Abstract
Background
Microvascular complications related to diabetes mellitus type II such as foot ulcers are the reason of many mortalities among T2DM patients. The role of microRNAs (miRNAs, miRs) as potent regulators of gene expression is studied in different diseases such as diabetes mellitus and primary studies revealed their importance as early detecting biomarkers. Therefore, in this study it is tried to evaluate the expression level of some miRNAs (miR-770-5p, miR-661 and miR-571) in serum and tissue samples of T2DM related foot ulcer among Iranian patients.
Methods
30 samples of blood and 30 muscle tissue were collected from T2DM patients suffering foot ulcer (T2DM + FU), 30 blood samples collected from T2DM patients without foot ulcer (T2DM-FU). 30 tissue samples collected from patients with trauma and 30 blood samples were selected as healthy controls.
Results
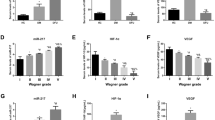
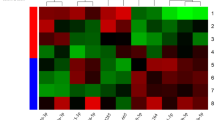
The three studied miRNAs were statistically significant in all groups in comparison to control blood group. Also, comparison between other groups showed a significant increase of all studied miRNAs especially in the blood and tissues of T2DM + FU patients. The only significant correlation detected between the FBS level and miR-571 expression pattern in blood samples of T2DM + FU group. Finally, the results showed that miR-571, -661, and -770 has a statistically significant discriminative character for differentiating T2DM + FU patients from T2DM-FU both in tissue and blood samples.
Conclusion
Although more studies are essential for certifying these findings, our results showed that miR-770-5p, miR-661 and miR-571 are correlated with the microvascular complications related with T2DM such as foot ulcer.




Similar content being viewed by others
Data availability
Upon reasonable request authors will send relevant documentation or data.
Code availability
Not applicable.
References
Martin A, Komada MR, Sane DC (2003) Abnormal angiogenesis in diabetes mellitus [Internet]. Medicinal Research Reviews. Med Res Rev. https://doi.org/10.1002/med.10024
Boulton AJM (2013). The pathway to foot ulceration in diabetes [Internet]. Medical Clinics of North America. Med Clin North Am. https://doi.org/10.1016/j.mcna.2013.03.007
Moura LIF, Dias AMA, Carvalho E, De Sousa HC (2013) Recent advances on the development of wound dressings for diabetic foot ulcer treatment - a review [Internet]. Acta Biomaterialia. Acta Biomater. https://doi.org/10.1016/j.actbio.2013.03.033
Da Silva L, Carvalho E, Cruz MT (2010) Role of neuropeptides in skin inflammation and its involvement in diabetic wound healing [Internet]. Expert Opinion on Biological Therapy. Expert Opin Biol Ther. https://doi.org/10.1517/14712598.2010.515207
Iversen MM, Tell GS, Riise T, Hanestad BR, Østbye T, Graue M et al (2009) History of foot ulcer increases mortality among individuals with diabetes: ten-year follow-up of the Nord-Trøndelag health study. Norway Diabetes Care Diabetes Care 32:2193–2199. https://doi.org/10.2337/dc09-0651
Sanjeev K Gupta SKS. Diabetic foot: a continuing challenge - PubMed [Internet]. [cited 26 May 2021]. Available: https://pubmed.ncbi.nlm.nih.gov/23393676/
Wright E, Scism-Bacon JL, Glass LC (2006) Oxidative stress in type 2 diabetes: The role of fasting and postprandial glycaemia [Internet]. International Journal of Clinical Practice. Int J Clin Pract. https://doi.org/10.1111/j.1368-5031.2006.00825.x
Daiber A, Oelze M, Schuhmacher S (2010) Organic nitrates and nitrate resistance in diabetes: The role of vascular dysfunction and oxidative stress with emphasis on antioxidant properties of pentaerithrityl tetranitrate. Exp Diabetes Res. Exp Diabetes Res. https://doi.org/10.1155/2010/213176
Wang C, Wan S, Yang T, Niu D, Zhang A, Yang C et al (2016) Increased serum microRNAs are closely associated with the presence of microvascular complications in type 2 diabetes mellitus. Sci Rep Nature Publishing Group 6:1–9. https://doi.org/10.1038/srep20032
Flynt AS, Lai EC (2008) Biological principles of microRNA-mediated regulation: Shared themes amid diversity [Internet]. Nature Reviews Genetics. Nat Rev Genet. https://doi.org/10.1038/nrg2455
