Abstract
Background
In viral infections, mitochondria act as one of the main hubs of the pathogenesis. Recent findings present new insights into the potential role of circulating cell-free mitochondrial DNA (ccf-mtDNA) in COVID-19 pathogenesis by the induction of immune response and aggressive cytokine storm in SARS-CoV-2 infection.
Methods and results
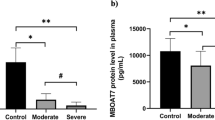
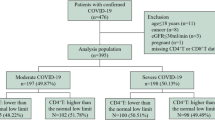
The levels of ccf-mtDNA were investigated in 102 hospitalized patients with COVID-19 using the quantitative PCR (q-PCR) method. Statistical analysis confirmed a strong association between the levels of ccf-mtDNA and and mortality, ICU admission, and intubation. Also, our findings highlighted the pivotal role of comorbidities as a risk factor for COVID-19 mortality and severity.
Conclusion
Higher levels of ccf-mtDNA can serve as a potential early indicator for progress and poor prognosis of COVID-19.

Similar content being viewed by others
Data Availability
Data and materials are available upon request.
Abbreviations
- COVID-19:
-
Coronavirus disease 2019
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus-2
- DAMP:
-
Damage-associated molecular pattern
- mt-DAMP:
-
Mitochondrial damage-associated molecular pattern
- ccf-mtDNA:
-
Circulating cell-free mitochondrial DNA
- ICU:
-
Intensive care unit
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- CYB:
-
Cytochrome b
- COX3:
-
Cytochrome c oxidase subunit III
- SD:
-
Standard deviation
- CVD:
-
Cardiovascular diseases
- HTN:
-
Hypertension
- DM type2:
-
Diabetes mellitus type 2
- ROC:
-
Receiver operating characteristic
- TLR9:
-
Toll-like receptors 9
- cGAS-STING:
-
Cyclic guanosine monophosphate -adenosine monophosphate synthase stimulator of interferon genes
- OS:
-
Oxidative stress
References
Wang C, Horby PW, Hayden FG, Gao GF (2020) A novel coronavirus outbreak of global health concern. The Lancet 395(10223):470–473
Lynch SM, Guo G, Gibson DS, Bjourson AJ, Rai TS (2021) Role of senescence and aging in SARS-CoV-2 infection and COVID-19 disease. Cells 10(12):3367
Mahmoodpoor A, Sanaie S, Ostadi Z et al (2022) Roles of mitochondrial DNA in dynamics of the immune response to COVID-19. Gene 836:146681
Mahmoodpoor A, Sanaie S, Roudbari F, Sabzevari T, Sohrabifar N, Kazeminasab S (2022) Understanding the role of telomere attrition and epigenetic signatures in COVID-19 severity. Gene 811:146069
Bhowal C, Ghosh S, Ghatak D, De R (2023) Pathophysiological involvement of host mitochondria in SARS-CoV-2 infection that causes COVID-19: a comprehensive evidential insight. Mol Cell Biochem 478(6):1325–1343
West AP, Shadel GS, Ghosh S (2011) Mitochondria in innate immune responses. Nat Rev Immunol 11(6):389–402
Piantadosi CA (2020) Mitochondrial DNA, oxidants, and innate immunity. Free Radic Biol Med 152:455–461
Scozzi D, Cano M, Ma L et al (2021) Circulating mitochondrial DNA is an early indicator of severe illness and mortality from COVID-19. JCI Insight. ;6(4)
Herst PM, Rowe MR, Carson GM, Berridge MV (2017) Functional mitochondria in health and disease. Front Endocrinol 8:296
Moro L (2020) Mitochondria at the Crossroads of Physiology and Pathology. MDPI, p 1971
Gallucci S, Maffei ME (2017) DNA sensing across the tree of life. Trends Immunol 38(10):719–732
Valdes-Aguayo JJ, Garza-Veloz I, Badillo-Almaraz JI et al (2021) Mitochondria and mitochondrial DNA: key elements in the pathogenesis and exacerbation of the inflammatory state caused by COVID-19. Medicina 57(9):928
Itagaki K, Kaczmarek E, Lee YT et al (2015) Mitochondrial DNA released by trauma induces neutrophil extracellular traps. PLoS ONE 10(3):e0120549
