Abstract
Background
Chondrocyte proliferation and differentiation play pivotal roles in regulating cartilage formation, endochondral bone formation, and repair. Cartilage damage and underdevelopment may cause severe joint diseases. Various transcription factors regulate cartilage development. Nuclear factor 1 B (Nfib) is a transcription factor that plays a regulatory role in various organs. However, the effect and mechanism of Nfib on the proliferation and differentiation of chondrocytes in cartilage are still largely unknown.
Methods and results
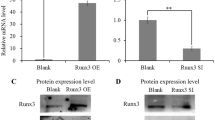
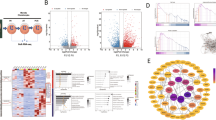
In the present study, we investigated the gene expression patterns in primary chondrocytes with Nfib overexpression or silencing by RNA sequencing (RNA-seq) technology. The results showed that Nfib overexpression significantly up-regulated genes that are related to chondrocyte proliferation and extracellular matrix (ECM) synthesis and significantly down-regulated genes related to chondrocyte differentiation and ECM degradation. However, with Nfib silencing, the genes involved in promoting chondrocyte differentiation were significantly up-regulated, whereas those involved in promoting chondrocyte proliferation were significantly down-regulated. Furthermore, quantitative real-time PCR (qRT-PCR), western blot, alcian blue staining and immunofluorescence staining assays further confirmed that Nfib potentially promotes chondrocyte proliferation and extracellular synthesis but inhibits differentiation.
Conclusions
The molecular mechanism of Nfib in promoting chondrocyte proliferation and inhibiting differentiation was probably achieved by stimulating Sox9 and its downstream genes. Thus, this study adds new insights regarding the underlying molecular mechanism of transcriptional regulation in cartilage.




Similar content being viewed by others
Data availability
The RNA-seq data set are available in the NCBI Sequence Read Archive (SRA) database (Accession Number: PRJNA707136).
References
Oh CD, Lu Y, Liang S, Mori-Akiyama Y, Chen D, de Crombrugghe B, Yasuda H (2014) SOX9 regulates multiple genes in chondrocytes, including genes encoding ECM proteins, ECM modification enzymes, receptors, and transporters. PLoS ONE 9:e107577. https://doi.org/10.1371/journal.pone.0107577
Akiyama H, Chaboissier MC, Martin JF, Schedl A, de Crombrugghe B (2002) The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev 16:2813–2828. https://doi.org/10.1101/gad.1017802
Wuelling M, Vortkamp A (2011) Chondrocyte proliferation and differentiation. Endocr Dev 21:1–11. https://doi.org/10.1159/000328081
Kim IS, Otto F, Zabel B, Mundlos S (1999) Regulation of chondrocyte differentiation by Cbfa1. Mech Dev 80:159–170. https://doi.org/10.1016/s0925-4773(98)00210-x
Inada M, Yasui T, Nomura S, Miyake S, Deguchi K, Himeno M, Sato M, Yamagiwa H, Kimura T, Yasui N, Ochi T, Endo N, Kitamura Y, Kishimoto T, Komori T (1999) Maturational disturbance of chondrocytes in Cbfa1-deficient mice. Dev Dyn 214:279–290. https://doi.org/10.1002/(SICI)1097-0177(199904)214:4%3c279::AID-AJA1%3e3.0.CO;2-W
Cheng A, Genever PG (2010) SOX9 determines RUNX2 transactivity by directing intracellular degradation. J Bone Miner Res 25:2680–2689. https://doi.org/10.1002/jbmr.174
Komori T (2018) Runx2, an inducer of osteoblast and chondrocyte differentiation. Histochem Cell Biol 149:313–323. https://doi.org/10.1007/s00418-018-1640-6
Chen H, Ghori-Javed FY, Rashid H, Adhami MD, Serra R, Gutierrez SE, Javed A (2014) Runx2 regulates endochondral ossification through control of chondrocyte proliferation and differentiation. J Bone Miner Res 29:2653–2665. https://doi.org/10.1002/jbmr.2287
Zhou Z, Yao B, Zhao D (2020) Runx3 regulates chondrocyte phenotype by controlling multiple genes involved in chondrocyte proliferation and differentiation. Mol Biol Rep 47:5773–5792. https://doi.org/10.1007/s11033-020-05646-6
