Abstract
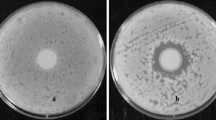
Pseudomonas aeruginosa and Aspergillus fumigatus are major microbes in cystic fibrosis (CF). We reported non-mucoid P. aeruginosa isolates more inhibitory to A. fumigatus than mucoid ones. Another CF P. aeruginosa phenotype, small colony variants (SCVs), is an unknown factor in intermicrobial competition with A. fumigatus. Clinical SCV isolates and reference CF non-mucoid isolate (Pa10, producing normal-sized colonies) were compared. Live cells of P. aeruginosa or filtrates from P. aeruginosa planktonic or biofilm cultures were co-incubated with A. fumigatus growing under conditions allowing biofilm formation or with preformed biofilm. Metabolic activity of A. fumigatus biofilm was then measured. When necessary, assays were done after adjustment for growth differences by adding fresh medium to the planktonic culture filtrate. Pyoverdine determinations were performed spectrophotometrically on the planktonic culture filtrates. In all experimental conditions (live cells and planktonic or biofilm culture filtrates of P. aeruginosa versus A. fumigatus biofilm formation or preformed biofilm), three SCV isolates were less inhibitory than Pa10, two equal or more inhibitory. Adjusting planktonic culture filtrates for growth differences showed SCV inhibition differences variably related to growth or deficient inhibitor production. Studies suggested the principal P. aeruginosa inhibitor to be pyoverdine. SCV isolates appear heterogeneous in their capacity to inhibit A. fumigatus biofilm. SCV isolates can be important in the CF microbiome, because they are capable of intermicrobial inhibition.



Similar content being viewed by others
References
Shoseyov D, Brownlee KG, Conway SP, et al. Aspergillus bronchitis in cystic fibrosis. Chest. 2006;130:222–6.
Amin R, Dupuis A, Aaron SD, et al. The effect of chronic infection with Aspergillus fumigatus on lung function and hospitalization in patients with cystic fibrosis. Chest. 2010;137:171–6.
Mangan A. Interactions between some aural Aspergillus species and bacteria. J Gen Microbiol. 1969;58:261–6.
Blyth W, Forey A. The influence of respiratory bacteria and their biochemical fractions on Aspergillus fumigatus. Sabouraudia. 1971;9:273–82.
Kerr JR, Taylor GW, Rutman A, et al. Pseudomonas aeruginosa pyocyanin and 1-hydroxyphenazine inhibit fungal growth. J Clin Pathol. 1999;52:385–7.
Briard B, Bomme P, Lechner BE, et al. Pseudomonas aeruginosa manipulates redox and iron homeostasis of its microbiota partner Aspergillus fumigatus via phenazines. Sci Rep. 2015;5:8220.
Briard B, Rasoldier V, Bomme P, et al. Dirhamnolipids secreted from Pseudomonas aeruginosa modify anjpegungal susceptibility of Aspergillus fumigatus by inhibiting β1,3glucan synthase activity. ISME J. 2017;11:1578–91.
Sass G, Nazik H, Penner J, et al. Studies of Pseudomonas aeruginosa mutants indicate pyoverdine as the central factor in inhibition of Aspergillus fumigatus biofilm. Infectious Disease Week, San Diego, 2017, Abstr.64764.
Secor PR, Sass G, Nazik H, et al. Effect of acute predation with bacteriophage on intermicrobial aggression by Pseudomonas aeruginosa. PLoS ONE. 2017;12:e0179659.
Williams HD, Davies JC. Basic science for the chest physician: Pseudomonas aeruginosa and the cystic fibrosis airway. Thorax. 2012;67:465–7.
Folkesson A, Jelsbak L, Yang L, et al. Adaptation of Pseudomonas aeruginosa to the cystic fibrosis airway: an evolutionary perspective. Nat Rev Microbiol. 2012;10:841–51.
Ferreira JAG, Penner JC, Moss RB, et al. Inhibition of Aspergillus fumigatus and its biofilm by Pseudomonas aeruginosa is dependent on the source, phenotype and growth conditions of the bacterium. PLoS ONE. 2015;10:e0134692.
Shirazi F, Ferreira JAG, Stevens DA, et al. Biofilm filtrates of Pseudomonas aeruginosa strains isolated from cystic fibrosis patients inhibit preformed Aspergillus fumigatus biofilms via apoptosis. PLoS ONE. 2016;11:e0150155.
Haussler S, Tummler B, Weisbrot H, et al. Small colony variants of Pseudomonas aeruginosa in cystic fibrosis. Clin Infect Dis. 1999;29:621–5.
Evans TJ. Small colony variants of Pseudomonas aeruginosa in chronic bacterial infection of the lung in cystic fibrosis. Future Microbiol. 2015;10:231–9.
Hogardt M, Heesemann J. Adaptation of Pseudomonas aeruginosa during persistence in the cystic fibrosis lung. Int J Med Microbiol. 2010;300:557–62.
Thomassen MJ, Demko CA, Boxerbaum B, et al. Multiple isolates of Pseudomonas aeruginosa with differing antimicrobial susceptibility patterns from patients with cystic fibrosis. J Infect Dis. 1979;140:873–80.
