Abstract
Background
MicroRNA (miRNA) expression analysis has been shown to provide them as biomarkers in several eye diseases and has a regulatory role in pathogenesis. However, miRNA expression analysis in the vitreous humor (VH) of intraocular tuberculosis (IOTB) is not studied. Thus, we aim to find miRNA expression signatures in the VH of IOTB patients to identify their regulatory role in disease pathogenesis and to find them as potential biomarkers for IOTB.
Methods and results
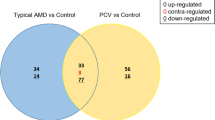
First, we profiled miRNAs in VH of three IOTB and three Macular hole (MH) samples as controls through small-RNA deep sequencing using Illumina Platform. In-house bioinformatics analysis identified 81 dysregulated miRNAs in IOTB. Further validation in VH of IOTB (n = 15) compared to MH (n = 15) using Real-Time quantitative PCR (RT-qPCR) identified three significantly upregulated miRNAs, hsa-miR-150-5p, hsa-miR-26b-5p, and hsa-miR-21-5p. Based on the miRNA target prediction, functional network analysis, and RT-qPCR analysis of target genes, the three miRNAs downregulating WNT5A, PRKCA, MAP3K7, IL7, TGFB2, IL1A, PRKCB, TNFA, and TP53 genes involving MAPK signaling pathway, PI3K-AKT signaling pathway, WNT signaling pathway, Cell cycle, TGF-beta signaling pathway, Long-term potentiation, and Sphingolipid signaling pathways, have a potential role in disease pathogenesis. The ROC analysis of RT-qPCR data showed that hsa-miR-150-5p with AUC = 0.715, hsa-miR-21-5p with AUC = 0.789, and hsa-miR-26b-5p with AUC = 0.738; however, the combination of hsa-miR-21-5p and hsa-miR-26b-5p with AUC = 0.796 could serve as a potential biomarker for IOTB.
Conclusions
This study provides the first report on miRNA expression signatures detected in VH for IOTB pathogenesis and also provides a potential biomarker for IOTB.




Similar content being viewed by others
Data Availability
The data is available upon request from authors.
Abbreviations
- OTB:
-
Ocular Tuberculosis
- Mtb :
-
Mycobacterium tuberculosis
- ETB:
-
Extrapulmonary tuberculosis
- TB:
-
Tuberculosis
- ATT:
-
Anti-tuberculosis therapy
- PPD:
-
Purified protein derivative
- IGRA:
-
Interferon-gamma release assay
- NAAT:
-
Nucleic acid amplification test
- AH:
-
Aqueous humor
- VH:
-
Vitreous humor
- IL:
-
6-Interleukin-6
- CXCL8:
-
C-X-C Motif Chemokine Ligand 8
- IL:
-
8- Interleukin-8
- CXCL9:
-
C-X-C Motif Chemokine Ligand 9
- CXCL10:
-
C-X-C Motif Chemokine Ligand 10
- ESAT:
-
6- Early secretory antigenic 6 kDa
- CFP:
-
10-Culture filtrate protein 10
- CXCL13:
-
C-X-C Motif Chemokine Ligand 13
- CCL17:
-
CC motif chemokine ligand 17
- IL:
-
17- Interleukin-17
- IOTB:
-
Intraocular tuberculosis
- PCR:
-
Polymerase chain reaction
- BD:
-
Becton Dickinson
- MH:
-
Macular hole
- RT:
-
Room temperature
- FTMH:
-
Full-thickness macular hole
- RNA:
-
Ribonucleic acid
- DNA:
-
Deoxy ribonucleic acid
- cDNA:
-
complementary DNA
- STAR:
-
Spliced transcripts alignment to a reference
- GRCh38:
-
Genome reference consortium human build 38
- DE:
-
Differential expression
- TMM:
-
Trimmed mean of M-values
- FC:
-
Fold change
- CPM:
-
Counts per million
- ROC curve:
-
Receiver operating characteristic curve
- RT:
-
qPCR-Real time quantitative PCR
- ACTB:
-
β-Actin
- DAVID:
-
Database for Annotation, Visualization, and Integrated Discovery
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- FDR:
-
False Discovery Rate
- MPB64:
-
Mannose binding protein 64
- miRNA:
-
MicroRNA
- MAPK:
-
Mitogen-activated protein kinase
- TGFB:
-
Transforming growth factor beta
- WNT:
-
Wingless-related integration site
- PI3K:
-
