Abstract
Background
Discoidin domain receptor 1 (DDR1), a member of receptor tyrosine kinase, has been implicated in tumor progression. However, the function and underlying mechanism of DDR1 in lung adenocarcinoma (LUAD) progression is unclear. Thus, we explored the molecular regulatory mechanism of DDR1 in the migration of LUAD.
Methods
Transwell assays, wound healing assays and xenograft tumor assays were performed to study the function of DDR1 in the progression of LUAD. Immunoblotting and quantitative real-time polymerase chain reaction (RT-qPCR) were used to detect the expression levels of genes. Co-immunoprecipitation (co-IP) assays were performed to detect the interaction between DDR1 and AKT. Immunofluorescence and immunohistochemistry assays were used to determine the expression level of proteins in cells and tissues, respectively.
Results
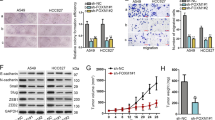
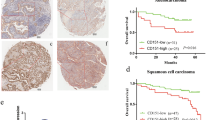
DDR1 expression was significantly higher in LUAD tissues than in normal lung tissues, and the level of DDR1 was inversely correlated with prognosis in patients. We found that DDR1 promoted the migration and invasion of LUAD cells in vitro. Furthermore, ectopic expression of DDR1 in LUAD cells altered EMT-related markers expression. Importantly, the DDR1 protein interacted with AKT and phosphorylated AKT. The AKT inhibitor MK2206 interrupted Snail upregulation in DDR1-overexpressing LUAD cells. Finally, our study revealed that depletion of DDR1 attenuated LUAD cell migration in a tumor xenograft mouse model.
Conclusion
Our findings uncovered that a high abundance of DDR1 increased the migration and invasion capability of LUAD cells via the AKT/Snail signaling axis and indicated that DDR1 could be a potential target for treating LUAD.





Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70(1):7–30
Herbst RS, Morgensztern D, Boshoff C (2018) The biology and management of non-small cell lung cancer. Nature 553(7689):446–454
Massague J, Obenauf AC (2016) Metastatic colonization by circulating tumour cells. Nature 529(7586):298–306
Lamouille S, Xu J, Derynck R (2014) Molecular mechanisms of epithelial–mesenchymal transition. Nat Rev Mol Cell Biol 15(3):178–196
Nieto MA, Huang RY, Jackson RA, Thiery JP (2016) Emt: 2016. Cell 166(1):21–45
Sinha D, Saha P, Samanta A, Bishayee A (2020) Emerging concepts of hybrid epithelial-to-mesenchymal transition in cancer progression. Biomolecules 10(11):1561
Dongre A, Weinberg RA (2019) New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol 20(2):69–84
Fazilaty H, Rago L, Kass Youssef K, Ocaña OH, Garcia-Asencio F, Arcas A et al (2019) A gene regulatory network to control EMT programs in development and disease. Nat Commun 10(1):1–16
Pastushenko I, Blanpain C (2019) EMT transition states during tumor progression and metastasis. Trends Cell Biol 29(3):212–226
Vogel W, Gish GD, Alves F, Pawson T (1997) The discoidin domain receptor tyrosine kinases are activated by collagen. Mol Cell 1(1):13–23
Kothiwale S, Borza CM, Lowe EW, Pozzi A, Meiler J (2015) Discoidin domain receptor 1 (DDR1) kinase as target for structure-based drug discovery. Drug Discov Today 20(2):255–261
Borza CM, Pozzi A (2014) Discoidin domain receptors in disease. Matrix Biol 34:185–192
Iwai LK, Luczynski MT, Huang PH (2014) Discoidin domain receptors: a proteomic portrait. Cell Mol Life Sci 71(17):3269–3279