Schneider M, Muhlemann K, Droz S, et al. Clinical characteristics associated with isolation of small-colony variants of Staphylococcus aureus and Pseudomonas aeruginosa from respiratory secretions of patients with cystic fibrosis. J Clin Microbiol. 2008;46:1832–4.
Cao H, Krishnan G, Goumnerov B, et al. A quorum sensing-associated virulence gene of Pseudomonas aeruginosa encodes a LysR-like transcription regulator with a unique self-regulatory mechanism. Proc Natl Acad Sci USA. 2001;98:14613–8.
Mayer-Hamblett N, Ramsey BW, Kulasekara HD, et al. Pseudomonas aeruginosa phenotypes associated with eradication failure in children with cystic fibrosis. Clin Infect Dis. 2014;59:624–31.
O’Toole GA, Kolter R. Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development. Mol Microbiol. 1998;30:295–304.
Lee DG, Urbach JM, Wu G, et al. Genomic analysis reveals that Pseudomonas aeruginosa virulence is combinatorial. Genome Biol. 2006;7:R90.
Liberati NT, Urbach JM, Miyata S, et al. An ordered, nonredundant library of Pseudomonas aeruginosa strain PA14 transposon insertion mutants. Proc Natl Acad Sci USA. 2006;103:2833–8.
Denning DW, Clemons KV, Hanson LH, et al. Restriction endonuclease analysis of total cellular DNA of Aspergillus fumigatus isolates of geographically and epidemiologically diverse origin. J Infect Dis. 1990;162:1151–8.
Denning DW, Stevens DA. Efficacy of cilofungin alone and in combination with amphotericin B in a murine model of disseminated aspergillosis. Antimicrob Agents Chemother. 1991;35:1329–33.
Anand R, Clemons KV, Stevens DA. Effect of anaerobiasis or hypoxia on Pseudomonas aeruginosa inhibition of Aspergillus fumigatus biofilm. Arch Microbiol. 2017;199:881–90.
Sabra W, Haddad AM, Zeng AP. Comparative physiologic study of the wild type and the small colony variant of Pseudomonas aeruginosa 20265 under controlled growth conditions. World J Microbiol Biotechnol. 2014;30:1027–36.
Kahl B, Herrmann M, Everding AS, et al. Persistent infection with small colony variant strains of Staphylococcus aureus in patients with cystic fibrosis. J Infect Dis. 1998;177:1023–9.
Besier S, Smaczny C, von Mallinckrodt C, et al. Prevalence and clinical significance of Staphylococcus aureus small-colony variants in cystic fibrosis lung disease. J Clin Microbiol. 2007;45:168–72.
Wolter DJ, Emerson JC, McNamara S, et al. Staphylococcus aureus small-colony variants are independently associated with worse lung disease in children with cystic fibrosis. Clin Infect Dis. 2013;57:384–91.
Garcia LG, Lemaire S, Kahl BC, et al. Antibiotic activity against small-colony variants of Staphylococcus aureus: review of in vitro, animal and clinical data. J Antimicrob Chemother. 2013;68:1455–64.
Malone JG. Role of small colony variants in persistence of Pseudomonas aeruginosa infections in cystic fibrosis lungs. Infect Drug Resist. 2015;8:237–47.
Lynch SV, Bruce KD. The cystic fibrosis airway microbiome. Cold Spring Harb Perspect Med. 2013;3:a009738.
Manavathu EK, Vager DL, Vazquez JA. Isolation and characterization of small colony variants of Pseudomonas aeruginosa from monomicrobial and P. aeruginosa-Aspergillus fumigatus mixed microbial biofilms. In: Interscience conference on antimicrobial agents and chemotherapy, 2012. Abstr. M-986.
Nazik H, Moss RB, Karna V, et al. Are cystic fibrosis Aspergillus fumigatus isolates different? Intermicrobial interactions with Pseudomonas. Mycopathologia. 2017;182:315–8.
Visaggio D, Pasqua M, Bonchi C, et al. Cell aggregation promotes pyoverdine-dependent iron uptake and virulence in Pseudomonas aeruginosa. Front Microbiol. 2015;6:902.
Acknowledgements
This work was presented in part to the 4th Meeting of the ECMM/ISHAM Working group, Fungal respiratory infections in Cystic Fibrosis, in Osuna (Seville), Spain, June 5–6, 2017. These studies were partially supported by a grant from the Child Health Research Institute, Stanford Transdisciplinary Initiatives Program, CIMR no. 3777, and by a gift from John Flatley, CIMR no. 3770. The funding sources had no involvement in study design, data handling or interpretation, writing or decision to publish.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no competing interests/conflicts of interest.
Informed Consent
CF isolates from patient respiratory cultures were obtained after written informed consent for biobanking of the patients’ specimens and subsequent use, approved by the Stanford Institutional Review Board.
Rights and permissions
About this article
Cite this article
Anand, R., Moss, R.B., Sass, G. et al. Small Colony Variants of Pseudomonas aeruginosa Display Heterogeneity in Inhibiting Aspergillus fumigatus Biofilm. Mycopathologia 183, 263–272 (2018). https://doi.org/10.1007/s11046-017-0186-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-017-0186-9