Abstract
Background
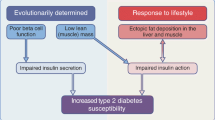
Type 2 diabetes mellitus (T2DM) is a metabolic disorder characterized by limited metabolic flexibility in the body. Such limitation implicates the pyruvate dehydrogenase kinase 4 (PDK4) gene Poor nutrition, frequently observed among Southeast Asians usually involves excessive intakes of carbohydrates and monosodium glutamate (MSG), that have been frequently linked to an increased risk of T2DM.
Methods
The 14-week study aimed to assess the effects of high-carbohydrate (HC), high-MSG (HMSG), and a combination of high-carbohydrate and high-MSG (HCHMSG) diets on the development of T2DM using male mice. To assess the effects, the male mice were divided into four groups: control (C), HC, HMSG, and HCHMSG for 14 weeks.
Results
After 14 weeks, both the HC and HCHMSG groups showed signs of T2DM (168.83 ± 32.33; 156.42 ± 32.46). The blood samples from the HMSG, HC, and HCHMSG groups (57.67 ± 2.882; 49.22 ± 7.36; 48.9 ± 6.43) as well as skeletal muscle samples from the HMSG, HC, and HCHMSG groups (57.78 ± 8.54; 42.13 ± 7.25; 37.57 ± 10.42) exhibited a gradual hypomethylation. The HC groups particularly displayed significant PDK4 gene expression in skeletal muscle. A progressive overexpression of the PDK4 gene was observed as well in the HMSG, HCHMSG, and HC groups (2.03 ± 3.097; 3.21 ± 2.94; 5.86 ± 2.54).
Conclusions
These findings suggest that T2DM can be induced by high-carbohydrate and high-MSG diets. However, the sole consumption of high MSG did not lead to the development of T2DM. Further research should focus on conducting long-term studies to fully comprehend the impact of a high MSG diet on individuals with pre-existing T2DM.





Similar content being viewed by others
Data availability
Data will be made available upon request.
Abbreviations
- C:
-
Control
- FBG:
-
Fasting blood glucose
- HC:
-
High-carbohydrate
- HCHMSG:
-
High-carbohydrate and high-MSG
- HMSG:
-
High-MSG
- MSG:
-
Monosodium glutamate
- MSP:
-
Methylation specific PCR
- OGTT:
-
Oral glucose tolerance test
- PDC:
-
Pyruvate dehydrogenase complex
- PDK4:
-
Pyruvate dehydrogenase kinase 4
- SPSS:
-
Statistical Package for the Social Sciences
- T2DM:
-
Type 2 diabetes mellitus
References
Khan MAB, Hashim MJ, King JK, Govender RD, Mustafa H, Al KJ (2019) Epidemiology of type 2 diabetes – global burden of disease and forecasted trends. J Epidemiol Glob Health 10:107. https://doi.org/10.2991/jegh.k.191028.001
Lin X, Xu Y, Pan X, Xu J, Ding Y, Sun X et al (2020) Global, regional, and national burden and trend of diabetes in 195 countries and territories: an analysis from 1990 to 2025. Sci Rep 10:1–11. https://doi.org/10.1038/s41598-020-71908-9
Yaegashi A, Kimura T, Hirata T, Iso H, Tamakoshi A (2023) Association between low-carbohydrate diet score and incidence of type 2 diabetes among Japanese adults: the JACC Study. J Nutr Sci 12:1–7. https://doi.org/10.1017/jns.2022.122
Yu H, Wang R, Zhao Y, Song Y, Sui H, Wu Y et al (2023) Monosodium glutamate intake and risk assessment in China nationwide, and a comparative analysis worldwide. Nutrients 15:2444. https://doi.org/10.3390/nu15112444
Bahadoran Z, Mirmiran P, Ghasemi A (2019) Monosodium glutamate (MSG)-induced animal model of type 2 diabetes. Methods Mol Biol 1916:49–65. https://doi.org/10.1007/978-1-4939-8994-2_3
