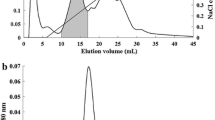
Abstract
Background
Hepatocellular carcinoma (HCC) is the main types of primary liver cancer, which shows some abnormal glycosylation, such as the increase of fucose. Lens culinaris agglutinin (LCA), a natural plant lectin that can bind to mannose and fucose, has been reported to be antiproliferative to may tumors. However, the effect of LCA on the vitality and migration ability of human hepatoma cells is not demonstrated. Therefore, the aim of this study is to investigate the effects of LCA on vitality and migration in human hepatoma cells and its potential mechanisms.
Methods and results
LCA had no significant effect on viability of human hepatoma cells (HCCLM3, MHCC97L and HepG2) and hepatocytes (L02) by CCK-8 kit, but it could inhibit human hepatoma cells migration significantly without affecting hepatocytes by Transwell method. Sugar inhibition assay was used to verify the possible binding site between LCA and human hepatoma cells. The result showed that Mannose- and fucose- related sites were associated with LCA inhibiting human hepatoma cells migration. Moreover, LCA could affect HCCLM3 migration by activating ERK1/2 and JNK1/2/3 signalling pathways. LCA did not affect MMP-2 and MMP-9 of HCCLM3 through gelatinase zymography. However, the results of immunofluorescence standing showed that LCA could reduce the F-actin formation in HCCLM3 via ERK1/2 and JNK1/2/3 signalling pathways.
Conclusions
LCA might inhibit human hepatoma cell migration by reducing the F-actin formation via the mannose and fucose-mediated ERK1/2 and JNK1/2/3 signalling pathway. This result will deepen people’s understanding on plant lectin as a drug in tumor glycobiology.





Similar content being viewed by others
References
Feng M, Pan Y, Kong R, Shu S (2020) Therapy of primary liver cancer. The Innovation 1:100032. https://doi.org/10.1016/j.xinn.2020.100032
Oliveira-Ferrer L, Legler K, Milde-Langosch K (2017) Role of protein glycosylation in cancer metastasis. Semin Cancer Bio 44:141–152. https://doi.org/10.1016/j.semcancer.2017.03.002
Sachs J, Kadioglu O, Weber A, Mundorf V, Betz J, Efferth T, Pietruszka J, Teusch N (2019) Selective inhibition of P-gp transporter by goniothalamin derivatives sensitizes resistant cancer cells to chemotherapy. J Nat Med 73:226–235. https://doi.org/10.1007/s11418-018-1230-x
Lu L, Zhou J, Shi J, Peng XJ, Qi XX, Wang Y, Li FY, Zhou FY, Liu L, Liu ZQ (2015) Drug-metabolizing activity, protein and gene expression of UDP-glucuronosyltransferases are significantly altered in hepatocellular carcinoma patients. PLoS ONE 10:e0127524. https://doi.org/10.1371/journal.pone.0127524
Barre A, Bourne Y, Van Damme EJ, Rougé P (2019) Overview of the structure-function relationships of mannose-specific lectins from plants, algae and fungi. Int J Mol Sci 20:254. https://doi.org/10.3390/ijms20020254
van Holle S, van Damme EJM (2019) Messages from the past: new insights in plant lectin evolution. Front Plant Sci 10:36. https://doi.org/10.3389/fpls.2019.00036
Mazalovska M, Kouokam JC (2020) Plant-derived lectins as potential cancer therapeutics and diagnostic tools. Bio Med Res Int 19:1–13. https://doi.org/10.1155/2020/1631394
van Holle S, van Damme EJ (2015) Distribution and evolution of the lectin family in soybean (Glycine max). Molecules 20:2868–2891. https://doi.org/10.3390/molecules20022868
Diwan D, Shinkai K, Tetsuka T, Cao B, Arai H, Koyama T, Hatano K, Matsuoka K (2017) Synthetic assembly of mannose moieties using polymer chemistry and the biological evaluation of its interaction towards concanavalin a. Molecules 22:157. https://doi.org/10.3390/molecules22010157
Wang W, Yang Y, Wang S, Nagaraj VJ, Liu Q, Wu J, Tao N (2012) Label-free measuring and mapping of binding kinetics of membrane proteins in single living cells. Nat Chem 4:846–853. https://doi.org/10.1038/nchem
Mishra A, Behura A, Mawatwal S, Kumar A, Naik L, Mohanty SS, Manna D, Dokania P, Mishra A, Patra S, Dhiman R (2019) Structure-function and application of plant lectins in disease biology and immunity. Food Chem Toxicol 134:110827. https://doi.org/10.1016/j.fct.2019.110827