Akt- phosphoinositide-3-kinase-protein kinase B
- TP53:
-
Tumor protein 53
- IL:
-
1A-Interleukin-1A
- PRKCB:
-
Protein Kinase C beta
- PRKCA:
-
Protein Kinase C Alpha
- MAP3K7:
-
Mitogen-activated protein kinase kinase kinase 7
- IL:
-
7-Interleukin-7
- TNFα or TNFA:
-
Tumor necrosis factor alpha
- WNT5A:
-
WNT family member 5A
- AUC:
-
Area under the ROC curve
- CI:
-
Confidence interval
- MPT53:
-
Mycobacterium protein tuberculosis 53
- IFNɣ:
-
Interferon ɣ
References
Neuhouser AJ, Sallam A (2023) Ocular tuberculosis. StatPearls Treasure Island (FL) ineligible companies. Ahmed Sallam declares no relevant financial relationships with ineligible companies, Disclosure
Moule MG, Cirillo JD (2020) Mycobacterium tuberculosis Dissemination plays a critical role in Pathogenesis. Front Cell Infect Microbiol 10:65. https://doi.org/10.3389/fcimb.2020.00065
Lin CJ, Hsia NY, Hwang DK, Hwang YS, Chang YC, Lee YC, Hsu YR, Yeh PT, Lin CP, Chen HF et al (2022) Clinical Manifestations and Outcomes of Tubercular Uveitis in Taiwan-A ten-year Multicenter Retrospective Study. Med (Kaunas) 58(3):376. https://doi.org/10.3390/medicina58030376
Testi I, Agrawal R, Mehta S, Basu S, Nguyen Q, Pavesio C, Gupta V (2020) Ocular tuberculosis: where are we today? Indian J Ophthalmol 68(9):1808–1817. https://doi.org/10.4103/ijo.IJO_1451_20
Chawla R, Singh MK, Singh L, Shah P, Kashyap S, Azad S, Venkatesh P, Sen S (2022) Tubercular DNA PCR of ocular fluids and blood in cases of presumed ocular tuberculosis: a pilot study. Ther Adv Ophthalmol 14:25158414221123522. https://doi.org/10.1177/25158414221123522
Ang M, Cheung G, Vania M, Chen J, Yang H, Li J, Chee SP (2012) Aqueous cytokine and chemokine analysis in uveitis associated with tuberculosis. Mol Vis 18:565–573
Alam A, Imam N, Ahmed MM, Tazyeen S, Tamkeen N, Farooqui A, Malik M, Ishrat R (2019) Identification and classification of differentially expressed genes and Network Meta-Analysis reveals potential Molecular Signatures Associated with Tuberculosis. Front Genet 10:932. https://doi.org/10.3389/fgene.2019.00932
Alam K, Sharma G, Forrester JV, Basu S (2023) Antigen-Specific intraocular cytokine responses distinguish ocular tuberculosis from undifferentiated uveitis in tuberculosis-immunoreactive patients. Am J Ophthalmol 246:31–41. https://doi.org/10.1016/j.ajo.2022.08.029
Bansal R, Khan MM, Dasari S, Verma I, Goodlett DR, Manes NP, Nita-Lazar A, Sharma SP, Kumar A, Singh N (2021) Proteomic profile of vitreous in patients with tubercular uveitis. Tuberculosis (Edinb) 126:102036. https://doi.org/10.1016/j.tube.2020.102036
Hutchinson PE, Kee AR, Agrawal R, Yawata N, Tumulak MJ, Connolly JE, Chee SP, Siak J (2021) Singapore Ocular Tuberculosis Immunity Study (SPOTIS): role of T-lymphocyte profiling in patients with presumed ocular tuberculosis. Ocul Immunol Inflamm 29(7–8):1489–1495. https://doi.org/10.1080/09273948.2020.1767791
Chadalawada S, Kathirvel K, Lalitha P, Rathinam S, Devarajan B (2022) Dysregulated expression of microRNAs in aqueous humor from intraocular tuberculosis patients. Mol Biol Rep 49:1–11. https://doi.org/10.1007/s11033-021-06846-4
Mammadzada P, Bayle J, Gudmundsson J, Kvanta A, Andre H (2019) Identification of Diagnostic and Prognostic microRNAs for recurrent vitreous hemorrhage in patients with proliferative Diabetic Retinopathy. J Clin Med 8(12):2217. https://doi.org/10.3390/jcm8122217
Toro M, Reibaldi M, Avitabile T, Bucolo C, Salomone S, Rejdak R, Nowomiejska K, Tripodi S, Posarelli C, Ragusa M et al (2020) MicroRNAs in the vitreous humor of patients with retinal detachment and a different grading of proliferative vitreoretinopathy: a pilot study MicroRNAs and proliferative vitreoretinopathy. Transl Vis Sci Technol 9(6):23. https://doi.org/10.1167/tvst.9.6.23
