Abstract
Backgrounds
Malignant melanoma is an aggressive skin tumor with a rapidly increasing incidence and there is not yet a successful treatment strategy. Vulpinic acid (VA) is derived from secondary metabolites from lichen species. In the current study, we, for the first time, investigated the anti-cancer effects of VA and the underlying mechanism VA induced programmed cell death in melanoma.
Methods
The anti-cancer effects of VA on melanoma cells were evaluated by the xCELLigence system, flow cytometry, caspase-3 activity and RT-PCR analysis.
Results
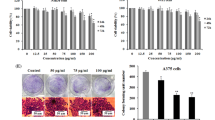
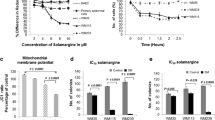
Our results showed that VA had a strong anti-proliferative effect on A-375 melanoma cells without damaging human epidermal melanocyte cells. Additionally, VA promoted apoptotic cell death through G2/M arrest and the activation of both intrinsic and extrinsic apoptosis pathways according to the analysis of 88 genes associated with apoptosis by qRT-PCR.
Conclusions
Our findings suggest that VA could become an alternative topical and transdermal treatment strategy in the treatment of maligned melanoma cancer. However, further investigations are needed to assess the underlying molecular mechanism of VA mediated apoptotic cell death in the treatment of melanoma.




Similar content being viewed by others
References
O’Neill CH, Scoggins CR (2019) Melanoma. J Surg Oncol 120(5):873–881. https://doi.org/10.1002/jso.25604
American Cancer Society (2019) Cancer facts & Fig. 2019. Atlanta: American Cancer Society 1 – 4
Lo JA, Fisher DE (2014) The melanoma revolution: from UV carcinogenesis to a new era in therapeutics. Science 346(6212):945–949. https://doi.org/10.1126/science.1253735
Jenkins RW, Fisher DE (2021) Treatment of Advanced Melanoma in 2020 and Beyond. J Invest Dermatol 141(1):23–31. https://doi.org/10.1016/j.jid.2020.03.943
Micali G, Lacarrubba F, Nasca MR, Schwartz RA (2014) Topical pharmacotherapy for skin cancer: part I. Pharmacology. J Am Acad Dermatol 70(6):965. https://doi.org/10.1016/j.jaad.2013.12.045. .e1 – 978
Abbas K, Qadir M, Anwar S (2019) The Role of Melanin in Skin Cancer. Crit Rev Eukaryot Gene Expr 29(1):17–24. https://doi.org/10.1615/CritRevEukaryotGeneExpr.2018024980
Gray-Schopfer V, Wellbrock C, Marais R (2007) Melanoma biology and new targeted therapy. Nature 445(7130):851–857. https://doi.org/10.1038/nature05661
Hammerová J, Uldrijan S, Táborská E, Slaninová I (2011) Benzo[c]phenanthridine alkaloids exhibit strong anti-proliferative activity in malignant melanoma cells regardless of their p53 status. J Dermatol Sci 62(1):22–35. https://doi.org/10.1016/j.jdermsci.2011.01.006
Yi SA, Nam KH, Kim S, So HM, Ryoo R, Han JW, Kim KH, Lee J (2019) Vulpinic Acid Controls Stem Cell Fate toward Osteogenesis and Adipogenesis. Genes 11(1):18. https://doi.org/10.3390/genes11010018
[Kılıç N, Derici MK, Büyük İ, Aydın SS, Aras S, Cansaran-Duman D (2018) Evaluation of in vitro Anticancer Activity of Vulpinic Acid and its Apoptotic Potential Using Gene Expression and Protein Analysis. Indian J Pharm Edu Res 52(4):626–634. https://doi.org/10.5530/ijper.52.4.73
Kwon Y, Cha J, Chiang J, Tran G, Giaever G, Nislow C, Hur JS, Kwak YS (2016) A chemogenomic approach to understand the antifungal action of Lichen-derived vulpinic acid. J Appl Microbiol 121(6):1580–1591. https://doi.org/10.1111/jam.13300
Kwon HY, Kim KS, Baik JS, Moon HI, Lee JW, Kim CH et al (2013) Tryptoplide-Mediated Apoptosis by Suppression of Focal Adhesion Kinase through Extrinsic and Intrinsic Pathways in Human Melanoma Cells. Evid Based Compl Alter Med 172548. https://doi.org/10.1155/2013/172548
