Abstract
Background
Pongamia is considered an important biofuel species worldwide. Drought stress in the early growth stages of Pongamia influences negatively on the germination and seedling development. Due to lack of cultivar stability under drought stress conditions, establishment of successful plantation in drought hit areas becomes a major problem. To address this issue, drought stress response of four Pongamia genotypes was studied at morphological, physio-chemical and transcriptome levels.
Methods and results
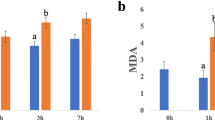
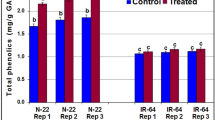
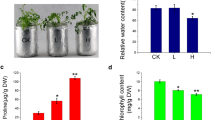
Drought stress was levied by limiting water for 15 days on three months old seedlings of four genotypes. A significant effect of water stress was observed on the traits considered. The genotype NRCP25 exhibited superior morpho-physiological, biochemical drought responses. Also, the genotype had higher root length, photosynthetic pigments, higher antioxidant enzymes and solute accumulation compared to other genotypes. In addition, transcript profiling of selected drought responsive candidate genes such as trehalose phosphate synthase 1 (TPS1), abscisic acid responsive elements-binding protein 2 (ABF2-2), heat shock protein 17 (HSP 17 kDa), tonoplast intrinsic protein 1 (TIP 1–2), zinc finger homeodomain protein 2 (ZFP 2), and xyloglucan endotransglucolase 13 (XET 13) showed only up-regulation in NRCP25. Further, the transcriptome responses are in line with key physio-chemical responses exhibited by NRCP25 for drought tolerance.
Conclusions
As of now, there are no systematic studies on Pongamia drought stress tolerance; therefore this study offers a comprehensive understanding of whole plant drought stress responsiveness of Pongamia. Moreover, the results support important putative trait indices with potential candidate genes for drought tolerance improvement of Pongamia.



Similar content being viewed by others
Availability of data and material
Authors are comply with field standards of this journal.
References
Oshunsanya SO, Nwosu NJ, Li Y (2019)Sustainable Agriculture, Forest and Environmental Management
Ahmad P, Prasad MNV (2011) Abiotic stress responses in plants: metabolism, productivity and sustainability. Springer Science & Business Media
Sturrock RN, Frankel SJ, Brown AV et al (2011) Climate change and forest diseases. Plant Pathol 60:133–149. https://doi.org/10.1111/j.1365-3059.2010.02406.x
Taria S, Rane J, Alam B et al (2020) Combining IR imaging, chlorophyll fluorescence and phenomic approach for assessing diurnal canopy temperature dynamics and desiccation stress management in Azadirachta indica and Terminalia mantaly. Agrofor Syst 94:941–951. https://doi.org/10.1007/s10457-019-00461-w
McDowell N, Pockman WT, Allen CD et al (2008) Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytol 178:719–739. https://doi.org/10.1111/j.1469-8137.2008.02436.x
Rao N, Dong L, Li J, Zhang H (2008) Influence of Environmental Factors on Seed Germination and Seedling Emergence of American Sloughgrass (Beckmannia syzigachne). Weed Sci 56:529–533. https://doi.org/10.1614/ws-07-158.1
Harreh D, Saleh AA, Reddy ANR, Hamdan S (2018) An Experimental Investigation of Karanja Biodiesel Production in Sarawak, Malaysia. https://doi.org/10.1155/2018/4174205. J Eng (United Kingdom) 2018:
Kesari V, Madurai Sathyanarayana V, Parida A, Rangan L (2010) Molecular marker-based characterization in candidate plus trees of Pongamia pinnata, a potential biodiesel legume. AoB Plants 2010
Karmee SK, Chadha A (2005) Preparation of biodiesel from crude oil of Pongamia pinnata. Bioresour Technol 96:1425–1429
Pavithra HR, Gowda B, Prasanna KT, Shivanna MB (2013) Pod and seed traits in candidate plus trees of Pongamia pinnata (L.) Pierre from southern peninsular India in relation to provenance variation and genetic variability. J Crop Sci Biotechnol 16:131–142
