Abstract
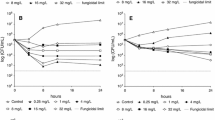
Caspofungin activity was determined in vitro and in vivo against three Candida orthopsilosis, three C. metapsilosis, two C. parapsilosis sensu stricto and two C. albicans isolates. MIC values and killing activity were determined in RPMI-1640 plus 50 % human serum. Neutropenic (cyclophosphamide-treated) mice were infected intravenously. Five-day intraperitoneal treatment with caspofungin was started after 24 h postinfection. Kidney burden was analyzed using the Kruskal–Wallis test with Dunn’s post-test. In killing studies, caspofungin was fungistatic and fungicidal against C. albicans at ≥0.25 and ≥2 μg/ml concentrations, respectively. Caspofungin was fungistatic at ≥8–16, ≥2–8 and at ≥2–8 μg/ml against C. parapsilosis, C. orthopsilosis and C. metapsilosis, respectively. In the murine model, C. albicans was inhibited by 1, 2 and 5 mg/kg of caspofungin (P < 0.001 compared to the controls). Against C. parapsilosis, only 5 mg/kg caspofungin was effective against both isolates (P < 0.05). Two and five mg/kg of caspofungin was effective against all C. orthopsilosis and C. metapsilosis isolates (P < 0.05 to <0.001). Serum-based killing tests proved to be useful in predicting in vivo efficacy of caspofungin against four Candida species. Caspofungin at clinically attainable concentrations proved to be effective against all four species.



Similar content being viewed by others
References
Pfaller MA, Diekema DJ. Epidemiology of invasive candidiasis: a persistent public health problem. Clin Microbiol Rev. 2007;20:133–63.
Lockhart SR, Messer SA, Pfaller MA, Diekema DJ. Geographic distribution and antifungal susceptibility of the newly described species Candida orthopsilosis and Candida metapsilosis, in comparison to the closely-related species Candida parapsilosis. J Clin Microbiol. 2008;46:2659–64.
Garcia-Effron G, Katiyar SK, Park S, Edlind TD, Perlin DS. A naturally occurring proline-to-alanine amino acid change in Fks1p in Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis accounts for reduced echinocandin susceptibility. Antimicrob Agents Chemother. 2008;52:2305–12.
Tavanti A, Davidson AD, Gow NAR, Maiden MCJ, Odds FC. Candida orthopsilosis and Candida metapsilosis spp. nov., to replace Candida parapsilosis groups II and III. J Clin Microbiol. 2005;43:284–92.
van Asbeck E, Clemons KV, Martinez M, Tong AJ, Stevens DA. Significant differences in drug susceptibility among species in the Candida parapsilosis group. Diagn Microbiol Infect Dis. 2008;62:106–9.
Gómez-López A, Alastruey-Izquierdo A, Rodríguez D, Almirante B, Pahissa A, Rodríguez- Tudela JL, Cuenca-Estrella M. The Barcelona Candidemia Project Study Group: Prevalence and susceptibility profile of Candida metapsilosis and Candida orthopsilosis: results from population-based surveillance of candidemia in Spain. Antimicrob Agents Chemother. 2008;52:1506–9.
Cantón E, Espinel-Ingroff A, Pemán J, del Castillo L. In vitro fungicidal activities of echinocandins against Candida metapsilosis, C. orthopsilosis, and C. parapsilosis evaluated by time-kill studies. Antimicrob Agents Chemother. 2010;54:2194–7.
Garcia-Effron GS, Park S, Perlin DS. Improved detection of Candida sp. fks hot spot mutants by using the method of the CLSI M27-A3 Document with the addition of bovine serum albumin. Antimicrob Agents Chemother. 2011;55:2245–55.
Ishikawa J, Maeda T, Matsumura I, Yasumi M, Ujiie H, Masaie H, Nakazawa T, Mochizuki N, Kishino S, Kanakura Y. Antifungal activity of micafungin in serum. Antimicrob Agents Chemother. 2009;53:4559–62.
Odabasi Z, Paetznick V, Rex JH, Ostrosky-Zeichner L. Effects of serum on in vitro susceptibility testing of echinocandins. Antimicrob Agents Chemother. 2007;51:4214–6.
Paderu P, Garcia-Effron G, Balashov S, Delmas G, Park S, Perlin DS. Serum differentially alters the antifungal properties of echinocandin drugs. Antimicrob Agents Chemother. 2007;51:2253–6.
Varga I, Sóczó G, Kardos G, Borbély A, Szabó Z, Kemény-Beke A, Majoros L. Comparison of killing activity of caspofungin against Candida parapsilosis, C. orthopsilosis and C. metapsilosis. J Antimicrob Chemother. 2008;62:1466–8.
Szabó Z, Szilágyi J, Tavanti A, Kardos G, Rozgonyi F, Bayegan S, Majoros L. In vitro efficacy of 5 antifungal agents against Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis as determined by time-kill methodology. Diagn Microbiol Infect Dis. 2009;64:283–8.