Nascimento APM, Wolin IA, Welter PG, Heinrich IA, Zanotto-Filho A, Osterne VJS, Lossio CF, Silva MTL, Nascimento KS, Cavada BS, Leal RB (2019) Lectin from Dioclea violacea induces autophagy in U87 glioma cells. Int J Biol Macromol 134:660–672. https://doi.org/10.1016/j.ijbiomac.2019.04.203
Peng H, Lv H, Wang Y, Liu HY, Li CY, Meng L, Chen F, Bao JK (2009) Clematis montana lectin, a novel mannose-binding lectin from traditional Chinese medicine with antiviral and apoptosis-inducing activities. Peptides 30:1805–1815. https://doi.org/10.1016/j.peptides.2009.06.027
Perduca M, Dalle Carbonare L, Bovi M, Innamorati G, Cheri S, Cavallini C, Scupoli MT, Mori A, Valenti MT (2017) Runx2 downregulation, migration and proliferation inhibition in melanoma cells treated with BEL β-trefoil. Oncol Rep 37:2209–2214. https://doi.org/10.3892/or.2017.5493
Matsuda A, Kuno A, Ishida H, Kawamoto T, Shoda J, Hirabayashi J (2008) Development of an all-in-one technology for glycan profiling targeting formalin-embedded tissue sections. Biochem Biophys Res Commun 370:259–263. https://doi.org/10.1016/j.bbrc.2008.03.090
Loris R, Casset F, Bouckaert J, Pletinckx J, Dao-Thi MH, Poortmans F, Imberty A, Perez S, Wyns L (1994) The monosaccharide binding site of lentil lectin: an X-ray and molecular modelling study. Glycoconj J 11:507–517. https://doi.org/10.1007/bf00731301
Hwang H, Jeong HK, Lee HK, Park GW, Lee JY, Lee SY, Kang YM, An HJ, Kang JG, Ko JH, Kim JY, Yoo JS (2020) Machine learning classifies core and outer fucosylation of N-glycoproteins using mass spectrometry. Sci Rep 10:2879. https://doi.org/10.1038/s41598-019-57274-1
Wongjarupong N, Negron-Ocasio G, Chaiteerakij R, Addissie B, Mohamed E, Mara K, Harmsen W, Theobald JP, Peters BE, Balsanek JG (2018) Model combining pre-transplant tumor biomarkers and tumor size shows more utility in predicting hepatocellular carcinoma recurrence and survival than the BALAD models. World J Gastroentero 24:1321–1331. https://doi.org/10.3748/wjg.v24.i12.1321
Comunale MA, Lowman M, Long RE, Krakover J, Philip R, Seeholzer S, Evans AA, Hann HW, Block TM, Mehta AS (2006) Proteomic analysis of serum associated fucosylated glycoproteins in the development of primary hepatocellular carcinoma. J Proteome Res 5:308–315. https://doi.org/10.1021/pr050328x
Kang X, Wang N, Pei C, Sun L, Sun R, Chen J, Liu Y (2012) Glycan-related gene expression signatures in human metastatic hepatocellular carcinoma cells. Exp Ther Med 3:415–422. https://doi.org/10.3892/etm.2011.430
Shang S, Li W, Qin X, Zhang S, Liu YK (2017) Aided diagnosis of hepatocellular carcinoma using serum fucosylated haptoglobin ratios. J Cancer 8:887–893. https://doi.org/10.7150/jca.17747
Tang ZY, Ye SL, Liu YK, Qin LX, Sun HC, Ye QH, Wang L, Zhou J, Qiu SJ, Li Y (2004) A decade’s studies on metastasis of hepatocellular carcinoma. J Cancer Res Clin Oncol 130:187–196. https://doi.org/10.1007/s00432-003-0511-1
Tang H, Li R, Liang P, Zhou YL, Wang GW (2015) miR125a inhibits the migration and invasion of liver cancer cells via suppression of the PI3K/AKT/mTOR signaling pathway. Oncol Lett 10:681–686. https://doi.org/10.3892/ol.2015.3264
Luo G, Chao YL, Tang B, Li BS, Xiao YF, Xie R, Wang SM, Wu YY, Dong H, Liu XD, Yang SM (2015) miR-149 represses metastasis of hepatocellular carcinoma by targeting actin-regulatory proteins PPM1F. Oncotarget 6:37808–37823. https://doi.org/10.18632/oncotarget.5676
Chan YS, Yu H, Xia L, Ng TB (2015) Lectin from green speckled lentil seeds (Lens culinaris) triggered apoptosis in nasopharyngeal carcinoma cell lines. Chin Med 10:25. https://doi.org/10.1186/s13020-015-0057-6
Lei HY, Chang CP (2009) Lectin of Concanavalin A as an anti-hepatoma therapeutic agent. J Biomed Sci 6:10. https://doi.org/10.1186/1423-0127-16-10
Hung LD, Trinh PTH (2021) Structure and anticancer activity of a new lectin from the cultivated red alga, Kappaphycus striatus. J Nat Med 75:223–231. https://doi.org/10.1007/s11418-020-01455-0
Hyuk M, Simon WR (2021) Mapk/erk signaling pathway in hepatocellular carcinoma. Cancers 13:3026. https://doi.org/10.3390/cancers13123026