da Silva TA, de Carvalho-Filho CJ, de Sousa Barcelos Barroqueiro E, Chagas DC das, Nascimento F via RF, Guerra RNM (2016) The effect of high carbohydrate consumption on glucose levels and antibody production in nonobese diabetic mice. Food Nutr Sci 7:866–873. https://doi.org/10.4236/fns.2016.710086
Vigorelli V, Resta J, Bianchessi V, Lauri A, Bassetti B, Agrifoglio M et al (2019) Abnormal DNA Methylation Induced by Hyperglycemia Reduces CXCR4 Gene Expression in CD34+ Stem Cells. J Am Heart Assoc. https://doi.org/10.1161/JAHA.118.010012
Kumar S, Chowdhury S, Razdan A, Kumari D, Purty RS, Ram H et al (2021) Downregulation of candidate gene expression and neuroprotection by piperine in streptozotocin-induced hyperglycemia and memory impairment in rats. Front Pharmacol 11:1–18. https://doi.org/10.3389/fphar.2020.595471
Jeon JH, Thoudam T, Choi EJ, Kim MJ, Harris RA, Lee IK (2021) Loss of metabolic flexibility as a result of overexpression of pyruvate dehydrogenase kinases in muscle, liver and the immune system: Therapeutic targets in metabolic diseases. J Diabetes Investig 12:21–31. https://doi.org/10.1111/jdi.13345
Putra SED, Singajaya S, Thesman F, Pranoto DA, Sanjaya R, Vianney YM et al (2020) Aberrant PDK4 promoter methylation preceding hyperglycemia in a mouse model. Appl Biochem Biotechnol 190:1023–1034. https://doi.org/10.1007/s12010-019-03143-6
van Otterdijk SD, Binder AM et al (2017) DNA methylation of candidate genes in peripheral blood from patients with type 2 diabetes or the metabolic syndrome. PLoS ONE 12:e0180955. https://doi.org/10.1371/journal.pone.0180955
Kamo T, Kurose S, Ohno H, Murata M, Hashiyada M, Saito T et al (2019) Epigenetic mechanism controls PDK4 gene activation before and after exercise therapy following artificial knee arthroplasty. Clin Interv Aging 14:1433–1443. https://doi.org/10.2147/CIA.S213154
Benedé-Ubieto R, Estévez-Vázquez O, Ramadori P, Cubero FJ, Nevzorova YA (2020) Guidelines and considerations for metabolic tolerance tests in mice. Diabetes Metab Syndr Obes Targets Ther 13:439–450. https://doi.org/10.2147/DMSO.S234665
Logan IE, Bobe G, Miranda CL, Vasquez-Perez S, Choi J, Lowry MB et al (2020) Germ-free swiss webster mice on a high-fat diet develop obesity, hyperglycemia, and dyslipidemia. Microorganisms 8:1–25. https://doi.org/10.3390/microorganisms8040520
Sharples AP, Seaborne RA. Exercise and DNA methylation in skeletal muscle. Sport. Exerc. Nutr. Genomics, Elsevier; 2019, p. 211–29. https://doi.org/10.1016/B978-0-12-816193-7.00010-5.
Stephens NA, Brouwers B, Eroshkin AM, Yi F, Cornnell HH, Meyer C et al (2018) Exercise response variations in skeletal muscle PCR recovery rate and insulin sensitivity relate to muscle epigenomic profiles in individuals with type 2 diabetes. Diabetes Care 41:2245–2254. https://doi.org/10.2337/dc18-0296
Khadka R, Tian W, Hao X, Koirala R (2018) Risk factor, early diagnosis and overall survival on outcome of association between pancreatic cancer and diabetes mellitus: Changes and advances, a review. Int J Surg 52:342–346. https://doi.org/10.1016/j.ijsu.2018.02.058
Baraille F, Ayari S, Carrière V, Osinski C, Garbin K, Blondeau B et al (2015) Glucose tolerance is improved in mice invalidated for the nuclear receptor HNF-4g: a critical role for enteroendocrine cell lineage. Diabetes 64:2744–2756. https://doi.org/10.2337/db14-0993
Morley TS, Xia JY, Scherer PE. Selective enhancement of insulin sensitivity in the mature adipocyte is sufficient for systemic metabolic improvements. Nat Commun 2015;6. https://doi.org/10.1038/ncomms8906.