PaVela R (2009) Effectiveness of some botanical insecticides against Spodoptera littoralis Boisduvala (Lepidoptera: Noctudiae), Myzus persicae Sulzer (Hemiptera: Aphididae) and Tetranychus urticae Koch (Acari: Tetranychidae). Plant Prot Sci 45:161–167
Swapna B, Rajendrudu G (2015) Seed Germination of Pongamia Pinnata (L.) Pierre under Water Stress. 4:62–66
Rajarajan K, Handa AK (2020) Drought stress responses in seedlings of three multipurpose agroforestry trees species of central India. Range Manag Agrofor 41:182–187
Sultana N, Limon SH, Rahman MS et al (2021) Germination and growth responses to water stress of three agroforestry tree species from Bangladesh. Environ Challenges 5:100256. https://doi.org/10.1016/j.envc.2021.100256
ICAR-CAFRI (2019) Annual Report. Jhansi, India
Krause GHWE (1991) Chlorophyll fluorescence and photosynthesis: the basics. Annu Rev Plant Physiol Plant Mol Biol 42:313–349
Lichtenthaler HK, Buschmann C (2001) Chlorophylls and carotenoids: Measurement and characterization by UV-VIS spectroscopy. Curr Protoc food Anal Chem 1:F4–F3
Rao L, Perez D, White E (1996) Lamin proteolysis facilitates nuclear events during apoptosis. J Cell Biol 135:1441–1455
Jebara S, Jebara M, Limam F, Aouani ME (2005) Changes in ascorbate peroxidase, catalase, guaiacol peroxidase and superoxide dismutase activities in common bean (Phaseolus vulgaris) nodules under salt stress. J Plant Physiol 162:929–936
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Lee YP, Takahashi T (1966) An improved colorimetric determination of amino acids with the use of ninhydrin. Anal Biochem 14:71–77
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Rozen S, Skaletsky H (2000) Primer3 on the WWW for general users and for biologist programmers. Bioinformatics methods and protocols. Springer, pp 365–386
Kozera B, Rapacz M (2013) Reference genes in real-time PCR. J Appl Genet 54:391–406
Alam B, Singh R, Uthappa AR et al (2018) Different genotypes of Dalbergia sissoo trees modified microclimate dynamics differently on understory crop cowpea (Vigna unguiculata) as assessed through ecophysiological and spectral traits in agroforestry system. Agric For Meteorol 249:138–148
Mafakheri A, Siosemardeh A, Bahramnejad B et al (2010) Effect of drought stress on yield, proline and chlorophyll contents in three chickpea cultivars. Aust J Crop Sci 4:580–585
Rajarajan K, Ganesamurthy K, Raveendran M et al (2021) Differential responses of sorghum genotypes to drought stress revealed by physio-chemical and transcriptional analysis. Mol Biol Rep 48:2453–2462
Kapanigowda MH(2011) Quantitative trait locus (QTL) mapping of transpiration efficiency related to pre-flower drought tolerance in sorghum [Sorghum bicolor (L.) Moench]. Texas A&M University
Hessini K, Kronzucker HJ, Abdelly C, Cruz C (2017) Drought stress obliterates the preference for ammonium as an N source in the C4 plant Spartina alterniflora. J Plant Physiol 213:98–107. https://doi.org/10.1016/j.jplph.2017.03.003
Awasthi R, Kaushal N, Vadez V, Turner NC, Berger J, Siddique KH (2014) Individual and combined effects of transient drought and heat stress on carbon assimilation and seed filling in chickpea. Funct Plant Biol 41:1148–1167
Chen D, Wang S, Cao B et al (2016) Genotypic variation in growth and physiological response to drought stress and re-watering reveals the critical role of recovery in drought adaptation in maize seedlings. Front Plant Sci 6:1241
Camilo S, Odindo AO, Kondwakwenda A, Sibiya J (2021) Root traits related with drought and phosphorus tolerance in common bean (Phaseolus vulgaris L.). Agronomy 11:552
Eldhuset TD, Nagy NE, Volařík D et al (2013) Drought affects tracheid structure, dehydrin expression, and above-and belowground growth in 5-year-old Norway spruce. Plant Soil 366:305–320
Zang C, Hartl-Meier C, Dittmar C et al (2014) Patterns of drought tolerance in major European temperate forest trees: climatic drivers and levels of variability. Glob Chang Biol 20:3767–3779