Hepokoski ML, Odish M, Lam MT et al (2022) Absolute quantification of plasma mitochondrial DNA by droplet digital PCR marks COVID-19 severity over time during intensive care unit admissions. Am J Physiology-Lung Cell Mol Physiol 323(1):L84–L92
Leonard A, Rolfsen M, Ix J et al (2022) Circulating, cell-free mitochondrial DNA Marks Disease Severity in COVID-19. B45 PREDICTING COVID SEVERITY AND OUTCOMES. American Thoracic Society, pp A2927–A
Aswani A, Manson J, Itagaki K et al (2018) Scavenging circulating mitochondrial DNA as a potential therapeutic option for multiple organ dysfunction in trauma hemorrhage. Front Immunol 9:891
Alyammahi SK, Abdin SM, Alhamad DW, Elgendy SM, Altell AT, Omar HA (2021) The dynamic association between COVID-19 and chronic disorders: an updated insight into prevalence, mechanisms and therapeutic modalities. Infect Genet Evol 87:104647
Chatterjee S, Nalla LV, Sharma M et al (2023) Association of COVID-19 with Comorbidities: an update. ACS Pharmacol Translational Sci 6(3):334–354
Reyna-Villasmil E, Caponcello MG, Maldonado N et al (2022) Association of Patients’ epidemiological characteristics and comorbidities with severity and related mortality risk of SARS-CoV-2 infection: results of an umbrella systematic review and Meta-analysis. Biomedicines 10(10):2437
Saleh J, Peyssonnaux C, Singh KK, Edeas M (2020) Mitochondria and microbiota dysfunction in COVID-19 pathogenesis. Mitochondrion 54:1–7
Valdés-Aguayo JJ, Garza-Veloz I, Vargas-Rodríguez JR et al (2021) Peripheral blood mitochondrial DNA levels were modulated by SARS-CoV-2 infection severity and its lessening was associated with mortality among hospitalized patients with COVID-19. Front Cell Infect Microbiol 11:1285
Acknowledgements
Not applicable.
Funding
This work was supported by Tabriz University of Medical Sciences [grant code 68454].
Author information
Authors and Affiliations
Contributions
A.M. administered the project and provided final approval of the manuscript, M.M. and R.A. helped with sample collection,M.T. and A.I. revised the manuscript, M.R. and A.J.P. reviewed and edited the draft, B.E. performed methodology and validated data, N.S. and S.K. conceived the study, performed the statistical analysis and wrote the original draft.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Tabriz University of medical sciences (IR.TBZMED.REC.1399.542). All participants signed the informed consent.
Consent for publication
Consent to publish had been obtained from all participants, and all authors agreed to publish this paper.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
: A) Association of ccf-mtDNA copy number with mortality in patients with COVID-19. B) Receiver operating characteristic (ROC) curves and predictive value of ccf-mtDNA copy number in predicting mortality
Figure S2: A) Association of ccf-mtDNA copy number with ICU admission in patients with COVID-19. B) Receiver operating characteristic (ROC) curves and predictive value of ccf-mtDNA copy number in predicting ICU admission
Figure S3: A) Association of ccf-mtDNA copy number with intubation in patients with COVID-19. B) Receiver operating characteristic (ROC) curves and predictive value of ccf-mtDNA copy number in predicting intubation.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mahmoodpoor, A., Mohammadzadeh, M., Asghari, R. et al. Prognostic potential of circulating cell free mitochondrial DNA levels in COVID-19 patients. Mol Biol Rep 50, 10249–10255 (2023). https://doi.org/10.1007/s11033-023-08841-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08841-3