Bi W, Deng JM, Zhang Z, Behringer RR, de Crombrugghe B (1999) Sox9 is required for cartilage formation. Nat Genet 22:85–89. https://doi.org/10.1038/8792
Ikegami D, Akiyama H, Suzuki A, Nakamura T, Nakano T, Yoshikawa H, Tsumaki N (2011) Sox9 sustains chondrocyte survival and hypertrophy in part through Pik3ca-Akt pathways. Development 138:1507–1519. https://doi.org/10.1242/dev.057802
Uchihashi T, Kimata M, Tachikawa K, Koshimizu T, Okada T, Ihara-Watanabe M, Sakai N, Kogo M, Ozono K, Michigami T (2007) Involvement of nuclear factor I transcription/replication factor in the early stage of chondrocytic differentiation. Bone 41:1025–1035. https://doi.org/10.1016/j.bone.2007.08.028
Hsu YC, Osinski J, Campbell CE, Litwack ED, Wang D, Liu S, Bachurski CJ, Gronostajski RM (2011) Mesenchymal nuclear factor I B regulates cell proliferation and epithelial differentiation during lung maturation. Dev Biol 354:242–252. https://doi.org/10.1016/j.ydbio.2011.04.002
Becker-Santos DD, Lonergan KM, Gronostajski RM, Lam WL (2017) Nuclear factor I/B: a master regulator of cell differentiation with paradoxical roles in cancer. EBioMedicine 22:2–9. https://doi.org/10.1016/j.ebiom.2017.05.027
Gründer A, Ebel TT, Mallo M, Schwarzkopf G, Shimizu T, Sippel AE, Schrewe H (2002) Nuclear factor I-B (Nfib) deficient mice have severe lung hypoplasia. Mech Dev 112:69–77. https://doi.org/10.1016/s0925-4773(01)00640-2
Steele-Perkins G, Plachez C, Butz KG, Yang G, Bachurski CJ, Kinsman SL, Litwack ED, Richards LJ, Gronostajski RM (2005) The transcription factor gene Nfib is essential for both lung maturation and brain development. Mol Cell Biol 25:685–698. https://doi.org/10.1128/MCB.25.2.685-698.2005
Rolando C, Erni A, Grison A, Beattie R, Engler A, Gokhale PJ, Milo M, Wegleiter T, Jessberger S, Taylor V (2016) Multipotency of adult hippocampal NSCs in vivo is restricted by drosha/NFIB. Cell Stem Cell 19:653–662. https://doi.org/10.1016/j.stem.2016.07.003
Mellas RE, Kim H, Osinski J, Sadibasic S, Gronostajski RM, Cho M, Baker OJ (2015) NFIB regulates embryonic development of submandibular glands. J Dent Res 94:312–319. https://doi.org/10.1177/0022034514559129
Chang CY, Pasolli HA, Giannopoulou EG, Guasch G, Gronostajski RM, Elemento O, Fuchs E (2013) NFIB is a governor of epithelial-melanocyte stem cell behaviour in a shared niche. Nature 495:98–102. https://doi.org/10.1038/nature11847
Chen L, Kostadima M, Martens JHA, Canu G, Garcia SP, Turro E, Downes K, Macaulay IC, Bielczyk-Maczynska E, Coe S, Farrow S, Poudel P, Burden F, Jansen SBG, Astle WJ, Attwood A, Bariana T, de Bono B, Breschi A, Chambers JC, Consortium B, Choudry FA, Clarke L, Coupland P, van der Ent M, Erber WN, Jansen JH, Favier R, Fenech ME, Foad N, Freson K, van Geet C, Gomez K, Guigo R, Hampshire D, Kelly AM, Kerstens HHD, Kooner JS, Laffan M, Lentaigne C, Labalette C, Martin T, Meacham S, Mumford A, Nürnberg S, Palumbo E, van der Reijden BA, Richardson D, Sammut SJ, Slodkowicz G, Tamuri AU, Vasquez L, Voss K, Watt S, Westbury S, Flicek P, Loos R, Goldman N, Bertone P, Read RJ, Richardson S, Cvejic A, Soranzo N, Ouwehand WH, Stunnenberg HG, Frontini M, Rendon A (2014) Transcriptional diversity during lineage commitment of human blood progenitors. Science 345:1251033. https://doi.org/10.1126/science.1251033
Waki H, Nakamura M, Yamauchi T, Wakabayashi K, Yu J, Hirose-Yotsuya L, Take K, Sun W, Iwabu M, Okada-Iwabu M, Fujita T, Aoyama T, Tsutsumi S, Ueki K, Kodama T, Sakai J, Aburatani H, Kadowaki T (2011) Global mapping of cell type-specific open chromatin by FAIRE-seq reveals the regulatory role of the NFI family in adipocyte differentiation. PLoS Genet 7:e1002311. https://doi.org/10.1371/journal.pgen.1002311
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12:357–360. https://doi.org/10.1038/nmeth.3317
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515. https://doi.org/10.1038/nbt.1621
