Abstract
Background/aim
Long non-coding RNA TARID (lncRNA TARID) can activate the tumor suppressor TCF21 in tumorigenesis by inducing promoter demethylation. However, the impact on lncRNA TARID and its variants of coronary artery disease (CAD) are poorly understood.
Methods
We performed a case–control study enrolling 949 cases and 892 controls to assess genotype. Five variants were genotyped by TaqMan assay. 20 cases and 20 controls were used to evaluate the expression of lncRNA TARID. The cell proliferation rate was evaluated by CCK-8. The RT-qPCR and cell cycle analysis were applied to examine cell proliferation-related mRNA and cell distribution.
Results
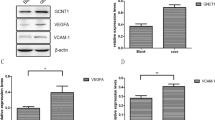
This study indicated that rs2327433 GG genotype was associated with CAD risk adjusting for traditional risk factors (OR = 2.74, 95%CI: 1.10–6.83, P = 0.03). Our results analyses revealed that the genotype of rs2327433 was related to the proportion of CAD patients with left anterior descending artery disease and left circumflex artery disease (P = 0.025 and P = 0.025, respectively). The results showed that the minor allele frequency of rs2327433 was significantly correlated with the severity of the disease (P = 0.029). The eQTL analysis showed that rs2327433 may affect the transcription factors TCF21 regulated by lncRNA TARID. We found that TARID silencing regulated cell proliferation and altered cell cycle progression by induced upregulation of CDK1 and PCNA.
Conclusions
SNP rs2327433 in lncRNA TARID was associated with CAD risk and the severity of CAD in the Chinese Han population. Furthermore, SNP rs2327433 may affect the expression of atherosclerosis-related transcription factor TCF21 regulated by lncRNA TARID. Finally, our study provided a new lncRNA-dictated regulatory mechanism participating in cell proliferation.


Similar content being viewed by others
References
Liu B, Pjanic M, Wang T et al (2018) Genetic regulatory mechanisms of smooth muscle cells map to coronary artery disease risk loci. Am J Hum Genet 103:377–388
Rizzacasa B, Amati F, Romeo F, Novelli G, Mehta JL (2019) Epigenetic modification in coronary atherosclerosis: JACC review topic of the week. J Am Coll Cardiol 74:1352–1365
Van Der Harst P, Verweij N (2018) Identification of 64 novel genetic loci provides an expanded view on the genetic architecture of coronary artery disease. Circul Res 122:433–443
Lu X, Wang L, Chen S et al (2012) Genome-wide association study in Han Chinese identifies four new susceptibility loci for coronary artery disease. Nat Genet 44:890–894
Lo Sardo V, Chubukov P, Ferguson W et al (2018) Unveiling the role of the most impactful cardiovascular risk locus through haplotype editing. Cell 175:1796–1810e20
Guo F, Tang C, Li Y, Liu Y, Lv P, Wang W, Mu Y (2018) The interplay of LncRNA ANRIL and mir-181b on the inflammation-relevant coronary artery disease through mediating NF-κb signalling pathway. J Cell Mol Med 22:5062–5075
Schunkert H, König IR, Kathiresan S et al (2011) Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat Genet 43:333–340
Boon RA, Jaé N, Holdt L, Dimmeler S (2016) Long noncoding RNAs from clinical genetics to therapeutic targets? J Am Coll Cardiol 67:1214–1226
Yang J, Gu L, Guo X, Huang J, Chen Z, Huang G, Kang Y, Zhang X, Long J, Su L (2018) LncRNA ANRIL expression and ANRIL gene polymorphisms contribute to the risk of ischemic stroke in the Chinese Han population. Cell Mol Neurobiol 38:1253–1269
Zhou L, Zhang X, He M, Cheng L, Chen Y, Hu FB, Wu T (2008) Associations between single nucleotide polymorphisms on chromosome 9p21 and risk of coronary heart disease in Chinese Han population. Arterioscler Thromb Vasc Biol 28:2085–2089
Jing J, Su L, Zeng Y, Tang X, Wei J, Wang L, Zhou L (2016) Variants in 9p21 predicts severity of coronary artery disease in a Chinese Han population. Ann Hum Genet 80:274–281
Li L, Wang L, Li H et al (2018) Characterization of LncRNA expression profile and identification of novel LncRNA biomarkers to diagnose coronary artery disease. Atherosclerosis 275:359–367
Vausort M, Wagner DR, Devaux Y (2014) Long noncoding RNAs in patients with acute myocardial infarction. Circul Res 115:668–677
Jiang C, Sun M, Li S, Tan J, Wang M, He Y (2021) Long non-coding RNA DICER1-AS1-low expression in arsenic-treated A549 cells inhibits cell proliferation by regulating the cell cycle pathway. Environ Toxicol Pharmacol 84:103617
