Abstract
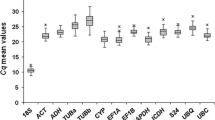
Quantitative real-time reverse transcription polymerase chain reaction (qPCR) has become the preferred method for studying low-abundant mRNA expression. Appropriate application of qPCR in such studies requires the use of reference gene(s) as an internal control in order to normalize the mRNA levels between different samples for an exact comparison of gene expression levels. Expression of the reference gene should be independent from development stage, cell/tissue types, treatments and environmental conditions. Recognizing the importance of reference gene(s) in normalization of qPCR data, various reference genes have been evaluated for stable expression under specific conditions in various organisms. In plants, only a few of them have been investigated, and very few reports about such reference genes in citrus. In the present study, seven candidate reference genes (18SrRNA, ACTB, rpII, UBQI, UBQ10, GAPDH and TUB) were tested, and three of them (18SrRNA, ACTB and rpII) proved to be the most stable ones among six leaf samples of different citrus genotypes. The three candidate reference genes were further analyzed for their stability of expression in five different tissues, and the results indicated that they were not completely stable. It is commonly accepted that gene expression studies should be normalized using more than one reference gene. Based on our results, we propose the use of the mean result rendered by18SrRNA, ACTB and rpII as reference genes to normalize mRNA levels in qPCR analysis of diverse cultivars and tissues of citrus. These results may provide a guideline for future works on gene expression in citrus by using qPCR.




Similar content being viewed by others
References
Bustin SA, Benes V, Nolan T, Pfaffl MW (2005) Quantitative real time RT-PCR—a perspective. J Mol Endocrinol 34:597–601
Gachon C, Mingam A, Charrier B (2004) Real-time PCR: What relevance to plant studies? J Exp Bot 55(402):1445–1454
Bustin SA (2005) Real-time, fluorescence-based quantitative PCR: a snapshot of current procedures and preferences. Expert Rev Mol Diagn 5(4):493–498
Bustin SA, Nolan T (2004) Pitfalls of quantitative real-time reverse transcription polymerase chain reaction. J Biomol Tech 15(3):155–166
Bustin SA (2002) Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J Mol Endocrinol 29:23–39
Andersen CL, Jensen JL, Orntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64(15):5245–5250
Von SD, Leverkoehne I, Von Samson-Himmelstjerna G, Gruber AD (2005) Impact of formalin-fixation and paraffin-embedding on the ratio between mRNA copy numbers of differently expressed genes. Histochem Cell Biol 124(2):177–188
Jain M, Nijhawan A, Tyagi AK, Khurana JP (2006) Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem Biophys Res Commun 345:646–651
Suzuki T, Higgins PJ, Crawford DR (2000) Control selection for RNA quantitation. Biotechniques 29(2):332–337
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR by geometric averaging of multiple internal control genes. Genome Biol 3(7):34
Tricarico C, Pinzani P, Bianchi S, Paglierani M, Distante V, Pazzagli M, Bustin SA, Orlando C (2002) Quantitative real-time reverse transcription polymerase chain reaction: normalization to rRNA or single housekeeping genes is inappropriate for human tissue biopsies. Anal Biochem 309(2):293–300
Perez S, Royo L, Astudillo A, Escudero D, Alvarez F, Rodriguez A, Gomez E, Otero J (2007) Identifying the most suitable endogenous control for determining gene expression in hearts from organ donors. BMC Mol Biol 8(1):114
Yang YF, Hou S, Cui GH, Chen SL, Wei JH, Huang LQ (2010) Characterization of reference genes for quantitative real-time PCR analysis in various tissues of Salvia miltiorrhiza. Mol Biol Rep 37:507–513
Jiang HB, Liu YH, Tang PA, Zhou AW, Wang JJ (2010) Validation of endogenous reference genes for insecticide-induced and developmental expression profiling of Liposcelis bostsrychophila (Psocoptera: Liposcelididae). Mol Biol Rep 37:1019–1029
Peng H, Cheng HY, Yu XW, Shi QH, Zhang H, Li JH, Ma H (2010) Molecular analysis of an actin gene, CarACT1, from chickpea (Cicer arietinum L.). Mol Biol Rep 37:1081–1088
Wu WJ, Ren ZQ, Wang Y, Chao Zh, Xu EQ, Xiong YZ (2010) Molecular characterization, expression patterns and polymorphism analysis of porcine Six1 gene. Mol Biol Rep. doi:10.1007/s11033-010-0403-9