Ye G, Zhang H, Chen B et al (2019) De novo genome assembly of the stress tolerant forest species Casuarina equisetifolia provides insight into secondary growth. Plant J 97:779–794. https://doi.org/10.1111/tpj.14159
Comas L, Becker S, Cruz VMV et al (2013) Root traits contributing to plant productivity under drought. Front Plant Sci 4:442
Armas C, Padilla FM, Pugnaire FI, Jackson RB (2010) Hydraulic lift and tolerance to salinity of semiarid species: Consequences for species interactions. Oecologia 162:11–21. https://doi.org/10.1007/s00442-009-1447-1
Contreras-Soto RI, Rafael DZ, Moiana LD et al(2022) Variation in Root-Related Traits Is Associated With Water Uptake in Lagenaria siceraria Genotypes Under Water-Deficit Conditions.Front Plant Sci13
Lohithaswa HC, Desai SA, Hanchinal RR et al(2014) Combining ability in tetraploid wheat for yield, yield attributing traits, quality and rust resistance over environments.Karnataka J Agric Sci26
Liu C, Liu Y, Guo K et al (2011) Effect of drought on pigments, osmotic adjustment and antioxidant enzymes in six woody plant species in karst habitats of southwestern China. Environ Exp Bot 71:174–183. https://doi.org/10.1016/j.envexpbot.2010.11.012
Uarrota VG, Stefen DLV, Leolato LS et al (2018) Revisiting carotenoids and their role in plant stress responses: from biosynthesis to plant signaling mechanisms during stress. Antioxidants and antioxidant enzymes in higher plants. Springer, pp 207–232
Khazaei Z, Esmaielpour B, Estaji A (2020) Ameliorative effects of ascorbic acid on tolerance to drought stress on pepper (Capsicum annuum L) plants. Physiol Mol Biol Plants 26:1649–1662
Hou X, Rivers J, León P et al (2016) Synthesis and function of apocarotenoid signals in plants. Trends Plant Sci 21:792–803
Yang F, Zhang Q, Yao Q et al(2020) Direct and indirect plant defenses induced by (Z)-3-hexenol in tomato against whitefly attack. J Pest Sci (2004) 93:1243–1254
Sarker U, Oba S (2018) Catalase, superoxide dismutase and ascorbate-glutathione cycle enzymes confer drought tolerance of Amaranthus tricolor. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-34944-0
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930. https://doi.org/10.1016/j.plaphy.2010.08.016
Khaleghi A, Naderi R, Brunetti C et al (2019) Morphological, physiochemical and antioxidant responses of Maclura pomifera to drought stress. Sci Rep 9:1–12
Petridis A, Therios I, Samouris G et al (2012) Effect of water deficit on leaf phenolic composition, gas exchange, oxidative damage and antioxidant activity of four Greek olive (Olea europaea L.) cultivars. Plant Physiol Biochem 60:1–11. https://doi.org/10.1016/j.plaphy.2012.07.014
Xing H, Fu X, Yang C et al (2018) Genome-wide investigation of pentatricopeptide repeat gene family in poplar and their expression analysis in response to biotic and abiotic stresses. Sci Rep 8:2817. https://doi.org/10.1038/s41598-018-21269-1
Wu S, Tian J, Ren Y, Wang Y(2022) Osmotic adjustment and antioxidant system regulated by nitrogen deposition improve photosynthetic and growth performance and alleviate oxidative damage in dwarf bamboo under drought stress.Front Plant Sci1009
Zhou Z, Li G, Chao W et al (2019) Physiological responses and tolerance evaluation of five poplar varieties to waterlogging. Not Bot Horti Agrobot Cluj-Napoca 47:658–667. https://doi.org/10.15835/nbha47311440
Galeano E, Vasconcelos TS, De Oliveira PN, Carrer H (2019) Physiological and molecular responses to drought stress in teak (Tectona grandis L.f.). PLoS ONE 14:1–26. https://doi.org/10.1371/journal.pone.0221571
Acosta-Pérez P, Camacho-Zamora BD, Espinoza-Sánchez EA et al (2020) Characterization of trehalose-6-phosphate synthase and trehalose-6-phosphate phosphatase genes and analysis of its differential expression in maize (Zea mays) seedlings under drought stress. Plants 9. https://doi.org/10.3390/plants9030315
Yang Y, Guo Y, Zhong J et al (2020) Root Physiological Traits and Transcriptome Analyses Reveal that Root Zone Water Retention Confers Drought Tolerance to Opisthopappus taihangensis. Sci Rep 10:2627. https://doi.org/10.1038/s41598-020-59399-0
