–1.
Kolarczyková D, Lysková P, Švarcová M, Kuklová I, Dobiáš R, Mallátová N, et al. Terbinafine resistance in Trichophyton mentagrophytes and Trichophyton rubrum in the Czech Republic: a prospective multicentric study. Mycoses. 2024;67:e13708.
Singh S, Subba N, Tilak R. Efficacy of terbinafine and itraconazole in different doses and in combination in the treatment of tinea infection: a randomized controlled parallel group open labeled trial with clinico-mycological correlation. Indian J Dermatol. 2020;65:284.
Ravindra Babu P, Pravin AJS, Deshmukh G, Dhoot D, Samant A, Kotak B. Efficacy and safety of terbinafine 500 mg once daily in patients with dermatophytosis. Indian J Dermatol. 2017;62:395.
Xie W, Kong X, Zheng H, Mei H, Ge N, Hu S, et al. Rapid Emergence of Recalcitrant Dermatophytosis by a Cluster of Multidrug-Resistant Trichophyton indotineae, China. Br J Dermatol [Internet]. 2024 [cited 2024 Jan 8]; Available from: https://pubmed-ncbi-nlm-nih-gov.myaccess.library.utoronto.ca/38180270/.
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AKG, SPR, TW, and AS have no conflict of interest to declare. WLB is an employee of Bako Diagnostics (Alpharetta, GA, USA).
Additional information
Handling Editor: Vishnu Chaturvedi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gupta, A.K., Polla Ravi, S., Wang, T. et al. Mapping the Global Spread of T. indotineae: An Update on Antifungal Resistance, Mutations, and Strategies for Effective Management. Mycopathologia 189, 45 (2024). https://doi.org/10.1007/s11046-024-00856-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11046-024-00856-z