Kot A, Kaczmarek R (2022) Exosomal miRNA profiling in vitreous humor in proliferative Diabetic Retinopathy. Cells 12(1):123. https://doi.org/10.3390/cells12010123
Bansal R, Dogra M, Chawla R, Kumar A (2020) Pars plana vitrectomy in uveitis in the era of microincision vitreous surgery. Indian J Ophthalmol 68(9):1844–1851. https://doi.org/10.4103/ijo.IJO_1625_20
Gupta A, Bansal R, Gupta V, Sharma A, Bambery P (2010) Ocular signs predictive of tubercular uveitis. Am J Ophthalmol 149(4):562–570. https://doi.org/10.1016/j.ajo.2009.11.020
Brandine G, Smith A (2019) Falco: high-speed FastQC emulation for quality control of sequencing data. F1000Res 8:1874. https://doi.org/10.12688/f1000research.21142.2
Marconcini M, Pischedda E, Houe V, Palatini U, Lozada-Chavez N, Sogliani D, Failloux AB, Bonizzoni M (2021) Profile of small RNAs, vDNA forms and viral integrations in late Chikungunya Virus infection of Aedes albopictus Mosquitoes. Viruses 13(4):553. https://doi.org/10.3390/v13040553
Dobin A, Davis CA, Schlesinger F, Drenkow J, Zaleski C, Jha S, Batut P, Chaisson M, Gingeras TR (2013) STAR: ultrafast universal RNA-seq aligner. Bioinf (Oxford England) 29(1):15–21. https://doi.org/10.1093/bioinformatics/bts635
Chisanga D, Liao Y, Shi W (2022) Impact of gene annotation choice on the quantification of RNA-seq data. BMC Bioinformatics 23(1):107. https://doi.org/10.1186/s12859-022-04644-8
Kozomara A, Birgaoanu M, Griffiths-Jones S (2019) miRBase: from microRNA sequences to function. Nucleic Acids Res 47(D1):D155–D162. https://doi.org/10.1093/nar/gky1141
Tarazona S, Furió-Tarí P, Turrà D, Pietro AD, Nueda MJ, Ferrer A, Conesa A (2015) Data quality aware analysis of differential expression in RNA-seq with NOISeq R/Bioc package. Nucleic Acids Res 43(21):e140–e140. https://doi.org/10.1093/nar/gkv711
Abbas-Aghababazadeh F, Li Q, Fridley BL (2018) Comparison of normalization approaches for gene expression studies completed with high-throughput sequencing. PLoS ONE 13(10):e0206312. https://doi.org/10.1371/journal.pone.0206312
Goedhart J, Luijsterburg MS (2020) VolcaNoseR is a web app for creating, exploring, labeling and sharing volcano plots. Sci Rep 10(1):20560. https://doi.org/10.1038/s41598-020-76603-3
McDermaid A, Monier B, Zhao J, Liu B, Ma Q (2019) Interpretation of differential gene expression results of RNA-seq data: review and integration. Brief Bioinform 20(6):2044–2054. https://doi.org/10.1093/bib/bby067
Sticht C, De La Torre C, Parveen A, Gretz N (2018) miRWalk: an online resource for prediction of microRNA binding sites. PLoS ONE 13(10):e0206239–e0206239. https://doi.org/10.1371/journal.pone.0206239
Otasek D, Morris JH, Boucas J, Pico AR, Demchak B (2019) Cytoscape automation: empowering workflow-based network analysis. Genome Biol 20(1):185. https://doi.org/10.1186/s13059-019-1758-4
Abhishek S, Ryndak MB, Choudhary A, Sharma S, Gupta A, Gupta V, Singh N, Laal S, Verma I (2019) Transcriptional signatures of Mycobacterium tuberculosis in mouse model of intraocular tuberculosis. Pathog Dis 77(5):ftz045. https://doi.org/10.1093/femspd/ftz045
Russo A, Ragusa M, Barbagallo C, Longo A, Avitabile T, Uva M, Bonfiglio V, Toro M, Caltabiano R, Mariotti C et al (2017) MiRNAs in the vitreous humor of patients affected by idiopathic epiretinal membrane and macular hole. PLoS ONE 12:e0174297. https://doi.org/10.1371/journal.pone.0174297