Tang X, Tang G, Özcan S (2008) Role of microRNAs in diabetes [Internet]. Biochimica et Biophysica Acta - Gene Regulatory Mechanisms. Biochim Biophys Acta. https://doi.org/10.1016/j.bbagrm.2008.06.010
Kantharidis P, Wang B, Carew RM, Lan HY (2011) Diabetes complications: The microRNA perspective [Internet]. Diabetes. Diabetes. https://doi.org/10.2337/db11-0082
Banerjee J, Chan YC, Sen CK (2011) MicroRNAs in skin and wound healing [Internet]. Physiological Genomics. Physiol Genomics. https://doi.org/10.1152/physiolgenomics.00157.2010
Fahs F, Bi X-L, Yu F-S, Zhou L, Mi Q-S (2016) Small RNAs Play Big Roles: MicroRNAs in Diabetic Wound Healing. Curr Mol Med 16:545–552. https://doi.org/10.2174/1566524016666160607091156
Schulte C, Zeller T (2015) microRNA-based diagnostics and therapy in cardiovascular disease-Summing up the facts. Cardiovasc Diagn Ther Cardiovasc Diagn Ther 5:17–36. https://doi.org/10.3978/j.issn.2223-3652.2014.12.03
Zampetaki A, Kiechl S, Drozdov I, Willeit P, Mayr U, Prokopi M et al (2010) Plasma MicroRNA profiling reveals loss of endothelial MiR-126 and other MicroRNAs in type 2 diabetes. Circ Res Circ Res 107:810–817. https://doi.org/10.1161/CIRCRESAHA.110.226357
Alhatemi AK, Majeed SR, Mubarak SMH (2021) Serum and tissue expression levels of microRNAs-661, -571 and -770–5p among diabetic foot ulcer patients compared to healthy controls. Gene Reports. Elsevier Inc. https://doi.org/10.1016/j.genrep.2020.100946
Brem H, Tomic-Canic M (2007) Cellular and molecular basis of wound healing in diabetes [Internet]. Journal of Clinical Investigation. J Clin Invest. https://doi.org/10.1172/JCI32169
Baltzis D, Eleftheriadou I, Veves A (2014) Pathogenesis and treatment of impaired wound healing in diabetes mellitus: new insights [Internet]. Adv Therapy. Springer Healthcare. https://doi.org/10.1007/s12325-014-0140-x
Moura J, Børsheim E, Carvalho E (2014) The role of micrornas in diabetic complications—special emphasis on wound healing [Internet]. Genes. MDPI AG. https://doi.org/10.3390/genes5040926
Zhi YF, Prins JB, Marwick TH (2004) Diabetic cardiomyopathy: Evidence, mechanisms, and therapeutic implications [Internet]. Endocrine Rev. Endocr Rev. https://doi.org/10.1210/er.2003-0012
Broughton G, Janis JE, Attinger CE (2006) The basic science of wound healing [Internet]. Plastic and Reconstructive Surgery. Plast Reconstr Surg. https://doi.org/10.1097/01.prs.0000225430.42531.c2
Pastar I, Stojadinovic O, Yin NC, Ramirez H, Nusbaum AG, Sawaya A, et al (2014) Epithelialization in wound healing: a comprehensive review. Adv Wound Care. Mary Ann Liebert Inc. https://doi.org/10.1089/wound.2013.0473
Eming SA, Martin P, Tomic-Canic M (2014) Wound repair and regeneration: Mechanisms, signaling, and translation [Internet]. Science Translational Medicine. American Association for the Advancement of Science. https://doi.org/10.1126/scitranslmed.3009337
Adeghate E (2004) Molecular and cellular basis of the aetiology and management of diabetic cardiomyopathy: a short review [Internet]. Molecular and Cellular Biochemistry. Mol Cell Biochem. https://doi.org/10.1023/B:MCBI.0000028755.86521.11
Ortega FJ, Mercader JM, Moreno-Navarrete JM, Rovira O, Guerra E, Esteve E, et al (2014) Profiling of circulating microRNAs reveals common microRNAs linked to type 2 diabetes that change with insulin sensitization. Diabetes Care. American Diabetes Association Inc. https://doi.org/10.2337/dc13-1847
Takahashi P, Xavier DJ, Evangelista AF, Manoel-Caetano FS, Macedo C, Collares CVA et al (2014) MicroRNA expression profiling and functional annotation analysis of their targets in patients with type 1 diabetes mellitus. Gene Gene 539:213–223. https://doi.org/10.1016/j.gene.2014.01.075
Salas-Pérez F, Codner E, Valencia E, Pizarro C, Carrasco E, Pérez-Bravo F (2013) MicroRNAs miR-21a and miR-93 are down regulated in peripheral blood mononuclear cells (PBMCs) from patients with type 1 diabetes. Immunobiology Immunobiology 218:733–737. https://doi.org/10.1016/j.imbio.2012.08.276
Baran-Gale J, Fannin EE, Kurtz CL, Sethupathy P (2013) Beta Cell 5′-Shifted isomiRs Are Candidate Regulatory Hubs in Type 2 Diabetes. PLoS One. PLoS One. https://doi.org/10.1371/journal.pone.0073240
