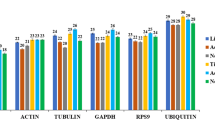
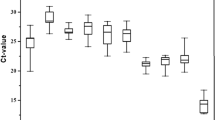
Abstract
Quantitative real time PCR (qRT-PCR) is a nucleic acid quantitative technique and is also considered as a validation tool. The Cry1Ia36 protein isolated from Bacillus thuringiensis (Bt) strain YC-10 has high nematicidal activity against nematodes. Caenorhabditis elegans is one of the major model organisms and a readily accessible source of biological material for gene expression studies. To evaluate the expression stability of 12 candidate reference genes of C. elegans for exposing to different concentrations of Cry1Ia36 protein and different treat time, five statistical approaches (the comparative delta-Ct method, BestKeeper, NormFinder, Genorm and RefFinder) were used to evaluate each individual candidate reference gene. The results indicated that cdc-42 and F35G12.2 were the best reference genes for performing reliable gene expression normalization in the impact of Cry1Ia36 protein. In addition, when C. elegans was exposed to Cry1Ia36 protein and other nematicides, avermectin and 5-aminolevulinic acid, cdc-42 was recommended as the most reliable reference genes. Y45F10D.4 was the least stable reference genes in our experimental settings. Therefore, cdc-42 was reliable reference gene for gene expression studies in C. elegans exposed to Cry1Ia36 protein and other nematicides.


Similar content being viewed by others
References
Rosa FE, Silveira SM, Silveira CG et al (2009) Quantitative real-time RT-PCR and chromogenic in situ hybridization: precise methods to detect HER-2 status in breast carcinoma. BMC Cancer 9:90. https://doi.org/10.1186/1471-2407-9-90
Hoogewijs D, Houthoofd K, Matthijssens F et al (2008) Selection and validation of a set of reliable reference genes for quantitative sod gene expression analysis in C. elegans. BMC Mol Biol 9(1):9. https://doi.org/10.1186/1471-2199-9-9
Taki FA, Zhang B (2013) Determination of reliable reference genes for multi-generational gene expression analysis on C. elegans exposed to abused drug nicotine. Psychopharmacology 230(1):77–88. https://doi.org/10.1007/s00213-013-3139-0
Wu H, Taki FA, Zhang Y et al (2014) Evaluation and identification of reliable reference genes for toxicological study in Caenorhabditis elegans. Mol Biol Rep 41(5):3445–3455. https://doi.org/10.1007/s11033-014-3206-6
Bravo A, Likitvivatanavong S, Gill SS et al (2011) Bacillus thuringiensis: a story of a successful bioinsecticide. Insect Biochem Mol Biol 41(7):423–431. https://doi.org/10.1016/j.ibmb.2011.02.006
Mohammed SH, El Saedy MA, Enan MR et al (2008) Biocontrol efficiency of Bacillus thuringiensis toxins against root-knot nematode, Meloidogyne incognita. J Cell Mol Biol 7(1):57–66
Read TD, Peterson SN, Tourasse N et al (2003) The genome sequence of Bacillus anthracis Ames and comparison to closely related bacteria. Nature 423(6935):81–86. https://doi.org/10.1038/nature01586
van Frankenhuyzen K (2009) Insecticidal activity of Bacillus thuringiensis crystal proteins. J Invertebr Pathol 101(1):1–16. https://doi.org/10.1016/j.jip.2009.02.009
Schnepf E, Nv Crickmore, Van Rie J et al (1998) Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol Mol Biol Rev 62(3):775–806. https://doi.org/10.0000/PMID9729609
Cheng F, Wang J, Song Z et al (2015) Complete genome sequence of Bacillus thuringiensis YC-10, a novel active strain against plant-parasitic nematodes. J Biotechnol 210:17–18. https://doi.org/10.1016/j.jbiotec.2015.06.395
Liu L, Knauth S, Eickhorst T (2018) Adsorption and desorption of Cry1Ab proteins on differently textured paddy soils. Pedosphere 28(1):94–102. https://doi.org/10.1016/S1002-0160(18)60006-2
Sajid M, Geng C, Li M, Wang Y et al (2018) Whole genomic analysis of Bacillus thuringiensis revealing partial genes as a source of novel Cry toxins. Appl Environ Microbiol 84(14):e00277-18. https://doi.org/10.1128/AEM.00277-18
Sanahuja G, Banakar R, Twyman RM et al (2011) Bacillus thuringiensis: a century of research, development and commercial applications. Plant Biotechnol J 9(3):283–300. https://doi.org/10.1111/j.1467-7652.2011.00595.x
Shi C, Zhou G, Liu Z et al (2018) A novel Bacillus thuringiensis Cry57 protein domain swap influence on insecticidal activity. J Northeast Agric Univ 25(3):44–52
Song Z (2017) Identification of several key crop pathogenic nematodes in hunan province and nematicidal role of Bt Cry1Ia36 protein. Hunan Agricultural University, Changsha