Shrestha G, Thompson A, Robison R, St Clair LL (2016) Letharia vulpina, a vulpinic acid containing lichen, targets cell membrane and cell division processes in methicillin-resistant Staphylococcus aureus. Pharm Biol 54(3):413–418. https://doi.org/10.3109/13880209.2015.1038754
Rundel PW (1978) The ecological role of secondary lichen substances. Biochem Syst Ecol 6:157–170
Molnár K, Farkas E (2010) Current results on biological activities of lichen secondary metabolites: a review. Z Naturforsch C J Biosci 65(3–4):157–173. https://doi.org/10.1515/znc-2010-3-401
Koparal AT (2015) Anti-angiogenic and antiproliferative properties of the lichen substances (-)-usnic acid and vulpinic acid. Z Naturforsch C J Biosci 70(5–6):159–164. https://doi.org/10.1515/znc-2014-4178
Kılıç N, Aras S, Cansaran-Duman D (2018) Determination of Vulpinic Acid Effect on Apoptosis and mRNA Expression Levels in Breast Cancer Cell Lines. Anticancer Agents Med Chem 18(14):2032–2041. https://doi.org/10.2174/1871520618666180903101803
Kim S, So H, Roh H, Kim J, Yu J, Lee S et al (2017) Vulpinic acid contributes to the cytotoxicity of Pulveroboletus rhubarbelii to human cancer cells by inducing apoptosis. RSC Adv 7:35297–35304
Adewale FO, Basiru AO, Ayorinde OO, Israel OI, Oluwafemi OA (2017) Regulation of apoptotic and necroptotic cell death in skin cancer. J Cancer Biol Res 5(4):1108
Broussard L, Howland A, Ryu S, Song K, Norris D, Armstrong CA, Song PI (2018) Melanoma Cell Death Mechanisms. Chonnam Med J 54(3):135–142. https://doi.org/10.4068/cmj.2018.54.3.135
Mattia G, Puglisi R, Ascione B, Malorni W, Carè A, Matarrese P (2018) Cell death-based treatments of melanoma:conventional treatments and new therapeutic strategies. Cell Death Dis 9(2):112. https://doi.org/10.1038/s41419-017-0059-7
Hussein MR, Haemel AK, Wood GS (2003) Apoptosis and melanoma: molecular mechanisms. J Pathol 199(3):275–288. https://doi.org/10.1002/path.1300
Galluzzi L, Vitale I, Aaronson SA et al (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death Differ 2018. Cell Death Differ 25:486–541. https://doi.org/10.1038/s41418-017-0012-4
Karaosmanoglu O, Sivas H, Tay T, Turkish A (2015) The vitro investigation of cytotoxic and apoptotic effects vulpinic acid on normal and cancer cells. J Biotechnol 208:S95
Varol M, Turk A, Candan M, Tay T, Koparal AT (2016) Photoprotective Activity of Vulpinic and Gyrophoric Acids Toward Ultraviolet B-Induced Damage in Human Keratinocytes. Phytother Res 30(1):9–15. https://doi.org/10.1002/ptr.5493
Cansaran-Duman D, Guney Eskiler G, Colak B, Sozen Kucukkara E (2021) Vulpinic acid as a natural compound inhibits the proliferation of metastatic prostate cancer cells by inducing apoptosis. Mol Biol Rep 48(8):6025–6034. https://doi.org/10.1007/s11033-021-06605-5
Hamsa TP, Kuttan G (2011) Harmine activates intrinsic and extrinsic pathways of apoptosis in B16F-10 melanoma. Chin Med 6(1):11. https://doi.org/10.1186/1749-8546-6-11
Ramirez M, Salvesen GS (2018) A primary on caspase mechanisms. Semin Cell Dev Biol 82:79–85. https://doi.org/10.1016/j.semcdb.2018.01.002
Arch RH, Gedrich RW, Thompson CB (1998) Tumor necrosis factor receptor-associated factors (TRAFs)--a family of adapter proteins that regulate life and death. Genes Dev 12:2821–2830. https://doi.org/10.1101/gad.12.18.2821
Luo Z, Zhang X, Zeng W, Water J, Yang K, Lu L et al (2016) TRAF6 regulates melanoma invasion and metastasis through ubiquitination of Basigin. Oncotarget 7(6):7179–7192. https://doi.org/10.18632/oncotarget.6886
Beroukhim R, Mermel CH, Porter D et al (2010) The landscape of somatic copy-number alteration across human cancers. Nature 463:899–905. https://doi.org/10.1038/nature08822
Starczynowski DT, Lockwood WW, Deléhouzée S, Chari R, Wegrzyn J, Fuller M, Tsao MS et al (2011) TRAF6 is an amplified oncogene bridging the RAS and NF-εB pathways in human lung cancer. J Clin Invest 121(10):4095–4105. https://doi.org/10.1172/JCI58818