Schulze MB, Weikert C, Pischon T, Bergmann MM, Al-Hasani H, Schleicher E et al (2009) Use of multiple metabolic and genetic markers to improve the prediction of type 2 diabetes: The EPIC-Potsdam study. Diabetes Care 32:2116–2119. https://doi.org/10.2337/dc09-0197
Zhang T, Li L, Shang Q, Lv CF, Wang CY, Su B (2015) Circulating miR-126 is a potential biomarker to predict the onset of type 2 diabetes mellitus in susceptible individuals. Biochem Biophys Res Commun. Academic Press Inc. https://doi.org/10.1016/j.bbrc.2015.05.017
Chen X, Ba Y, Ma L, Cai X, Yin Y, Wang K et al (2008) Characterization of microRNAs in serum: A novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res Cell Res 18:997–1006. https://doi.org/10.1038/cr.2008.282
Spinetti G, Fortunato O, Caporali A, Shantikumar S, Marchetti M, Meloni M et al (2013) MicroRNA-15a and MicroRNA-16 impair human circulating proangiogenic cell functions and are increased in the proangiogenic cells and serum of patients with critical limb ischemia. Circ Res Circ Res 112:335–346. https://doi.org/10.1161/CIRCRESAHA.111.300418
Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S, et al (2003) Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 114: 763–76. Available: http://www.ncbi.nlm.nih.gov/pubmed/14505575
Roderburg C, Mollnow T, Bongaerts B, Elfimova N, Cardenas DV, Berger K, et al (2012) Micro-RNA profiling in human serum reveals compartment-specific roles of miR-571 and miR-652 in liver cirrhosis. PLoS One. PLoS One. https://doi.org/10.1371/journal.pone.0032999
Vetter G, Saumet A, Moes M, Vallar L, Le Béchec A, Laurini C et al (2010) MiR-661 expression in SNAI1-induced epithelial to mesenchymal transition contributes to breast cancer cell invasion by targeting Nectin-1 and StarD10 messengers. Oncogene Oncogene 29:4436–4448. https://doi.org/10.1038/onc.2010.181
Liu X, Wu Y, Huang Q, Zou D, Qin W, Chen Z (2015) Grouping pentylenetetrazol-induced epileptic rats according to memory impairment and MicroRNA expression profiles in the hippocampus. PLoS One. Public Library of Science. https://doi.org/10.1371/journal.pone.0126123
FQ F, GS L, JC L, LZ C (2019) Overexpression of miR-770 indicates a favorable prognosis and suppresses tumorigenesis by modulating PI3K-AKT pathway in glioma. Eur Rev Med Pharmacol Sci. Eur Rev Med Pharmacol Sci. https://doi.org/10.26355/EURREV_201904_17565
Liu J, Luo R, Wang J, Luan X, Wu D, Chen H, et al (2012) Tumor Cell-Derived Exosomal miR-770 Inhibits M2 Macrophage Polarization via Targeting MAP3K1 to Inhibit the Invasion of Non-small Cell Lung Cancer Cells. Front Cell Dev Biol. Frontiers. https://doi.org/10.3389/FCELL.2021.679658
Zhang SZ, Qiu XJ, Dong SS, Zhou LN, Zhu Y, Wang MD, et al (2019) MicroRNA-770–5p is involved in the development of diabetic nephropathy through regulating podocyte apoptosis by targeting TP53 regulated inhibitor of apoptosis 1. Eur Rev Med Pharmacol Sci. Verduci Editore s.r.l. https://doi.org/10.26355/eurrev_201902_17018
Zhang Y, Sun X, Icli B, Feinberg MW (2017) Emerging roles for microRNAs in diabetic microvascular disease: Novel targets for therapy [Internet]. Endocrine Reviews. Endocrine Society. https://doi.org/10.1210/er.2016-1122
Acknowledgements
The authors wish to thank Shiraz University of Medical Sciences for the supporting
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Animal rights
Written consents were taken from all participants.
Ethical approval
The current study was approved by the Ethical committee of Shiraz University of Medical Sciences and also all the used protocols were in conformation with the ethical guidelines of Helsinki Declaration.
Consent to participate
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Rashin VatanIman, Seyed Hossein Malekpour, Afsoon Afshari and Maryam Zare. The first draft of the manuscript was written by Afsoon Afshari and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
VatanIman, R., Malekpour, S.H., Afshari, A. et al. MiR-770-5p, miR-661 and miR-571 expression level in serum and tissue samples of foot ulcer caused by diabetes mellitus type II in Iranian population. Mol Biol Rep 48, 7811–7818 (2021). https://doi.org/10.1007/s11033-021-06798-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06798-9