Lin Q, Yang J, Wang Q et al (2019) Overexpression of the trehalose-6-phosphate phosphatase family gene AtTPPF improves the drought tolerance of Arabidopsis thaliana. BMC Plant Biol 19:1–15. https://doi.org/10.1186/s12870-019-1986-5
Joshi R, Sahoo KK, Singh AK et al (2020) Enhancing trehalose biosynthesis improves yield potential in marker-free transgenic rice under drought, saline, and sodic conditions. J Exp Bot 71:653–668. https://doi.org/10.1093/jxb/erz462
Nuccio ML, Wu J, Mowers R et al (2015) Expression of trehalose-6-phosphate phosphatase in maize ears improves yield in well-watered and drought conditions. Nat Biotechnol 33:862–869
Sah SK, Reddy KR, Li J (2016) Abscisic acid and abiotic stress tolerance in crop plants. Front Plant Sci 7:1–26. https://doi.org/10.3389/fpls.2016.00571
Fujita Y, Fujita M, Shinozaki K, Yamaguchi-Shinozaki K (2011) ABA-mediated transcriptional regulation in response to osmotic stress in plants. J Plant Res 124:509–525. https://doi.org/10.1007/s10265-011-0412-3
Aghaie P, Tafreshi SAH (2020) Central role of 70-kDa heat shock protein in adaptation of plants to drought stress. Cell Stress Chaperones 25:1071–1081
Xiang J, Chen X, Hu W et al (2018) Overexpressing heat-shock protein OsHSP50.2 improves drought tolerance in rice. Plant Cell Rep 37:1585–1595. https://doi.org/10.1007/s00299-018-2331-4
Rodrigues MI, Takeda AAS, Bravo JP, Maia IG (2016) The eucalyptus tonoplast intrinsic protein (TIP) gene subfamily: Genomic organization, structural features, and expression profiles. Front Plant Sci 7:1–14. https://doi.org/10.3389/fpls.2016.01810
Khan MIR, Fatma M, Per TS et al (2015) Salicylic acid-induced abiotic stress tolerance and underlying mechanisms in plants. Front Plant Sci 6:1–17. https://doi.org/10.3389/fpls.2015.00462
Yuan X, Huang P, Wang R et al (2018) A zinc finger transcriptional repressor confers pleiotropic effects on rice growth and drought tolerance by down-regulating stress-responsive genes. Plant Cell Physiol 59:2129–2142. https://doi.org/10.1093/pcp/pcy133
Han G, Lu C, Guo J et al (2020) C2H2 Zinc Finger Proteins: Master Regulators of Abiotic Stress Responses in Plants. Front Plant Sci 11:1–13. https://doi.org/10.3389/fpls.2020.00115
Spollen WG, Lenoble ME, Samuels TD et al (2000) Abscisic acid accumulation maintains maize primary root elongation at low water potentials by restricting ethylene production. Plant Physiol 122:967–976. https://doi.org/10.1104/pp.122.3.967
Kuluev BR, Musin KhG, Mikhaylova ZAB, Zaikina EV(2019) EA Role of the expansin and xyloglucan endotransglycosylase genes in the regulation of tobacco growth under the influence of abiotic stress factors. Plant Biotechnol postgenomic era 135–136
Acknowledgements
The authors are grateful to Indian Council of Agricultural Research (ICAR), New Delhi, India, for facilities to complete this work.
Funding
No funding has been received separately for completion of this work.
Author information
Authors and Affiliations
Contributions
KR conceived the idea. KR, SS, HA and AA wrote the main manuscript text. KR, PT, ST, SS and AK prepared the manuscript. KR, AB, AJ, AR and revised the manuscript at different stages of the writing process.
Corresponding author
Ethics declarations
Conflict of interest
All the Authors declared that no conflicts of interest are associated with this publication.
Consent to Participate
This article does not contain any studies with human participants performed by any of the authors.
Consent to Publish
The Author’s hereby consents to publication of the Work in Molecular Biology Reports Journal.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rajarajan, K., Sakshi, S., Taria, S. et al. Whole plant response of Pongamia pinnata to drought stress tolerance revealed by morpho-physiological, biochemical and transcriptome analysis. Mol Biol Rep 49, 9453–9463 (2022). https://doi.org/10.1007/s11033-022-07808-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07808-0