Abstract
Purpose
Kaempferol is a natural flavonoid that has been reported to be active against many cancers, including prostate cancer, breast cancer and colon cancer. In our previous study, we found kaempferol could act as a selective androgen receptor modulator, thereby suppress development of benign prostatic hyperplasia. This finding inspired us to further explore the effect and the mechanism of action of kaempferol on prostate cancer.
Methods
Plate clone formation assay was performed to detect the effect of kaempferol on cell proliferation. Flow cytometry was used to detect the impact of kaempferol on cell apoptosis and cell cycle distribution. qPCR, immunofluorescence staining, and enzyme-linked immunosorbent assay were used to detect the expression of gene and protein of Ki67 which is a biomarker of cell proliferation.
Results
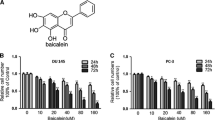
In the present study, we found kaempferol could dramatically suppress androgen-dependent and androgen-independent prostate cancer cells proliferation and induce their apoptosis. Furthermore, we found that kaempferol induced cell cycle to be arrested at G1 phase in 22Rv1 cells but at S and G2 phase in PC-3 cells. In addition, we detected the mRNA and protein of Ki67 which is corresponding to the cell proliferation and found that kaempferol could significantly inhibit Ki67 expression at mRNA level but increase its expression at protein levels in both androgen-dependent and androgen-independent prostate cancer cells.
Conclusion
Taken together, kaempferol inhibited the proliferation of androgen-dependent and androgen-independent prostate cancer cells by regulating the expression of Ki67. These findings further shed light on the mechanism of action of kaempferol on anti-prostate cancer.






Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A et al (2021) Global cancer statistics 2020: globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics. CA Cancer J Clin 72(1):7–33. https://doi.org/10.3322/caac.21708
de Wit R, de Bono J, Sternberg CN, Fizazi K, Tombal B, Wülfing C et al (2019) Cabazitaxel versus Abiraterone or Enzalutamide in metastatic prostate cancer. N Engl J Med 381(26):2506–2518
Hussain M, Fizazi K, Saad F, Rathenborg P, Shore N, Ferreira U et al (2018) Enzalutamide in men with nonmetastatic, castration-resistant prostate cancer. N Engl J Med 378(26):2465–2474
Chen AY, Chen YC (2013) A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem 138(4):2099–2107
Wang X, Yang Y, An Y, Fang G (2019) The mechanism of anticancer action and potential clinical use of kaempferol in the treatment of breast cancer. Biomed Pharmacother 117:109086
Wang X, Zhu J, Yan H, Shi M, Zheng Q, Wang Y et al (2021) Kaempferol inhibits benign prostatic hyperplasia by resisting the action of androgen. Eur J Pharmacol 907:174251
Da J, Xu M, Wang Y, Li W, Lu M, Wang Z (2019) Kaempferol promotes apoptosis while inhibiting cell proliferation via androgen-dependent pathway and suppressing vasculogenic mimicry and invasion in prostate cancer. Anal Cell Pathol (Amst) 2019:1907698
Knowles LM, Zigrossi DA, Tauber RA, Hightower C, Milner JA (2000) Flavonoids suppress androgen-independent human prostate tumor proliferation. Nutr Cancer 38(1):116–122
Berlin A, Castro-Mesta JF, Rodriguez-Romo L, Hernandez-Barajas D, González-Guerrero JF, Rodríguez-Fernández IA et al (2017) Prognostic role of Ki-67 score in localized prostate cancer: a systematic review and meta-analysis. Urol Oncol 35(8):499–506
Spratt DE (2018) Ki-67 remains solely a prognostic biomarker in localized prostate cancer. Int J Radiat Oncol Biol Phys 101(3):513–515
Wyatt RA, Trieu NPV, Crawford BD (2017) Zebrafish xenograft: an evolutionary experiment in tumour biology. Genes (Basel) 8(9):220
Brown HK, Schiavone K, Tazzyman S, Heymann D, Chico TJ (2017) Zebrafish xenograft models of cancer and metastasis for drug discovery. Expert Opin Drug Discov 12(4):379–389
Kapoor S (2018) Attenuating effect of kaempferol on neoplastic growth in different urological malignancies. Biomed Pharmacother 104:848
Dang Q, Song W, Xu D, Ma Y, Li F, Zeng J et al (2015) Kaempferol suppresses bladder cancer tumor growth by inhibiting cell proliferation and inducing apoptosis. Mol Carcinog 54(9):831–840
Mamouni K, Zhang S, Li X, Chen Y, Yang Y, Kim J et al (2018) A novel flavonoid composition targets androgen receptor signaling and inhibits prostate cancer growth in preclinical models. Neoplasia 20(8):789–799
Bandyopadhyay S, Romero JR, Chattopadhyay N (2008) Kaempferol and quercetin stimulate granulocyte-macrophage colony-stimulating factor secretion in human prostate cancer cells. Mol Cell Endocrinol 287(1–2):57–64
Sobecki M, Mrouj K, Colinge J, Gerbe F, Jay P, Krasinska L et al (2017) Cell-cycle regulation accounts for variability in ki-67 expression levels. Cancer Res. 77(10):2722–2734
Li LT, Jiang G, Chen Q, Zheng JN (2015) Ki67 is a promising molecular target in the diagnosis of cancer (review). Mol Med Rep 11(3):1566–1572
Green WJ, Ball G, Hulman G, Johnson C, Van Schalwyk G, Ratan HL et al (2016) KI67 and DLX2 predict increased risk of metastasis formation in prostate cancer-a targeted molecular approach. Br J Cancer 115(2):236–242
Lobo J, Rodrigues Â, Antunes L, Graça I, Ramalho-Carvalho J, Vieira FQ et al (2018) High immunoexpression of Ki67, EZH2, and SMYD3 in diagnostic prostate biopsies independently predicts outcome in patients with prostate cancer. Urol Oncol 36(4):161.e7-161.e17
Hammarsten P, Josefsson A, Thysell E, Lundholm M, Hägglöf C, Iglesias-Gato D et al (2019) Immunoreactivity for prostate specific antigen and Ki67 differentiates subgroups of prostate cancer related to outcome. Mod Pathol 32(9):1310–1319
Miller I, Min M, Yang C, Tian C, Gookin S, Carter D et al (2018) Ki67 is a graded rather than a binary marker of proliferation versus quiescence. Cell Rep 24(5):1105–1112
Funding
This work was supported by the natural science foundation of Guangxi Zhuang autonomous region (2018GXNSFBA281043), the scientific research and technology development of Guangxi Zhuang autonomous region (AD20159033), the Guangxi first-class discipline construction project of Guangxi university of Chinese medicine (2018XK062) and the development program of high-level talent team under Qihuang project of Guangxi university of Chinese medicine (Grant No.2021003).
Author information
Authors and Affiliations
Contributions
XW, GF, YP designed the study and developed the methodology. YZ, JC, WF, KL, FZ were involved in performing the experiments and collected the data. XW, YZ and JC analyzed and interpreted the data. XW, YZ, JC, XL and GF wrote the original draft. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no competing interests.
Ethical statement
Animal experiment involved in this study was approved by the institutional committee for animal welfare of the Guangxi University of Chinese Medicine.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Y., Chen, J., Fang, W. et al. Kaempferol suppresses androgen-dependent and androgen-independent prostate cancer by regulating Ki67 expression. Mol Biol Rep 49, 4607–4617 (2022). https://doi.org/10.1007/s11033-022-07307-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07307-2