Kadler KE, Baldock C, Bella J, Boot-Handford RP (2007) Collagens at a glance. J Cell Sci 120(Pt 12):1955–1958
Lemmon MA, Schlessinger J (2010) Cell signaling by receptor tyrosine kinases. Cell 141(7):1117–1134
Fu HL, Valiathan RR, Payne L, Kumarasiri M, Mahasenan KV, Mobashery S et al (2014) Glycosylation at Asn211 regulates the activation state of the discoidin domain receptor 1 (DDR1). J Biol Chem 289(13):9275–9287
Chen YL, Tsai WH, Ko YC, Lai TY, Cheng AJ, Shiah SG et al (2020) Discoidin domain receptor-1 (DDR1) is involved in angiolymphatic invasion in oral cancer. Cancers. https://doi.org/10.3390/cancers12040841
Yeh YC, Lin HH (1866) Tang MJ 2019 dichotomy of the function of DDR1 in cells and disease progression. Biochim Biophys Acta 11:118473
Rammal H, Saby C, Magnien K, Van-Gulick L, Garnotel R, Buache E et al (2016) Discoidin domain receptors: potential actors and targets in cancer. Front Pharmacol 7:55
Azizi R, Salemi Z, Fallahian F, Aghaei M (2019) Inhibition of didscoidin domain receptor 1 reduces epithelial–mesenchymal transition and induce cell-cycle arrest and apoptosis in prostate cancer cell lines. J Cell Physiol 234(11):19539–19552
Jin H, Ham IH, Oh HJ, Bae CA, Lee D, Kim YB et al (2018) Inhibition of discoidin domain receptor 1 prevents stroma-induced peritoneal metastasis in gastric carcinoma. Mol Cancer Res 16(10):1590–1600
Baltes F, Caspers J, Henze S, Schlesinger M, Bendas G (2020) Targeting discoidin domain receptor 1 (DDR1) signaling and its crosstalk with beta1-integrin emerges as a key factor for breast cancer chemosensitization upon collagen type 1 binding. Int J Mol Sci 21(14):4956
Vehlow A, Cordes N (2019) DDR1 (discoidin domain receptor tyrosine kinase 1) drives glioblastoma therapy resistance by modulating autophagy. Autophagy 15(8):1487–1488
Guan C, Zhang L, Wang S, Long L, Zhou H, Qian S et al (2019) Upregulation of MicroRNA-21 promotes tumorigenesis of prostate cancer cells by targeting KLF5. Cancer Biol Ther 20(8):1149–1161
Du L, Lee J-H, Jiang H, Wang C, Wang S, Zheng Z et al (2020) β-Catenin induces transcriptional expression of PD-L1 to promote glioblastoma immune evasion. J Exp Med. https://doi.org/10.1084/jem.20191115
Imani S, Hosseinifard H, Cheng J, Wei C, Fu J (2016) Prognostic value of EMT-inducing transcription factors (EMT-TFs) in metastatic breast cancer: a systematic review and meta-analysis. Sci Rep 6:28587
Lee MJ, Lee JH, Rubinsztein DC (2013) Tau degradation: the ubiquitin–proteasome system versus the autophagy-lysosome system. Prog Neurobiol 105:49–59
Hirsch FR, Scagliotti GV, Mulshine JL, Kwon R, Curran WJ, Wu Y-L et al (2017) Lung cancer: current therapies and new targeted treatments. Lancet 389(10066):299–311
Tan W-L, Jain A, Takano A, Newell EW, Iyer NG, Lim W-T et al (2016) Novel therapeutic targets on the horizon for lung cancer. Lancet Oncol 17(8):e347–e362
Yang SH, Baek HA, Lee HJ, Park HS, Jang KY, Kang MJ et al (2010) Discoidin domain receptor 1 is associated with poor prognosis of non-small cell lung carcinomas. Oncol Rep 24(2):311–319
Ford CE, Lau SK, Zhu CQ, Andersson T, Tsao MS, Vogel WF (2007) Expression and mutation analysis of the discoidin domain receptors 1 and 2 in non-small cell lung carcinoma. Br J Cancer 96(5):808–814
Miao L, Zhu S, Wang Y, Li Y, Ding J, Dai J et al (2013) Discoidin domain receptor 1 is associated with poor prognosis of non-small cell lung cancer and promotes cell invasion via epithelial-to-mesenchymal transition. Med Oncol 30(3):626