Wang L, Feng Z, Wang X, Wang X, Zhang X (2010) DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26:136–138. https://doi.org/10.1093/bioinformatics/btp612
Zhao RL, Zhang XM, Jia LN, Song W, Sun YL, Meng XY, Peng XX (2019) pNNS-Conjugated chitosan mediated IGF-1 and miR-140 overexpression in articular chondrocytes improves cartilage repair. Biomed Res Int 2019:2761241. https://doi.org/10.1155/2019/2761241
Surmann-Schmitt C, Dietz U, Kireva T, Adam N, Park J, Tagariello A, Onnerfjord P, Heinegård D, Schlötzer-Schrehardt U, Deutzmann R, von der Mark K, Stock M (2008) Ucma, a novel secreted cartilage-specific protein with implications in osteogenesis. J Biol Chem 283:7082–7093. https://doi.org/10.1074/jbc.M702792200
Seuffert F, Weidner D, Baum W, Schett G, Stock M (2018) Upper zone of growth plate and cartilage matrix associated protein protects cartilage during inflammatory arthritis. Arthritis Res Ther 20:88–100. https://doi.org/10.1186/s13075-018-1583-2
Poulet B, Liu K, Plumb D, Vo P, Shah M, Staines K, Sampson A, Nakamura H, Nagase H, Carriero A, Shefelbine S, Pitsillides AA, Bou-Gharios G (2016) Overexpression of TIMP-3 in chondrocytes produces transient reduction in growth plate length but permanently reduces adult bone quality and quantity. PLoS ONE 11:e0167971. https://doi.org/10.1371/journal.pone.0167971
Prideaux M, Staines KA, Jones ER, Riley GP, Pitsillides AA, Farquharson C (2015) MMP and TIMP temporal gene expression during osteocytogenesis. Gene Expr Patterns 18:29–36. https://doi.org/10.1016/j.gep.2015.04.004
Deckx S, Heymans S, Papageorgiou AP (2016) The diverse functions of osteoglycin: a deceitful dwarf, or a master regulator of disease? FASEB J 30:2651–2661. https://doi.org/10.1096/fj.201500096R
Wang A, Hu N, Zhang Y, Chen Y, Su C, Lv Y, Shen Y (2019) MEG3 promotes proliferation and inhibits apoptosis in osteoarthritis chondrocytes by miR-361-5p/FOXO1 axis. BMC Med Genomics 12:201–211. https://doi.org/10.1186/s12920-019-0649-6
Muttigi MS, Han I, Park HK, Park H, Lee SH (2016) Matrilin-3 role in cartilage development and osteoarthritis. Int J Mol Sci 17:590–599. https://doi.org/10.3390/ijms17040590
Huang Y, Wan G, Tao J (2017) C1q/TNF-related protein-3 exerts the chondroprotective effects in IL-1β-treated SW1353 cells by regulating the FGFR1 signaling. Biomed Pharmacother 85:41–46. https://doi.org/10.1016/j.biopha.2016.11.128
Tashima T, Nagatoishi S, Sagara H, Ohnuma S, Tsumoto K (2015) Osteomodulin regulates diameter and alters shape of collagen fibrils. Biochem Biophys Res Commun 463:292–296. https://doi.org/10.1016/j.bbrc.2015.05.053
Thaler R, Sturmlechner I, Spitzer S, Riester SM, Rumpler M, Zwerina J, Klaushofer K, van Wijnen AJ, Varga F (2015) Acute-phase protein serum amyloid A3 is a novel paracrine coupling factor that controls bone homeostasis. FASEB J 29:1344–1359. https://doi.org/10.1096/fj.14-265512
Rose BJ, Kooyman DL (2016) A tale of two joints: the role of matrix metalloproteases in cartilage biology. Dis Markers 2016:4895050. https://doi.org/10.1155/2016/4895050
Liao W, Li Z, Zhang H, Li J, Wang K, Yang Y (2015) Proteomic analysis of synovial fluid as an analytical tool to detect candidate biomarkers for knee osteoarthritis. Int J Clin Exp Pathol 8:9975–9989
Willcockson H, Ozkan H, Chubinskaya S, Loeser RF, Longobardi L (2021) CCL2 induces articular chondrocyte MMP expression through ERK and p38 signaling pathways. Osteoarthr Cartil Open 3:100136. https://doi.org/10.1016/j.ocarto.2020.100136
Ohlsson S, Tufvesson B, Polling A, Ohlsson K (1997) Distribution of the secretory leucocyte proteinase inhibitor in human articular cartilage. Biol Chem 378:1055–1058. https://doi.org/10.1515/bchm.1997.378.9.1055
Kim HE, Shin Y, Jung IJ, Yang JI, Chun CH, Kim HA, Chun JS (2021) Overexpression of secretory leukocyte peptidase inhibitor (SLPI) does not modulate experimental osteoarthritis but may be a biomarker for the disease. Osteoarthritis Cartilage 29:558–567. https://doi.org/10.1016/j.joca.2021.01.003