Miller CL, Anderson DR, Kundu RK et al (2013) Disease-related growth factor and embryonic signaling pathways modulate an enhancer of TCF21 expression at the 6q23.2 coronary heart disease locus. PLoS Genet. https://doi.org/10.1371/journal.pgen.1003652
Miller CL, Haas U, Diaz R et al (2014) Coronary heart disease-associated variation in TCF21 disrupts a miR-224 binding site and miRNA-mediated regulation. PLoS Genet. https://doi.org/10.1371/journal.pgen.1004263
Arab K, Park YJ, Lindroth AM et al (2014) Long noncoding RNA TARID directs demethylation and activation of the tumor suppressor TCF21 via GADD45A. Mol Cell 55:604–614
Nagao M, Lyu Q, Zhao Q et al (2020) Coronary disease-associated gene TCF21 inhibits smooth muscle cell differentiation by blocking the myocardin-serum response factor pathway. Circul Res. https://doi.org/10.1161/CIRCRESAHA.119.315968
Pozarowski P, Darzynkiewicz Z (2004) Analysis of cell cycle by flow cytometry. Methods Mol Biol (Clifton, NJ). https://doi.org/10.1385/1-59259-811-0:301
Zhou W, Xu B (2021) SNP-mediated lncRNA-ENTPD3-AS1 upregulation suppresses renal cell carcinoma via miR-155/HIF-1 α signaling. Cell Death Dis 12:1–9
Wirka RC, Wagh D, Paik DT et al (2019) Atheroprotective roles of smooth muscle cell phenotypic modulation and the TCF21 disease gene as revealed by single-cell analysis. Nat Med. https://doi.org/10.1038/s41591-019-0512-5
Chen D, Ma W, Ke Z, Xie F (2018) CircRNA hsa_circ_100395 regulates miR-1228/TCF21 pathway to inhibit lung cancer progression. Cell Cycle 17:2080–2090
Liang J, Le TH, Edwards DRV et al (2017) Single-trait and multi-trait genome-wide association analyses identify novel loci for blood pressure in African-ancestry populations. PLoS Genet. https://doi.org/10.1371/journal.pgen.1006728
Arab K, Karaulanov E, Musheev M, Trnka P, Schäfer A, Grummt I, Niehrs C (2019) GADD45A binds R-loops and recruits TET1 to CpG island promoters. Nat Genet 51:217–223
Wang Y, Wang L, Liu X, Zhang Y, Yu L, Zhang F, Liu L, Cai J, Yang X, Wang X (2014) Genetic variants associated with myocardial infarction and the risk factors in Chinese population. PLoS ONE. https://doi.org/10.1371/journal.pone.0086332
Dichgans M, Malik R, König IR et al (2014) Shared genetic susceptibility to ischemic stroke and coronary artery disease: a genome-wide analysis of common variants. Stroke 45:24–36
Dechamethakun S, Ikeda S, Arai T, Sato N, Sawabe M, Muramatsu M (2014) Associations between the CDKN2A/B, ADTRP and PDGFD polymorphisms and the development of coronary atherosclerosis in Japanese patients. J Atheroscler Thromb 21:680–690
Poon RYC (2016) Cell cycle control: a system of interlinking oscillators. Methods Mol Biol. https://doi.org/10.1007/978-1-4939-2957-3_1
Wang Z, Wang Y, Wang S et al (2018) Coxsackievirus A6 induces cell cycle arrest in G0/G1 phase for viral production. Front Cell Infect Microbiol. https://doi.org/10.3389/fcimb.2018.00279
González-Magaña A, Blanco FJ (2020) Human PCNA structure, function and interactions. Biomolecules. https://doi.org/10.3390/biom10040570
Mansilla SF, de la Vega MB, Calzetta NL, Siri SO, Gottifredi V (2020) Cdk-independent and pcna-dependent functions of p21 in dna replication. Genes. https://doi.org/10.3390/genes11060593
Strzalka W, Ziemienowicz A (2011) Proliferating cell nuclear antigen (PCNA): a key factor in DNA replication and cell cycle regulation. Ann Botany. https://doi.org/10.1093/aob/mcq243
Bednarek K, Kostrzewska-poczekaj M, Ustaszewski A, Janiszewska J (2021) Laryngeal squamous cell carcinoma cell lines show high tolerance for siRNA-mediated CDK1 knockdown. Am J Cancer Res 11:2081–2094
Acknowledgements
The authors are particularly grateful to all volunteers for participating in the present study. This work was supported by the National Natural Science Foundation of China (Grant No. 81773519 to L.Z.) and the Intelligent Medical Project of Chongqing Medical University (Grant Nos. ZHYX202026 and YJSZHYX202018).
Author information
Authors and Affiliations
Contributions
ZC contributed to conceptualization, methodology, and writing the original article; YZ, YZ, JF, YX, ML, HC, contributed to methodology; LZ contributed to review writing and editing, supervision, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cheng, Z., Zhang, Y., Zhuo, Y. et al. LncRNA TARID induces cell proliferation through cell cycle pathway associated with coronary artery disease. Mol Biol Rep 49, 4573–4581 (2022). https://doi.org/10.1007/s11033-022-07304-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07304-5