Valencia K, Ormazabal C, Zandueta C, Luis-Ravelo D, Anton I, Pajares MJ et al (2012) Inhibition of collagen receptor discoidin domain receptor-1 (DDR1) reduces cell survival, homing, and colonization in lung cancer bone metastasis. Clin Cancer Res 18(4):969–980
Miao L, Wang Y, Zhu S, Shi M, Li Y, Ding J et al (2014) Identification of novel driver mutations of the discoidin domain receptor 2 (DDR2) gene in squamous cell lung cancer of Chinese patients. BMC Cancer 24(14):369
Kobayashi-Watanabe N, Sato A, Watanabe T, Abe T, Nakashima C, Sueoka E, Kimura S, Sueoka-Aragane N (2017) Functional analysis of discoidin domain receptor 2 mutation and expression in squamous cell lung cancer. Lung Cancer 110:35–41
Gao H, Chakraborty G, Zhang Z, Akalay I, Gadiya M, Gao Y et al (2016) Multi-organ site metastatic reactivation mediated by non-canonical discoidin domain receptor 1 signaling. Cell 166(1):47–62
Riess JW, Neal JW (2012) Targeting fibroblast growth factor receptor and discoidin domain receptor 2 in non-small-cell lung cancer. J Thorac Oncol 7(16 Suppl 5):S385–S386
Payne LS, Huang PH (2014) Discoidin domain receptor 2 signaling networks and therapy in lung cancer. J Thorac Oncol 9(6):900–904
Guo G, Gong K, Wohlfeld B, Hatanpaa KJ, Zhao D, Habib AA (2015) Ligand-independent EGFR signaling. Cancer Res 75(17):3436–3441
Okabe T, Okamoto I, Tamura K, Terashima M, Yoshida T, Satoh T et al (2007) Differential constitutive activation of the epidermal growth factor receptor in non-small cell lung cancer cells bearing EGFR gene mutation and amplification. Cancer Res 67(5):2046–2053
Zhou BP, Deng J, Xia W, Xu J, Li YM, Gunduz M et al (2004) Dual regulation of Snail by GSK-3beta-mediated phosphorylation in control of epithelial–mesenchymal transition. Nat Cell Biol 6(10):931–940
Ryu K-J, Park S-M, Park S-H, Kim I-K, Han H, Kim H-J et al (2019) p38 stabilizes snail by suppressing DYRK2-mediated phosphorylation that is required for GSK3β-βTrCP–induced snail degradation. Cancer Res 79(16):4135–4148
Acknowledgements
We also thank the patients for their contribution in this study.
Funding
This work was supported by National Natural Science Foundation of China under (Grant No. 81902151 to L.D.); and Wenzhou Science & Technology Bureau Foundation (Grant No. Y20180074 to L.D).
Author information
Authors and Affiliations
Contributions
JL and LD conceived the study and designed the experiments; JZ, HC, LW, and WX performed the experiments and the statistical analyses; JW and QH collected the LUAD specimens; LD and JZ drafted the manuscript; and JL proofread the paper. All authors read and approved the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
All procedures performed in this study involving animal experiments approved by the Laboratory Animal Ethics Committee of Wenzhou Medical University.
Consent to participate
All the patients provided written informed consent to allow the use of their tissues in research and the use of human lung cancer specimens was approved by the Ethics Committee in first affiliated hospital of Wenzhou Medical University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhu, J., Cheng, H., Wang, L. et al. Discoidin domain receptor 1 promotes lung adenocarcinoma migration via the AKT/snail signaling axis. Mol Biol Rep 49, 7275–7286 (2022). https://doi.org/10.1007/s11033-022-07509-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07509-8