Brenner S (1974) The genetics of Caenorhabditis elegans. Genetics 77:71–94
Bone LW, Bottjer KP, Gill SS (1985) Trichostrongylus colubriformis: egg lethality due to Bacillus thuringiensis crystal toxin. Exp Parasitol 60(3):314. https://doi.org/10.1016/0014-4894(85)90037-2
Griffitts JS, Huffman DL, Whitacre JL et al (2003) Resistance to a bacterial toxin is mediated by removal of a conserved glycosylation pathway required for toxin-host interactions. J Biol Chem 278(46):45594–45602. https://doi.org/10.1074/jbc.M308142200
Marroquin LD, Elyassnia D, Griffitts JS et al (2000) Bacillus thuringiensis (Bt) toxin susceptibility and isolation of resistance mutants in the nematode Caenorhabditis elegans. Genetics 155(4):1693–1699. https://doi.org/10.1017/S0016672300004560
Wei JZ, Hale K, Carta L et al (2003) Bacillus thuringiensis crystal proteins that target nematodes. Proc Natl Acad Sci USA 100(5):2760–2765. https://doi.org/10.1073/pnas.0538072100
Kagias K, Podolska A, Pocock R (2014) reliable reference miRNAs for quantitative gene expression analysis of stress responses in Caenorhabditis elegans. BMC Genom 15(1):222. https://doi.org/10.1186/1471-2164-15-222
Peres TV, Arantes LP, Miah MR et al (2018) Role of Caenorhabditis elegans AKT-1/2 and SGK-1 in manganese toxicity. Neurotox Res 34(3):584–596. https://doi.org/10.1007/s12640-018-9915-1
Prasansuklab A, Meemon K, Sobhon P et al (2017) Ethanolic extract of Streblus asper leaves protects against glutamate-induced toxicity in HT22 hippocampal neuronal cells and extends lifespan of Caenorhabditis elegans. BMC Complement Altern Med 17(1):551. https://doi.org/10.1186/s12906-017-2050-3
Polak N, Read DS, Jurkschat K et al (2014) Metalloproteins and phytochelatin synthase may confer protection against zinc oxide nanoparticle induced toxicity in Caenorhabditis elegans. Comp Biochem Physiol C 160:75–85. https://doi.org/10.1016/j.cbpc.2013.12.001
Gao X, Teng Y, Luo J et al (2014) The survival motor neuron gene smn-1 interacts with the U2AF large subunit gene uaf-1 to regulate Caenorhabditis elegans lifespan and motor functions. RNA Biol 11(9):1148–1160. https://doi.org/10.4161/rna.36100
Lapierre LR, De Magalhaes Filho CD, McQuary PR et al (2013) The TFEB orthologue HLH-30 regulates autophagy and modulates longevity in Caenorhabditis elegans. Nat Commun 4:2267. https://doi.org/10.1038/ncomms3267
Zhang Y, Chen D, Smith MA et al (2012) Selection of reliable reference genes in Caenorhabditis elegans for analysis of nanotoxicity. PLoS ONE 7(3):e31849. https://doi.org/10.1371/journal.pone.0031849
Silver N, Best S, Jiang J et al (2006) Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol Biol 7:33. https://doi.org/10.1186/1471-2199-7-33
Pfaffl MW, Tichopad A, Prgomet C et al (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper–Excel-based tool using pair-wise correlations. Biotechnol Lett 26(6):509–515. https://doi.org/10.1023/B:BILE.0000019559.84305.47
Andersen CL, Jensen JL, Orntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64(15):5245–5250. https://doi.org/10.1158/0008-5472.CAN-04-0496
Vandesompele J, De Preter K, Pattyn F et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3(7):34. https://doi.org/10.1186/gb-2002-3-7-research0034
Bustin SA, Benes V, Garson JA et al (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55(4):611–622. https://doi.org/10.1373/clinchem.2008.112797
Conery AL, Larkins-Ford J, Ausubel FM et al (2014) High-throughput screening for novel anti-infectives using a C. elegans pathogenesis model. Curr Protoc Chem Biol 6(1):25–37. https://doi.org/10.1002/9780470559277.ch130160
Zhang C, Xie H, Xu CL et al (2012) Differential expression of Rs-eng-1b in two populations of Radopholus similis (Tylenchida: pratylecnchidae) and its relationship to pathogenicity. Eur J Plant Pathol 133:899–910. https://doi.org/10.1007/s10658-012-0015-4
Acknowledgements
This work was supported by National Key Research and Development Program of China (2018YFD0201208), National Natural Science Foundation of China (No. 31871941) and the Hunan Province Key Research and Development Program (2016NK2196).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, D., Liu, Y., Zhang, D. et al. Selection of reliable reference genes for gene expression studies in Caenorhabditis elegans exposed to crystals (Cry1Ia36) protein of Bacillus thuringiensis. Mol Biol Rep 46, 5767–5776 (2019). https://doi.org/10.1007/s11033-019-05010-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-019-05010-3