Loporchio DF, Tam EK, Cho J, Chung J, Jun GR, Xia W, Fiorello MG, Siegel NH, Ness S, Stein TD et al (2021) Cytokine levels in human vitreous in proliferative Diabetic Retinopathy. Cells 10(5):1069. https://doi.org/10.3390/cells10051069
El Faouri M, Ally N, Lippera M, Subramani S, Moussa G, Ivanova T, Patton N, Dhawahir-Scala F, Rocha-de-Lossada C, Ferrara M et al (2023) Long-term safety and efficacy of Pars Plana Vitrectomy for Uveitis: experience of a Tertiary Referral Centre in the United Kingdom. J Clin Med 12(9):3252. https://doi.org/10.3390/jcm12093252
Shakarchi F (2015) Ocular tuberculosis: current perspectives. Clin Ophthalmol 9:2223–2227. https://doi.org/10.2147/OPTH.S65254
Chaurasiya SK, Srivastava K (2009) Downregulation of protein kinase C- αenhances intracellular survival of Mycobacteria: role of PknG. BMC Microbiol 9:271. https://doi.org/10.1186/1471-2180-9-271
Shariq M, Quadir N, Sheikh JA, Singh AK, Bishai WR, Ehtesham NZ, Hasnain SE (2021) Post translational modifications in tuberculosis: ubiquitination paradox. Autophagy 17(3):814–817. https://doi.org/10.1080/15548627.2020.1850009
Olsen A, Chen Y, Ji Q, Zhu G, De Silva AD, Vilcheze C, Weisbrod T, Li W, Xu J, Larsen M et al (2016) Targeting Mycobacterium tuberculosis Tumor necrosis factor alpha-downregulating genes for the development of Antituberculous Vaccines. mBio 7(3):e01023–e01015. https://doi.org/10.1128/mBio.01023-15
Lim YJ, Lee J, Choi JA, Cho SN, Son SH, Kwon SJ, Son JW, Song CH (2020) M1 macrophage dependent-p53 regulates the intracellular survival of mycobacteria. Apoptosis 25(1–2):42–55. https://doi.org/10.1007/s10495-019-01578-0
Adankwah E, Harelimana JDD, Minadzi D, Aniagyei W, Abass K, Debrah L, Owusu O, Mayatepek D, Phillips E, Jacobsen R M (2020) Lower monocyte Interleukin-7 receptor expression impairs Anti-Mycobacterial Effector Functions in Tuberculosis Patients. J Immunol 206(10):2430–2440. https://doi.org/10.4049/jimmunol.2001256
Gern BH, Adams KN, Plumlee CR, Stoltzfus CR, Shehata L, Moguche AO, Busman-Sahay K, Hansen SG, Axthelm MK, Picker LJ et al (2021) TGFbeta restricts expansion, survival, and function of T cells within the tuberculous granuloma. Cell Host Microbe 29(4):594–606e596. https://doi.org/10.1016/j.chom.2021.02.005
Trinath J, Holla S, Mahadik K, Prakhar P, Singh V, Balaji KN (2014) The WNT signaling pathway contributes to dectin-1-dependent inhibition of toll-like receptor-induced inflammatory signature. Mol Cell Biol 34(23):4301–4314. https://doi.org/10.1128/MCB.00641-14
Takaki K, Ramakrishnan L, Basu S (2018) A zebrafish model for ocular tuberculosis. PLoS ONE 13(3):e0194982. https://doi.org/10.1371/journal.pone.0194982
Sharma RKM, Gupta VM, Bansal RM, Sharma KM, Gupta AM, Sachdeva NP (2018) Immune profiling of T cells infiltrating vitreous humor in Tubercular Uveitis. Immunol Invest 47(6):615–631. https://doi.org/10.1080/08820139.2018.1470640
Zheng L, Leung E, Lee N, Lui G, To KF, Chan RC, Ip M (2015) Differential MicroRNA expression in human macrophages with Mycobacterium tuberculosis infection of Beijing/W and Non-Beijing/W strain types. PLoS ONE 10(6):e0126018. https://doi.org/10.1371/journal.pone.0126018
Duroux-Richard I, Robin M, Peillex C, Apparailly F (2019) MicroRNAs: fine tuners of Monocyte Heterogeneity. Front Immunol 10:2145. https://doi.org/10.3389/fimmu.2019.02145
Koenig AL, Shchukina I, Amrute J, Andhey PS, Zaitsev K, Lai L, Bajpai G, Bredemeyer A, Smith G, Jones C et al (2022) Single-cell transcriptomics reveals cell-type-specific diversification in human heart failure. Nat Cardiovasc Res 1(3):263–280
Richter E, Ventz K, Harms M, Mostertz J, Hochgrafe F (2016) Induction of macrophage function in human THP-1 cells is Associated with rewiring of MAPK signaling and activation of MAP3K7 (TAK1) protein kinase. Front Cell Dev Biol 4:21. https://doi.org/10.3389/fcell.2016.00021
