Abstract
Background
The CRISPR/Cas9 system is widely used for genome editing in human, rat and mouse cells. In this study, we established Fzd6 mutant mice using CRISPR/Cas9 technology, and obtained Fzd6 homozygous mutant (Fzd6Q152E) mice through breeding. Fzd6 plays a role in depression, but there are few related reports. We used this model to investigate the mechanism of Fzd6 involved in depression, and build a solid foundation for subsequent in-depth studies.
Methods and Results
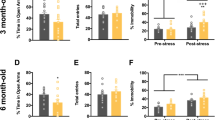
The target of Fzd6 mutation was obtained by CRISPR/Cas9 technology and hippocampal tissue was collected for Nissl staining and histological analysis. Blood was collected for enzyme linked immunosorbent assay (ELISA); The gene expression of Fzd6 and the related genes expression in wnt pathway was quantified by quantitative real-time PCR (qRT-PCR), and then expression of Fzd6 and proteins in the Wnt pathway were identified by western blotting. ELISA results showed that the expression levels of brain derived neurotrophic factor (BDNF), 5-hydroxytryptamine (5-HT), and Noradrenaline (NE) in serum were significantly decreased in Fzd6Q152E mice, whereas the mRNA expression of Lrp5, Lrp6, and Dkk2 is increased. The western blotting revealed that the expression of Fzd6 and Lrp6 is decreased, although the expression of Dkk2 and Gsk-3β increased.
Conclusion
Our study successfully established homozygous Fzd6 mutant mice model. The relationship between Fzd6-Wnt and depression was preliminarily clarified, which provides an ideal animal model for subsequent research on diseases induced by the Fzd6 mutation.





Similar content being viewed by others
References
Schenkelaars Q, Fierro-Constain L, Renard E, Hill AL, Borchiellini C (2015) Insights into Frizzled evolution and new perspectives. Evol Dev 17:160–169. https://doi.org/10.1111/ede.12115
Xu C, Tian G, Jiang C, Xue H, Kuerbanjiang M, Sun L, Gu L, Zhou H, Liu Y, Zhang Z, Xu Q (2019) NPTX2 promotes colorectal cancer growth and liver metastasis by the activation of the canonical Wnt/beta-catenin pathway via FZD6. Cell Death Dis 10:217. https://doi.org/10.1038/s41419-019-1467-7
Yan J, Liu T, Zhou X, Dang Y, Yin C, Zhang G (2016) FZD6, targeted by miR-21, represses gastric cancer cell proliferation and migration via activating non-canonical wnt pathway. Am J Transl Res 8:2354–2364
Han K, Lang T, Zhang Z, Zhang Y, Sun Y, Shen Z, Beuerman RW, Zhou L, Min D (2018) Luteolin attenuates Wnt signaling via upregulation of FZD6 to suppress prostate cancer stemness revealed by comparative proteomics. Sci Rep 8:8537. https://doi.org/10.1038/s41598-018-26761-2
Yuan Y, Wang Q, Ma SL, Xu LQ, Liu MY, Han B, Du N, Sun XL, Yin XL, Cao FF (2019) lncRNA PCAT-1 interacting with FZD6 contributes to the malignancy of acute myeloid leukemia cells through activating Wnt/β-catenin signaling pathway. Am J Transl Res 11:7104–7114
Golan T, Yaniv A, Bafico A, Liu G, Gazit A (2004) The human Frizzled 6 (HFz6) acts as a negative regulator of the canonical Wnt. beta-catenin signaling cascade. J Biol Chem 279:14879–14888. https://doi.org/10.1074/jbc.M306421200
Naz G, Pasternack SM, Perrin C, Mattheisen M, Refke M, Khan S, Gul A, Simons M, Ahmad W, Betz RC (2012) FZD6 encoding the Wnt receptor frizzled 6 is mutated in autosomal-recessive nail dysplasia. Br J Dermatol 166:1088–1094. https://doi.org/10.1111/j.1365-2133.2011.10800.x
Kasparis C, Reid D, Wilson NJ, Okur V, Cole C, Hansen CD, Bosse K, Betz RC, Khan M, Smith FJ (2016) Isolated recessive nail dysplasia caused by FZD6 mutations: report of three families and review of the literature. Clin Exp Dermatol 41:884–889. https://doi.org/10.1111/ced.12934
Voleti B, Tanis KQ, Newton SS, Duman RS (2012) Analysis of target genes regulated by chronic electroconvulsive therapy reveals role for Fzd6 in depression. Biol Psychiatry 71:51–58. https://doi.org/10.1016/j.biopsych.2011.08.004
Stuebner S, Faus-Kessler T, Fischer T, Wurst W, Prakash N (2010) Fzd3 and Fzd6 deficiency results in a severe midbrain morphogenesis defect. Dev Dyn 239:246–260. https://doi.org/10.1002/dvdy.22127
Cota-Coronado A, Díaz-Martínez NF, Padilla-Camberos E, Díaz-Martínez NE (2019) Editing the central nervous system through CRISPR/Cas9 systems. Front Mol Neurosci 12:110. https://doi.org/10.3389/fnmol.2019.00110
Li R, Miao J, Fan Z, Song S, Kong IK, Wang Y, Wang Z (2018) Production of genetically engineered golden syrian hamsters by pronuclear injection of the CRISPR/Cas9 complex. J Vis Exp. https://doi.org/10.3791/56263
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108. https://doi.org/10.1038/nprot.2008.73