Yamamoto K, Okano H, Miyagawa W, Visse R, Shitomi Y, Santamaria S, Dudhia J, Troeberg L, Strickland DK, Hirohata S, Nagase H (2016) MMP-13 is constitutively produced in human chondrocytes and co-endocytosed with ADAMTS-5 and TIMP-3 by the endocytic receptor LRP1. Matrix Biol 56:57–73. https://doi.org/10.1016/j.matbio.2016.03.007
Ushijima T, Okazaki K, Tsushima H, Ishihara K, Doi T, Iwamoto Y (2014) CCAAT/enhancer binding protein β regulates expression of Indian hedgehog during chondrocytes differentiation. PLoS ONE 9:e104547. https://doi.org/10.1371/journal.pone.0104547
Kim DH, Lee DH, Jo MR, Son DJ, Park MH, Hwang CJ, Park JH, Yuk DY, Yoon DY, Jung YS, Kim Y, Jeong JH, Han SB, Hong JT (2015) Exacerbation of collagen antibody-induced arthritis in transgenic mice overexpressing peroxiredoxin 6. Arthritis Rheumatol 67:3058–3069. https://doi.org/10.1002/art.39284
Pemmari A, Leppänen T, Hämäläinen M, Moilanen T, Vuolteenaho K, Moilanen E (2020) Widespread regulation of gene expression by glucocorticoids in chondrocytes from patients with osteoarthritis as determined by RNA-Seq. Arthritis Res Ther 22:271–284. https://doi.org/10.1186/s13075-020-02289-7
Ahmad R, Qureshi HY, El Mabrouk M, Sylvester J, Ahmad M, Zafarullah M (2007) Inhibition of interleukin 1-induced matrix metalloproteinase 13 expression in human chondrocytes by interferon gamma. Ann Rheum Dis 66:782–789. https://doi.org/10.1136/ard.2006.060269
Yamashita M, Inoue K, Saeki N, Ideta-Otsuka M, Yanagihara Y, Sawada Y, Sakakibara I, Lee J, Ichikawa K, Kamei Y, Iimura T, Igarashi K, Takada Y, Imai Y (2018) Uhrf1 is indispensable for normal limb growth by regulating chondrocyte differentiation through specific gene expression. Development 145:dev157412. https://doi.org/10.1242/dev.157412
Durand AL, Dufour A, Aubert-Foucher E, Oger-Desfeux C, Pasdeloup M, Lustig S, Servien E, Vaz G, Perrier-Groult E, Mallein-Gerin F, Lafont JE (2020) The lysine specific demethylase-1 negatively regulates the COL9A1 gene in human articular chondrocytes. Int J Mol Sci 21:6322–6337. https://doi.org/10.3390/ijms21176322
Klinger P, Surmann-Schmitt C, Brem M, Swoboda B, Distler JH, Carl HD, von der Mark K, Hennig FF, Gelse K (2011) Chondromodulin 1 stabilizes the chondrocyte phenotype and inhibits endochondral ossification of porcine cartilage repair tissue. Arthritis Rheum 63:2721–2731. https://doi.org/10.1002/art.30335
Kimura T, Okada A, Yatabe T, Okubo M, Toyama Y, Noda M, Okada Y (2010) RECK is up-regulated and involved in chondrocyte cloning in human osteoarthritic cartilage. Am J Pathol 176:2858–2867. https://doi.org/10.2353/ajpath.2010.091003
Acknowledgements
This work was supported by the National Key Research and Development Program of China (grant number 2018YFC1706605) and the National Natural Science Foundation of China (grant number 81702136).
Funding
This work was supported by the National Key Research and Development Program of China (Grant Number 2018YFC1706605), Innovation Team and Talents Cultivation Program of National Administration of Traditional Chinese Medicine (Grant Number ZYYCXTD-D-202001ZYYCXTD-D-202001) and the National Natural Science Foundation of China (Grant Number 81702136).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by DP and BQ. The first draft of the manuscript was written by DP. The review and editing of the manuscript was written by BY. The resources and supervision were performed by DZ. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures were performed in accordance with the guidelines of the Institutional Animal Ethics Committee of Changchun University of Chinese Medicine (No. ccucm-2017–0015).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pan, D., Qian, B., Zhao, D. et al. Nfib promotes chondrocyte proliferation and inhibits differentiation by mildly regulating Sox9 and its downstream genes. Mol Biol Rep 48, 7487–7497 (2021). https://doi.org/10.1007/s11033-021-06767-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06767-2