Bayegan S, Szilágyi J, Kemény-Beke Á, Földi R, Kardos G, Gesztelyi R, Juhasz B, Adnan A, Majoros L. Efficacy of a single 6 mg/kg versus two 3 mg/kg caspofungin doses for treatment of disseminated candidiasis caused by Candida albicans in a neutropenic mouse model. J Chemother. 2011;23:107–9.
Clinical and Laboratory Standards Institute. Reference method for broth dilution antifungal susceptibility testing of yeasts. Approved standard, 3rd ed. M27-A3. Clinical and Laboratory Standards Institute, Wayne, PA. 2008.
Stone JA, Holland SD, Wickersham PJ, Sterrett A, Schwartz M, Bonfiglio C, Hesney M, Winchell GA, Deutsch PJ, Greenberg H, Hunt TL, Waldman SA. Single- and multiple-dose pharmacokinetics of caspofungin in healthy men. Antimicrob Agents Chemother. 2002;46:739–45.
Szilágyi J, Földi R, Bayegan S, Kardos G, Majoros L. Effect of nikkomycin Z and 50 % human serum on the killing activity of high-concentration caspofungin against Candida species using time-kill methodology. J Chemother. 2012;24:18–25.
Andes D, Marchillo K, Stamstad T, Conklin R. In vivo pharmacodynamics of a new triazole, ravuconazole, in a murine candidiasis model. Antimicrob Agents Chemother. 2003;47:1193–9.
Migoya EM, Mistry GC, Stone JA, Comisar W, Sun P, Norcross A, Bi S, Winchell GA, Ghosh K, Uemera N, Deutsch PJ, Wagner JA. Safety and pharmacokinetics of higher doses of caspofungin in healthy adult participants. J Clin Pharmacol. 2011;51:202–11.
Flattery AM, Hickey E, Gill CJ, Powles MA, Misura AS, Galgoci AM, Ellis JD, Zhang R, Sandhu P, Ronan J, Abruzzo GK. Efficacy of caspofungin in a juvenile mouse model of central nervous system candidiasis. Antimicrob Agents Chemother. 2011;55:3491–7.
Pfaller MA, Diekema DJ, Andes D, Arendrup MC, Brown SD, Lockhart SR, Motyl M, Perlin DS. The CLSI Subcommittee for antifungal testing: clinical breakpoints for the echinocandins and Candida revisited: Integration of molecular, clinical, and microbiological data to arrive at species-specific interpretive criteria. Drug Resist Updat. 2011;14:164–76.
Pappas PG, Kauffman CA, Andes D, Benjamin DK Jr. infectious diseases society of America: clinical practice guidelines for the management of candidiasis: update by the infectious diseases society of america. Clin Infect Dis. 2009;48:503–35.
Kabbara N, Lacroix C. Breakthrough C. parapsilosis and C. guilliermondii blood stream infections in allogeneic hematopoietic stem cell transplant recipients receiving long-term caspofungin therapy. Haematologica. 2008;93:639–40.
Betts RF, Nucci M, Talwar D, Gareca M, Queiroz-Telles F, Bedimo RJ, Herbrecht R, Ruiz-Palacios G, Young JA, Baddley JW, Strohmaier KM, Tucker KA, Taylor AF, Kartsonis NA. Caspofungin high-dose study group: A multicenter, double-blind trial of a high-dose caspofungin treatment regimen versus a standard caspofungin treatment regimen for adult patients with invasive candidiasis. Clin Infect Dis. 2009;48:1676–84.
Barchiesi F, Spreghini E, Tomassetti S, Della Vittoria A, Arzeni D, Manso E, Scalise G. Effects of caspofungin against Candida guilliermondii and Candida parapsilosis. Antimicrob Agents Chemother. 2006;50:2719–27.
Acknowledgments
Caspofungin pure powder was kindly provided by Merck Research Laboratories, Rahway, NJ., USA. R. Földi was supported by a Richter Gedeon Pharma PhD scholarship, L. Majoros was supported by a Bolyai Research Scholarship of the Hungarian Academy of Sciences. The work of R. Földi and L. Majoros was supported by the TÁMOP 4.2.1./B-09/1/KONV-2010-0007 project (New Hungary Development Plan, co-financed by the European Social Fund and the European Regional Development Fund). L. Majoros received conference travel grants from MSD and Pfizer.
Conflict of interest
R. Földi was supported by a Richter Gedeon Pharma PhD scholarship, L. Majoros was supported by a Bolyai Research Scholarship of the Hungarian Academy of Sciences. The work of R. Földi and L. Majoros was supported by the TÁMOP 4.2.1./B-09/1/KONV-2010-0007 project (New Hungary Development Plan, co-financed by the European Social Fund and the European Regional Development Fund).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Földi, R., Kovács, R., Gesztelyi, R. et al. Comparison of In Vitro and Vivo Efficacy of Caspofungin Against Candida parapsilosis, C. orthopsilosis, C. metapsilosis and C. albicans . Mycopathologia 174, 311–318 (2012). https://doi.org/10.1007/s11046-012-9554-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-012-9554-7