Ni D, Xu P, Gallagher S (2017) Immunoblotting and Immunodetection. Curr Protoc Protein Sci. https://doi.org/10.1002/cpps.32
Harro J (2019) Animal models of depression: pros and cons. Cell Tissue Res 377:5–20. https://doi.org/10.1007/s00441-018-2973-0
Dong Y, Li H, Zhao L, Koopman P, Zhang F, Huang JX (2019) Genome-wide off-target analysis in crispr-cas9 modified mice and their offspring. G3 (Bethesda) 9:3645–3651. https://doi.org/10.1534/g3.119.400503
Fan Z, Li W, Lee SR, Meng Q, Shi B, Bunch TD, White KL, Kong IK, Wang Z (2014) Efficient gene targeting in golden Syrian hamsters by the CRISPR/Cas9 system. PLoS ONE. https://doi.org/10.1371/journal.pone.0109755
Hwang WY, Fu Y, Reyon D, Maeder ML, Tsai SQ, Sander JD, Peterson RT, Yeh JR, Joung JK (2013) Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol 31:227–229. https://doi.org/10.1038/nbt.2501
Yu H, Lv D, Shen M, Zhang Y, Zhou D, Chen Z, Wang C (2019) BDNF mediates the protective effects of scopolamine in reserpine-induced depression-like behaviors via up-regulation of 5-HTT and TPH1. Psychiatry Res 271:328–334. https://doi.org/10.1371/journal.pone.0109755
Zhao Y, Wang Q, Jia M, Fu S, Pan J, Chu C, Liu X, Liu X, Liu Z (2019) (+)-Sesamin attenuates chronic unpredictable mild stress-induced depressive-like behaviors and memory deficits via suppression of neuroinflammation. J Nutr Biochem 64:61–71. https://doi.org/10.1016/j.jnutbio.2018.10.006
Gould TD, Manji HK (2002) The Wnt signaling pathway in bipolar disorder. Neuroscientist 8:497–511. https://doi.org/10.1177/107385802237176
Gray JD, Rubin TG, Hunter RG, McEwen BS (2014) Hippocampal gene expression changes underlying stress sensitization and recovery. Mol Psychiatry 19:1171–1178. https://doi.org/10.1038/mp.2013.175
He X, Semenov M, Tamai K, Zeng X (2004) LDL receptor-related proteins 5 and 6 in Wnt/beta-catenin signaling: arrows point the way. Development 131:1663–1677. https://doi.org/10.1242/dev.01117
Muneer A (2017) Wnt and GSK3 signaling pathways in bipolar disorder: clinical and therapeutic implications. Clin Psychopharmacol Neurosci 15:100–114. https://doi.org/10.9758/cpn.2017.15.2.100
Neuhaus J, Weimann A, Berndt-Paetz M (2021) Immunocytochemical analysis of endogenous frizzled-(Co-)receptor interactions and rapid Wnt pathway activation in mammalian cells. Int J Mol Sci. https://doi.org/10.3390/ijms222112057
Ko SB, Mihara E, Park Y, Roh K, Kang C, Takagi J, Bang I, Choi HJ (2022) Functional role of the Frizzled linker domain in the Wnt signaling pathway. Commun Biol 5:421. https://doi.org/10.1038/s42003-022-03370-4
Peng H, Wang HB, Wang L, Zhou B, Li XY, Tan J (2018) Gsk3β aggravates the depression symptoms in chronic stress mouse model. J Integr Neurosci 17:169–175. https://doi.org/10.31083/jin-170050
Devotta A, Hong CS, Saint-Jeannet JP (2018) Dkk2 promotes neural crest specification by activating Wnt/β-catenin signaling in a GSK3β independent manner. Elife. https://doi.org/10.7554/eLife.34404
Acknowledgements
This work was funded and supported by the scientific research project of Shanxi Provincial Health Commission (2021031) and the Provincial Key Laboratory of Biological Psychiatry, the 2020 "Four Batches" Science and Technology Innovation Program (2020SYS10). We would like to thank Editage (www.editage.cn) for English language editing.
Funding
This work was funded and supported by the scientific research project of Shanxi Provincial Health Commission (2021031) and the Provincial Key Laboratory of Biological Psychiatry, the 2020 "Four Batches" Science and Technology Innovation Program (Grant No. 2020SYS10).
Author information
Authors and Affiliations
Contributions
XY and GS: contributed to the study conception, The first draft of the manuscript was written by XY and all authors commented on previous versions of the manuscript. XY, JY, LX, GX, LW and YQ: performed animal experiments, molecular experiments and data analysis.
Corresponding author
Ethics declarations
Conflicts of interest
All authors declare that they have no conflicts of interest.
Ethical approval
The protocol was approved by the Institutional Animal Care and Use Committee of Shanxi Medical University (SYDL2021012).
Informed consent
Informed consent was obtained all participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yan, X., Yang, J., Xiao, L. et al. Construction of Fzd6Q152E mice through CRISPR/Cas9 technology and their reproduction and identification. Mol Biol Rep 49, 9575–9584 (2022). https://doi.org/10.1007/s11033-022-07848-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07848-6