Abstract
Background
During gamete development and spermatogenesis, certain genes on the Y chromosome (Yq) in the Male-Specific Region (MSR) are responsible for human gametes formation. The long arm Yq is composed of both euchromatin and the genetically inactive heterochromatin regions. This region contains the Azoospermia factors AZFa, AZFb and AZFc. In the case of male infertility, microdeletions on the Yq chromosome appear to be structural chromosomal anomalies linked to sperm abnormality.
Methods
The present study aimed to look at the incidence, of Asthenospermia (AS), Teratospermia (TS), Oligospermia (OS) and Oligoasthenoteratospermia (OAT) patterns of Y chromosomal microdeletions in Indian infertile men with an (AZF a, b, c). This study was conducted with 75 infertile men as cases and 75 fertile men as a control for AZF locus microdeletion utilizing sequence-tagged sites.
Results
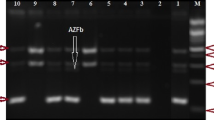
The AZFc region of germ cell DNA (50.6%) was the most deleted section in infertile men when compared to blood DNA (21.3%), followed by deletions in the AZFb region (21.3%) in germ cell DNA whereas blood DNA had no microdeletion in the AZFa region in both germ cell DNA and blood DNA. Infertile men displayed significant Yq microdeletion in both AZFb and also AZFc. Around 33% (25) of 75 infertile men had AZF (a, b, c) region microdeletion in blood DNA, compared to it germ cell DNA had a larger percentage of 72% (54) of Y chromosome microdeletions in the study samples.
Conclusion
A high-frequency rate of microdeletions seen in germ cell DNA. PCR-based Y chromosome microdeletion screening using germ cell DNA along with Genomic DNA might help in screening for genetic abnormality in infertile men who endure assisted reproductive technology treatments. This study might be attributable to the interplay of lifestyle and genetic factors, both contributing to the risk of developing these germ-line deletions.


Similar content being viewed by others
References
Smith LB, Walker WH (2014) The regulation of spermatogenesis by androgens. Semen Cell Dev Biol 30:2–13. https://doi.org/10.1016/j.semcdb.2014.02.012
Wang X, Sharma RK, Sikka SC, Thomas Jr AJ, Falcone T, Agarwal A (2003) Oxidative stress is associated with increased apoptosis leading to spermatozoa DNA damage in patients with male factor infertility. Fertil Steril 80(3):531–535
Krausz C, Rosta V, Swerdloff RS, Wang C (2022) Genetics of male infertility. In: Emery and Rimoin’s principles and practice of medical genetics and genomics. pp 121–147
Fainberg J, Kashanian JA (2019) Recent advances in understanding and managing male infertility. F1000Research. https://doi.org/10.12688/f1000research.17076.1
Leaver RB (2016) Male infertility: an overview of causes and treatment options. Br J Nurs 25(18):S35–S40
Ohlander SJ, Lindgren MC, Shultz L (2016) Testosterone and male infertility. Urol Clin 43(2):195–202
World Health Organization (2010) Laboratory manual for the examination and processing of human semen 2010, 5th edn. Cambridge University Press, Cambridge
Jensen CFS, Østergren P, Dupree JM, Ohl DA, Sønksen J, Fode M (2017) Varicocele and male infertility. Nat Rev Urol 14(9):523–533
Jenardhanan P, Panneerselvam M, Mathur PP (2016) Effect of environmental contaminants on spermatogenesis. Semin Cell Dev Biol 59:126–140
Sharma R, Biedenharn KR, Fedor JM, Agarwal A (2013) Lifestyle factors and reproductive health: taking control of your fertility. Reprod Biol Endocrinol 11(1):1–15
Stouffs K, Seneca S, Lissens W (2014) Genetic causes of male infertility. Annales d’endocrinologie 75(2):109–111
Colaco S, Modi D (2019) Consequences of Y chromosome microdeletions beyond male infertility. J Assist Reprod Genet 36(7):1329–1337
Ceylan GG, Ceylan C, Elyas H (2009) Genetic anomalies in patients with severe oligozoospermia and azoospermia in eastern Turkey: a prospective study. Genet Mol Res 8:915–922
Dong Y, Du R-C, Jiang Y-T, Wu J, Li L-L, Liu R-Z (2012) Impact of chromosomal translocations on male infertility, semen quality, testicular volume and reproductive hormone levels. J Intl Med Res 40:2274
Ambulkar P, Chaudhary A, Waghmare J, Tanaka A, Pal A (2015) Prevalence of Y chromosome microdeletions in idiopathic azoospermia cases in central Indian men. J Clin Diagn Res. https://doi.org/10.7860/JCDR/2015/15249.6515
Rabinowitz MJ, Huffman PJ, Haney NM, Kohn TP (2021) Y-chromosome microdeletions: a review of prevalence, screening, and clinical considerations. Apply Clin Genet 14:51–59. https://doi.org/10.2147/TACG.S267421
Funding
The authors received support from the organization Sri Ramachandra Institute of Higher Education and Research for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Archana, S., Vijayalakshmi, J., Usha Rani, G. et al. The comparison of Y chromosome microdeletion incidence in blood DNA and sperm cell DNA. Mol Biol Rep 50, 1203–1208 (2023). https://doi.org/10.1007/s11033-022-08076-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-08076-8