Li GL, Roy B, Li XH, Yue WF, Wu XF, Liu JM, Zhang CX, Miao YG (2009) Quantification of silkworm coactivator of MBF1 mRNA by SYBR Green I real-time RT-PCR reveals tissue- and stage-specific transcription levels. Mol Biol Rep 36:1217–1223
Schmittgen TD, Zakrajsek BA (2000) Effect of experimental treatment on housekeeping gene expression: validation by real-time quantitative RT-PCR. J Biochem Biophys Methods 46(1–2):69–81
Foss DL, Baarsch MJ, Murtaugh MP (1998) Regulation of hypoxanthine phosphor ribosyl transferase, glyceraldehyde-3-phosphate dehydrogenase and beta-actin mRNA expression in porcine immune cells and tissues. Anim Biotechnol 9(1):67–78
Thellin O, Zorzi W, Lakaye B, De Borman B, Coumans B, Hennen G, Grisar T, Igout A, Heinen E (1999) Housekeeping genes as internal standards: use and limits. J Biotechnol 75(2–3):291–295
Lee PD, Sladek R, Greenwood CM, Hudson TJ (2002) Control genes and variability: absence of ubiquitous reference transcripts in diverse mammalian expression studies. Genome Res 12(2):292–297
Tomasz C, Mark S, Thomas A, Udvardi MK, Scheible W-R (2005) Genome-wide identification and testing of superior reference genes for transcript normalization in Arabidopsis1[w]. Plant Physiol 139:5–17
Aoki T, Naka H, Katagiri T, Hirono I (2000) Cloning and characterization of glyceraldehyde-3-phosphate dehydrogenase cDNA of Japanese flounder Paralichthys olivaceus. Fish Sci 66:737–742
Olsvik PA, Lie KK, Jordal AEO, Nilsen TO, Hordvik I (2005) Evaluation of potential reference genes in real-time RT-PCR studies of Atlantic salmon. BMC Mol Biol 6:21
Zhang Z, Hu J (2007) Development and validation of endogenous reference genes for expression profiling of medaka (Oryzias latipes) exposed to endocrine disrupting chemicals by quantitative real-time RT-PCR. Toxicol Sci 95(2):356–368
Huggett J, Dheda K, Bustin SA, Zumla A (2005) Real-time RT-PCR normalization: strategies and considerations. Genes Immun 6(4):279–284
Ohl F, Jung M, Radonic A, Sachs M, Loening SA, Jung K (2006) Identification and validation of suitable endogenous reference genes for gene expression studies of human bladder cancer. J Urol 175(5):1915–1920
Dheda K, Huggett J, Chang J, Kim L, Bustin S, Johnson M, Rook G, Zumla A (2005) The implications of using an inappropriate reference gene for real-time reverse transcription PCR data normalization. Anal Biochem 344:141–143
Sturzenbaum SR, Kille P (2001) Control genes in quantitative molecular biological techniques: the variability of invariance. Comp Biochem Physiol B Biochem Mol Biol 130(3):281–289
Radonic A, Thulke S, Mackay IM, Landt O, Siegert W, Nitsche A (2004) Guideline to reference gene selection for quantitative realtime PCR. Biochem Biophys Res Commun 313(4):856–862
Brunner AM, Yakovlev IA, Strauss SH (2004) Validating internal controls for quantitative plant gene expression studies. BMC Plant Biol 4:14
Nathalie N, Jean FH, Lucien H, Evers D (2005) Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56(421):2907–2914
Ding J, Jia J, Yang L, Wen H, Zhang C, Liu W, Zhang D (2004) Validation of a rice specific gene, sucrose phosphate synthase, used as the endogenous reference gene for qualitative and real-time quantitative PCR detection of transgenes. J Agric Food Chem 52(11):3372–3377
Kim BR, Nam HY, Kim SU, Kim SI, Chang YJ (2003) Normalization of reverse transcription quantitative-PCR with housekeeping genes in rice. Biotechnol Lett 25(21):1869–1872
Gunnhild WT, Ian KT, May BB (2007) Evaluation of reference genes for real-time RT-PCR expression studies in the plant pathogen Pectobacterium atrosepticum. BMC Plant Biol 7:50
Jian B, Liu B, Bi Y, Hou W, Wu C, Han T (2008) Validation of internal control for gene expression study in soybean by quantitative real-time PCR. BMC Mol Biol 9(1):59
Libault M, Thibivilliers S, Bilgin D, Radwan O, Benitez M, Clough S, Stacey G (2008) Identification of four soybean reference genes for gene expression normalization. Plant Genome 1:44–54
Tong ZG, Gao ZH, Wang F, Zhou J, Zhang Z (2009) Selection of reliable reference genes for gene expression studies in peach using real-time PCR. BMC Mol Biol 10:71
Carla FB, Fábio ES, Mirian PM, Ivan GM (2009) Identification of suitable internal control genes for expression studies in Coffea arabica under different experimental conditions. BMC Mol Biol 10:1
Hayati MI, Robert SS, Rosanne EC, Graham DB, Donald JM, John MM (2004) Comparison of reference genes for quantitative real-time polymerase chain reaction analysis of gene expression in sugarcane. Plant Mol Biol Report 22:325–337