Meng Q, Zheng M, Liu H, Song C, Zhang W, Yan J, Qin L, Liu X (2012) TRAF6 regulates proliferation, apoptosis, and invasion of osteosarcoma cell. Mol Cell Biochem 371(1–2):177–186. https://doi.org/10.1007/s11010-012-1434-4
Chung JY, Park YC, Eat H, Wu H (2002) All TRAFs are not created equal: common and distinct molecular mechanisms of TRAF-mediated signal transduction. J Cell Sci 115(Pt 4):679–688
Rajandram R, Bennett NC, Wang Z, Perry-Keene J, Vesey DA, Johnson DW, Gobe GC (2012) Patient samples of renal cell carcinoma show reduced expression of TRAF1 compared with normal kidney and functional studies in vitro indicate TRAF1 promotes apoptosis: potential for targeted therapy. Pathology 44(5):453–459. https://doi.org/10.1097/PAT.0b013e3283557748
Vince JE, Pantaki D, Feltham R, Mace PD, Cordier SM, Schmukle AC, Davidson AJ, e (2009) TRAF2 must bind to cellular inhibitors of apoptosis for tumor necrosis factor (tnf) to efficiently activate nf-{kappa}b and to prevent tnf-induced apoptosis. J Biol Chem 284(51):35906–35915. https://doi.org/10.1074/jbc.M109.072256
Elbadawy M, Usui T, Yamawaki H, Sasaki K (2018) Novel functions of death-associated protein kinases through mitogen-activated protein kinase-related signals. Int J Mol Sci 19(10):3031. https://doi.org/10.3390/ijms19103031
van den Oord JJ, Maes A, Stas M, Nuyts J, Battocchio S, Kasran A, De Wolf-Peeters C (1996) CD40 is a prognostic marker in primary cutaneous malignant melanoma. Am J Pathol 149(6):1953
Georgopoulos NT, Steele LP, Thomson MJ, Selby PJ, Southgate J, Trejdosiewicz LK (2006) A novel mechanism of CD40-induced apoptosis of carcinoma cells involving TRAF3 and JNK/AP-1 activation. Cell Death Differ 13(10):1789–1801. https://doi.org/10.1038/sj.cdd.4401859
Wong RS (2011) Apoptosis in cancer: from pathogenesis to treatment. J Exp Clin Cancer Res 30(1):87. https://doi.org/10.1186/1756-9966-30-87
Akl H, Vervloessem T, Kiwiluoto S, Bittremieux M, Parys JB, De Smedt H, Bultynck G (2014) A dual role for the anti-apoptoctic Bcl-2 protein in cancer: mitochondria versus endoplasmic reticulum. Biochim Biophys Acta 1843(10):2240–2252. https://doi.org/10.1016/j.bbamcr.2014.04.017
Lindqvist LM, Simon AK, Baehrecke EH (2015) Current questions and possible controversies in autophagy. Cell Death Discov 1:15036. https://doi.org/10.1038/cddiscovery.2015.36
Hatok J, Racay P (2016) Bcl-2 family proteins: master regulators of cell survival. Biomol Concepts 7(4):259–270. https://doi.org/10.1515/bmc-2016-0015
Shakeri R, Kheirollahi A, Davoodi J (2017) Apaf-1: Regulation and function in cell death. Biochimie 135:111–125. https://doi.org/10.1016/j.biochi.2017.02.001
Steeland S, Libert C, Vandenbroucke RE (2018) A new venue of TNF targeting. Int J Mol Sci 19(5):1442. https://doi.org/10.3390/ijms19051442
Acknowledgements
This research was partly supported by the research grants from the Scientific and Technological Research Council of Turkey (TUBITAK, Project no. 216S583).
Author information
Authors and Affiliations
Contributions
Conceptualization, D.C.D.; investigation, S.Y.; writing—original draft preparation, S.Y., G.G.E., S.A.; writing—review and editing, D.C.D.; supervision, D.C.D. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics approval
This study does not require an ethics committee decision.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yangın, S., Cansaran-Duman, D., Eskiler, G.G. et al. The molecular mechanisms of vulpinic acid induced programmed cell death in melanoma. Mol Biol Rep 49, 8273–8280 (2022). https://doi.org/10.1007/s11033-022-07619-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07619-3