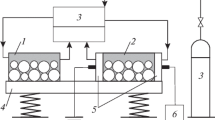
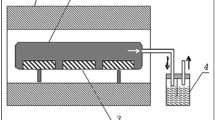
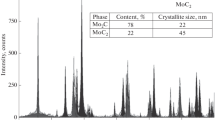
The main factors limiting the application of mechanochemical synthesis to tungsten carbide in the WO3 + Mg + C system are determined. It is shown that carbon modifications obtained by the pyrolysis of a vegetable raw material can be used to synthesize tungsten carbide with a minimal content of sulfur.








Similar content being viewed by others
References
G. V. Samsomov, V. K. Vitryanyuk, and F. I. Chaplygin, Tungsten Carbides [in Russian], Naukova Dumka, Kiev (1974).
E. G. Avvakumov, Mechanical Methods of Activating Chemical Processes [in Russian], Nauka, Novosibirsk (1986).
A. A. Popovich, V. P. Reva, and V. N. Vasilenko, “Physico-chemical laws governing the mechanochemical reduction of metals and nonmetals and their oxides,” Izv. Vyssh. Uchebn. Zaved., Chern. Metall., No. 5, 6–9 (1992).
A. A. Popovich, Mechanochemical Synthesis of Refractory Compounds [in Russian], Izd-vo DVGTU, Vladivostok (2003).
D. V. Onishchenko, A. A. Popovich, and Van Sin Shen, “Synthesis of nanosized tungsten carbide powder for creating functional nanocomposites,” Izv. Vyssh. Uchebn. Zaved., Poroshk. Metall. Funktsion. Pokrytiya, No. 2, 9–15 (2011).
D. V. Onishchenko and V. P. Reva, “Production of tungsten carbide nanopowder by mechanical activation,” Fiz. Khim. Obrab. Mater., No. 2, 71–77 (2011).
G. A. Gorokhovskii, V. G. Chernyshev, V. P. Reva, et al., “Production of metallic powders by the comminution of chip wastes,” Poroshk. Metall., No. 12, 1–8 (1988).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Metallovedenie i Termicheskaya Obrabotka Metallov, No. 5, pp. 48 – 54, May, 2013.
Rights and permissions
About this article
Cite this article
Reva, V.P., Onishchenko, D.V. Tungsten carbide obtained by mechanochemical synthesis with the use of different carbon agents. Met Sci Heat Treat 55, 275–280 (2013). https://doi.org/10.1007/s11041-013-9619-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11041-013-9619-3