Expósito-Rodríguez M, Borges AA, Borges-Pérez A, Pérez JA (2008) Selection of internal control genes for quantitative real-time RT-PCR studies during tomato development process. BMC Plant Biol 8:131
Reid KE, Olsson N, Schlosser J, Peng F, Lund ST (2006) An optimized grapevine RNA isolation procedure and statistical determination of reference genes for real-time RT-PCR during berry development. BMC Plant Biol 6:27
Gonzalez-Verdejo CI, Die JV, Nadal S, Jimenez-Marin A, Moreno MT, Roman B (2008) Selection of housekeeping genes for normalization by real-time RTPCR: analysis of Or-MYB1 gene expression in Orobanche ramose development. Anal Biochem 379(2):176–181
Dong LM, Sui C, Liu YJ, Yang Y, Wei JH, Yang YF (2010) Validation and application of reference genes for quantitative gene expression analyses in various tissues of Bupleurum chinense. Mol Biol Rep. doi:10.1007/s11033-010-0648-3
Hong S-Y, Seo PJ, Yang M-S, Xiang F, Park C-M (2008) Exploring valid reference genes for gene expression studies in Brachypodium distachyon by real-time PCR. BMC Plant Biol 8:112
Mallona I, Lischewski S, Weiss J, Hause B, Egea-Cortines M (2010) Validation of reference genes for quantitative real-time PCR during leaf and flower development in Petunia hybrida. BMC Plant Biol 10:4
Maroufi A, Van Bockstaele E, De Loose M (2010) Validation of reference genes for gene expression analysis in chicory (Cichorium intybus) using quantitative real-time. PCR BMC Mol Biol 11:15
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper-Excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515
Mason MG, Schmidt S (2002) Rapid isolation of total RNA and genomic DNA from Hakea actities. Funct Plant Biol 29:1013–1016
Rasmussen R, Meuer S, Wittwer C, Nakagawara K (eds) (2001) Quantification on the LightCycler. In: Rapid cycle real-time PCR, methods and applications. Springer, Berlin, pp 21–34
BestKeeper Software. http://www.gene-quantification.de/bestkeeper.html. (Ref. Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper-Excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515)
geNorm Software. http://medgen.ugent.be/~jvdesomp/genorm/. (Ref. Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR by geometric averaging of multiple internal control genes. Genome Biol 3(7):34)
Barrett HC, Rhodes AM (1976) A numerical taxonomic study of affinity relationships in cultivated citrus and its close relatives. Syst Bot 1:105–136
Nicolosi E, Deng ZN, Gentile A, La Malfa S, Continella G, Tribulato E (2000) Citrus phylogeny and genetic origin of important species as investigated by molecular markers. Theor Appl Genet 100:1155–1166
Deng ZN, La Malfa S, Xie YM, Xiong XY, Gentile A (2007) Identification and evaluation of chloroplast uni- and trinucleotide sequence repeats in citrus. Hortic Sci 111:186–192
Spiegel-Roy P, Goldschmidt EE (1996) Biology of citrus. Cambridge University Press, Cambridge, p 230
Steinau M, Rajeevan MS, Unger ER (2006) DNA and RNA references for qRT-PCR assays in exfoliated cervical cells. J Mol Diagn 8:113–118
Ohl F, Jung M, Xu C, Stephan C, Rabien A, Burkhardt M, Nitsche A, Kristiansen G, Loening SA, Radonic A, Jung K (2005) Gene expression studies in prostate cancer tissue: which reference gene should be selected for normalization? J Mol Med 83(12):1014–1024
Silver N, Best S, Jiang J, Thein SL (2006) Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time. PCR BMC Mol Biol 7:33
Claus LA, Jens LJ, Ørntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64:5245–5250
Duncan A, Dylan NC, Fiona S, Philip JR (2007) Expression stability of commonly used reference genes in canine articular connective tissues. BMC Vet Res 3:7
Hellemans J, Mortier G, Paepe AD, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8:R19
Acknowledgments
We thank A.P. Xiaojun Wang for assistance in data analysis and the Center of Analytical Service of Hunan Agricultural University for devices support. This work was supported by the National Natural Science Foundation of China (30871702) and the China Modern Agricultural Research System (CARS-27).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, J., Yuan, F., Long, G. et al. Selection of reference genes for quantitative real-time RT-PCR analysis in citrus. Mol Biol Rep 39, 1831–1838 (2012). https://doi.org/10.1007/s11033-011-0925-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-0925-9