Dong XZ, Song Y, Lu YP, Hu Y, Liu P, Zhang L (2019) Sanguinarine inhibits the proliferation of BGC-823 gastric cancer cells via regulating miR-96-5p/miR-29c-3p and the MAPK/JNK signaling pathway. J Nat Med 73:777–788. https://doi.org/10.1007/s11418-019-01330-7
Kurosaki H, Nakatake M, Sakamoto T, Kuwano N, Yamane M, Ishii K, Fujiwara Y, Nakamura T (2021) Anti-tumor effects of MAPK-dependent tumor-selective oncolytic vaccinia virus armed with CD/UPRT against pancreatic ductal adenocarcinoma in mice. Cells 10:985. https://doi.org/10.3390/cells10050985
Xie SJ, Diao LT, Cai N, Zhang LT, Xiang S, Jia CC, Qiu DB, Liu C, Sun YJ, Lei H (2021) mascRNA and its parent lncRNA MALAT1 promote proliferation and metastasis of hepatocellular carcinoma cells by activating ERK/MAPK signaling pathway. Cell Death Discov 7:110. https://doi.org/10.1038/s41420-021-00497-x
Min J, Feng Q, Liao W, Liang Y, Gong C, Li E, He W, Yuan R, Wu L (2018) IFITM3 promotes hepatocellular carcinoma invasion and metastasis by regulating MMP9 through p38/MAPK signaling. FEBS Open Bio 8:1299–1311. https://doi.org/10.1002/2211-5463.12479
Simiczyjew A, Mazur AJ, Popow-Woz´niak A, Malicka-Błaszkiewicz M, Nowak D (2014) Effect of overexpression of β- and γ-actin isoforms on actin cytoskeleton organization and migration of human colon cancer cells. Histochem Cell Bio 142:307–322. https://doi.org/10.1007/s00418-014-1199-9
Ullo MF, Logue JS (2021) ADF and cofilin-1 collaborate to promote cortical actin flow and the leader bleb-based migration of confined cells. https://doi.org/10.1101/2021.01.11.426289. eLife 10
Rudzka DA, Spennati G, McGarry DJ, Chim YH, Neilson M, Ptak A, Munro J, Kalna G, Hedley A, Moralli D, Green C, Mason S, Blyth K, Mullin M, Yin H, Olson MF (2019) Migration through physical constraints is enabled by MAPK-induced cell softening via actin cytoskeleton re-organization. J Cell Sci 132:jcs224071. https://doi.org/10.1242/jcs.224071
Li H, Qiu Z, Li F, Wang C (2017) The relationship between MMP-2 and MMP-9 expression levels with breast cancer incidence and prognosis. Oncol Lett 14:5865–5870. https://doi.org/10.3892/ol.2017.6924
Oliveira I, Nunes A, Lima A, Borralho P, Rodrigues C, Ferreira RB, Ribeiro AC (2019) New lectins from mediterranean flora. activity against HT29 colon cancer cells. Int J Mol Sci 20:3059. https://doi.org/10.3390/ijms20123059
Jian Q, Yang Z, Shu J, Liu XW, Zhang J, Zheng L (2018) Lectin BS-I inhibits cell migration and invasion via AKT/GSK-3β/β-catenin pathway in hepatocellular carcinoma. J Cell Mol Med 22:315–329. https://doi.org/10.1111/jcmm.13320
Chen H, Qu J, Huang X, Kurundkar A, Zhu L, Yang N, Venado A, Ding Q, Liu G, Antony VB, Thannickal VJ, Zhou Y (2016) Mechanosensing by the α6-integrin confers an invasive fibroblast phenotype and mediates lung fibrosis. Nat Commun 18:12564. https://doi.org/10.1038/ncomms12564
Lee MS, Jung JI, Kwon SH, Lee SM, Morita K, Her S (2012) TIMP-2 fusion protein with human serum albumin potentiates anti-angiogenesis-mediated inhibition of tumor growth by suppressing MMP-2 expression. PLoS ONE 7:e35710. https://doi.org/10.1371/journal.pone.00357
Acknowledgements
This work was supported by the Science and Technology Research Program of Chongqing Municipal Education Commission [KJQN201800601, KJQN201900644] and the Natural Science Foundation of Chongqing [cstc2020jcyj-msxmX0760].
Funding
This work was supported by the Science and Technology Research Program of Chongqing Municipal Education Commission [KJQN201800601, KJQN201900644] and the Natural Science Foundation of Chongqing [cstc2020jcyj-msxmX0760].
Author information
Authors and Affiliations
Contributions
Conceptualization: Bingyu Zhang and Zhangyong Li; Methodology: Bingyu Zhang and Haoran Jiang; Formal analysis and investigation: Haoran Jiang, Xianxin Wen and Xue Zhang; Writing-original draft preparation: Haoran Jiang; Review and editing: Bingyu Zhang; Resources: Bingyu Zhang and Xianhua Zhong. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jiang, H., Wen, X., Zhang, X. et al. Lens culinaris agglutinin inhibits human hepatoma cell migration via mannose and fucose-mediated ERK1/2 and JNK1/2/3 signalling pathway. Mol Biol Rep 49, 7665–7676 (2022). https://doi.org/10.1007/s11033-022-07582-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07582-z