Bergenfelz C, Janols H, Wullt M, Jirstrom K, Bredberg A, Leandersson K (2013) Wnt5a inhibits human monocyte-derived myeloid dendritic cell generation. Scand J Immunol 78(2):194–204. https://doi.org/10.1111/sji.12075
Saarela J, Kallio SP, Chen D, Montpetit A, Jokiaho A, Choi E, Asselta R, Bronnikov D, Lincoln MR, Sadovnick AD et al (2006) PRKCA and multiple sclerosis: association in two independent populations. PLoS Genet 2(3):e42. https://doi.org/10.1371/journal.pgen.0020042
Ondee T, Jaroonwitchawan T, Pisitkun T, Gillen J, Nita-Lazar A, Leelahavanichkul A, Somparn P (2019) Decreased protein kinase C-β type II Associated with the prominent endotoxin exhaustion in the macrophage of FcGRIIb–/– Lupus Prone mice is revealed by phosphoproteomic analysis. Int J Mol Sci 20:1354. https://doi.org/10.3390/ijms20061354
Sinigaglia A, Peta E, Riccetti S, Venkateswaran S, Manganelli R, Barzon L (2020) Tuberculosis-Associated MicroRNAs: from pathogenesis to Disease biomarkers. Cells 9(10):2160. https://doi.org/10.3390/cells9102160
Acknowledgements
The authors thank Dr. Radhika Thundikandy, Dr. Vedhanayaki Rajesh, Dr. Anjana Somanath, Mrs. Kokila, and the Uvea Clinic, Aravind Eye Hospital, Madurai, India, for the sample collection and clinical assessment. Balagiri Sundar, biostatistician for ROC curve preparation. Dr. Shanthi R, pathologist for histopathological examinations.
Funding
Project supported by the Department of Biotechnology (DBT), India (No: BT/PR20733/MED/29/1075/2016), Swathi Chadalawada is funded by a Senior Research Fellowship provided by the Indian Council of Medical Research (ICMR), India (Sanction letter No. 2019–4013/Gen-BMS dt.30.09.2019).
Author information
Authors and Affiliations
Contributions
SC: Execution, Data curation, Formal analysis, Writing-Original draft preparation RS: Resources, Investigation, Reviewing and Editing PL: Reviewing and Editing NBK: Resources, Reviewing and Editing BD: Conceptualization, Methodology, Writing- Reviewing and Editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interes
All the authors declared that they have no conflict of interest.
Ethical approval
This study was approved by the Institutional Ethics Committee of Aravind Eye Hospital, Madurai, Tamil Nadu, India (IRB2017007BAS).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material: Table S1
. Clinical features of patients with IOTB, Macular hole, and Noninfectious Uveitis samples. Table S2. NGS data statistics. Table S3. List of primers used for the RT-qPCR analysis. Table S4. ROC analysis of VH miRNAs. Table S5. Serum miRNAs validated using RT-qPCR. Table S6. Differential expression analysis of miRNAs using NOISeq
Supplementary Fig. S1
. Log2 FC values of miRNAs in serum of intraocular tuberculosis. Real-time qPCR validation of differentially expressed miRNAs in serum of intraocular tuberculosis (n = 12) and Noninfectious Uveitis samples (n = 8)
Supplementary Fig. S2
. Selected target genes functional network for RT-qPCR validation
Supplementary Fig. S3
. Low power view of haematoxylin and eosin stained section of a enucleated eye with necrotic granuloma within the vitreous cavity and degenerated retina
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chadalawada, S., Rathinam, S., Lalitha, P. et al. Detection of microRNAs expression signatures in vitreous humor of intraocular tuberculosis. Mol Biol Rep 50, 10061–10072 (2023). https://doi.org/10.1007/s11033-023-08819-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08819-1