Kim JY, Michaliszyn SF, Nasr A, Lee SJ, Tfayli H, Hannon T et al (2016) The shape of the glucose response curve during an oral glucose tolerance test heralds biomarkers of type 2 diabetes risk in obese youth. Diabetes Care 39:1431–1439. https://doi.org/10.2337/dc16-0352
Cho Y, Hong N, Kim K, Cho S, Lee M, Lee Y et al (2019) The Effectiveness of intermittent fasting to reduce body mass index and glucose metabolism: a systematic review and meta-analysis. J Clin Med 8:1645. https://doi.org/10.3390/jcm8101645
Wong S, Chin K-Y, Suhaimi F, Ahmad F, Ima-Nirwana S (2018) The effects of a modified high-carbohydrate high-fat diet on metabolic syndrome parameters in male rats. Exp Clin Endocrinol Diabetes 126:205–212. https://doi.org/10.1055/s-0043-119352
Yamada LTP, de Oliveira MC, Batista NV, Fonseca RC, Sousa Pereira RV, Perez DA et al (2016) Immunologic and metabolic effects of high-refined carbohydrate-containing diet in food allergic mice. Nutrition 32:273–280. https://doi.org/10.1016/j.nut.2015.08.011
Ceolin P, De França SA, Froelich M, Dos Santos MP, Pereira MP, Queiroz TS et al (2019) A low-protein, high carbohydrate diet induces increase in serum adiponectin and preserves glucose homeostasis in rats. An Acad Bras Cienc 91:1–12. https://doi.org/10.1590/0001-3765201920180452
Sutton EF, Beyl R, Early KS, Cefalu WT, Ravussin E, Peterson CM (2018) Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metab 27:1212-1221.e3. https://doi.org/10.1016/j.cmet.2018.04.010
Hutchison AT, Liu B, Wood RE, Vincent AD, Thompson CH, O’Callaghan NJ et al (2019) Effects of intermittent versus continuous energy intakes on insulin sensitivity and metabolic risk in women with overweight. Obesity 27:50–58. https://doi.org/10.1002/oby.22345
Jørgensen SW, Hjort L, Gillberg L, Justesen L, Madsbad S, Brøns C et al (2021) Impact of prolonged fasting on insulin secretion, insulin action, and hepatic versus whole body insulin secretion disposition indices in healthy young males. Am J Physiol Metab 320:E281–E290. https://doi.org/10.1152/ajpendo.00433.2020
Coskun ZM, Ersoz M, Adas M, Hancer VS, Boysan SN, Gonen MS et al (2019) Kruppel-like transcription factor-4 gene expression and DNA methylation status in type 2 diabetes and diabetic nephropathy patients. Arch Med Res 50:91–97. https://doi.org/10.1016/j.arcmed.2019.05.012
Tareen SHK, Kutmon M, Arts ICW, de Kok TM, Evelo CT, Adriaens ME (2019) Logical modelling reveals the PDC-PDK interaction as the regulatory switch driving metabolic flexibility at the cellular level. Genes Nutr 14:27. https://doi.org/10.1186/s12263-019-0647-5
Park B, Jeon J, Go Y, Ham HJ, Kim J, Yoo EK et al (2018) PDK4 deficiency suppresses hepatic glucagon signaling by decreasing cAMP levels. Diabetes 67:2054–2068. https://doi.org/10.2337/db17-1529
Humardani FM, Tanaya LTA, Mulyanata LT, Kesuma D, Wijono H, Ikawaty R et al (2023) The effect of high carbohydrate and high msg intake on body weight and white adipose tissue. AIP Con Proc. https://doi.org/10.1063/5.0118548
Ibegbulem CO, Chikezie PC, Ukoha AI, Opara CN (2016) Effects of diet containing monosodium glutamate on organ weights, acute blood steroidal sex hormone levels, lipid profile and erythrocyte antioxidant enzymes activities of rats. J Acute Dis 5:402–407. https://doi.org/10.1016/j.joad.2016.08.007
Egusquiza RJ, Blumberg B (2020) Environmental obesogens and their impact on susceptibility to obesity: new mechanisms and chemicals. Endocrinol (US) 161:1–14. https://doi.org/10.1210/endocr/bqaa024
Acknowledgements
The researchers would like to thank the laboratory personnel at the Faculty of Biotechnology, University of Surabaya, for their invaluable support throughout the research.
Funding
The authors wish to acknowledge the financial assistance provided by the Indonesian Ministry of Research, Technology, and Higher Education for this study (058/SP-Lit/LPPM01/KemendikbudRistek/Multi/FTB/V/2022 and 004/SP2H/PT/LL7/2022).
Author information
Authors and Affiliations
Contributions
SEDP, RI, and FMH performed a major part of the research work, performed statistical analysis, interpreted results, and wrote the initial draft. LTM revised the figures, improved the initial draft, and helped in lab work. LTAT also helped in lab work. HW, DK, HWS conceived the idea, supervised the work, revised, modified, and approved the final draft.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Consent for publication
This study was published with the informed consent of all authors.
Ethical approval
The study underwent a rigorous review from the Research Ethics Committee of the University of Surabaya in Indonesia to ensure that the study was conducted in a manner that respects ethical considerations and the welfare of the subjects involved. The research design received approval from the Committee on September 1, 2020 (No: 138/KE/VIII/2020). The Committee confirmed that the study met the mandatory ethical standards and abided by the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Putra, S.E.D., Humardani, F.M., Mulyanata, L.T. et al. Exploring diet-induced promoter hypomethylation and PDK4 overexpression: implications for type 2 diabetes mellitus. Mol Biol Rep 50, 8949–8958 (2023). https://doi.org/10.1007/s11033-023-08794-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08794-7