Abstract
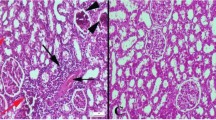
We investigated the antioxidant preventive effect of betaine on isoprenaline-induced myocardial infarction in male albino rats. Isoprenaline induced myocardial infarction was manifested by a moderate elevation in the levels of diagnostic marker enzymes (alanine aminotransferase, aspartate aminotransferase, lactate dehydrogenase and creatine phosphokinase) and homocysteine in plasma of experimental rats. Significant rise in the level of lipid peroxidation with a concomitant decline in the levels of myocardial non-enzymic (reduced glutathione) and enzymic antioxidants (glutathione peroxidase, glutathione-S-transferase, catalase and superoxide dismutase) was also observed. Oral pretreatment with betaine significantly prevented isoprenaline-induced alterations in the levels of diagnostic marker enzymes and homocysteine in plasma of experimental groups of rats. It counteracted the isoprenaline-induced lipid peroxidation and maintained the myocardial antioxidant defense system at near normal. Histopathological observations also confirmed the protective effect of betaine against isoprenaline-induced myocardial infarction. The results of the present investigation indicate that the protective effect of betaine is probably related to its ability to strengthen the myocardial membrane by its membrane stabilizing action or to a counteraction of free radicals by its antioxidant property.





Similar content being viewed by others
References
Mackay J, Mensha GA (2004) In deaths from coronary artery disease. In: Atlas of heart disease and stroke. World Health Organization in collaboration with the Centre for Disease Control and Prevention, Geneva, pp 48–49
Patil SS, Joshi R, Gupta G, Reddy MV, Pai M, Kalantri SP (2004) Risk factors for acute myocardial infarction in a rural population of central India: a hospital-based case-control study. Natl Med J India 17:189–194
Cooper R, Cutler J, Desvignes-Nickens P, Fortmann SP, Friedman L, Havlik R, Hogelin G, Marler J, McGovern P, Morosco G, Mosca L, Pearson T, Stamler J, Stryer D, Thom T (2000) Trends and disparities in coronary heart disease, stroke, and other cardiovascular disease in the United States; findings of the National Conference on cardiovascular disease prevention. Circulation 102:3137–3147
Craig SA (2004) Betaine in human nutrition. Am J Clin Nutr 80:539–549
Niculescu MD, Zeisel SH (2002) Diet, methyl donors and DNA methylation: interactions between dietary folate, methionine and choline. J Nutr 132:2333S–2335S
Kim SK, Kim YC (2002) Attenuation of bacterial lipopolysaccharide-induced hepatotoxicity by betaine or taurine in rats. Food Chem Toxicol 40:545–549. doi:10.1016/S0278-6915(01)00102-8
Erman F, Balkan J, Cevikbas U, Kocak-Toker N, Uysal M (2004) Betaine or taurine administration prevents fibrosis and lipid peroxidation induced by rat liver by ethanol plus carbon tetrachloride intoxication. Amino Acids 27:199–205. doi:10.1007/s00726-004-0105-5
Ganesan B, Rajesh R, Anandan R, Dhandapani N (2007) Biochemical studies on the protective effect of betaine on mitochondrial function in experimentally induced myocardial infarction in rats. J Health Sci 53:671–681. doi:10.1248/jhs.53.671
Ganesan B, Rajesh R, Anandan R, Dhandapani N (2007) Protective effect of betaine on changes in the levels of protein, glycoproteins and amino acids in isoprenaline-induced myocardial infarction in rats. Afr J Biochem Res 1:117–123
Ganesan B, Anandan R, Rajesh R (2008) Protective effect of betaine on changes in lipid profile, lipoproteins and fatty acid composition in experimentally induced myocardial infarction in Wistar rats. Int J Biomed Pharm Sci 2:65–69
Shiny KS, Kumar SH, Farvin KH, Anandan R, Devadasan K (2005) Protective effect of taurine on myocardial antioxidant status in isoprenaline-induced myocardial infarction in rats. J Pharm Pharmacol 57:1313–1317. doi:10.1211/jpp.57.10.0010
Farvin KHS, Anandan R, Kumar SHS, Shiny KS, Mathew S, Sankar TV, Nair PGV (2006) Cardioprotective effect of squalene on lipid profile in isoprenaline-induced myocardial infarction in rats. J Med Food 9:531–536. doi:10.1089/jmf.2006.9.531
Karthick M, Stanely Mainzen Prince P (2006) Preventive effect of rutin, a bioflavonoid, on lipid peroxides and antioxidants in isoproterenol-induced myocardial infarction in rats. J Pharm Pharmacol 58:701–707. doi:10.1211/jpp.58.5.0016
Padmanabhan M, Prince PS (2006) Preventive effect of S-allylcysteine on lipid peroxides and antioxidants in normal and isoproterenol-induced cardiotoxicity in rats: a histopathological study. Toxicology 224:128–137. doi:10.1016/j.tox.2006.04.039
Anandan R, Asha KK, Ammu K, Mathew S, Viswanathan Nair PG (2003) Effects of peroxidised PUFA on tissue defense system in experimentally induced myocardial infarction in rats. In: Surendran PK, Mathew PT, Thampuran N, Nambiar N, Joseph J, Boopendranath MR, Lakshmanan PT, Viswanathan Nair PG (eds) Seafood safety. Society of Fisheries Technologists, Cochin, pp 330–335
Mohur A, Cook IJY (1957) Simple methods for measuring serum levels of glutamic-oxalo acetic and glutamic-pyruvic transaminase in routine laboratories. J Clin Pathol 10:394–399. doi:10.1136/jcp.10.4.394
King J (1965) Lactate dehydrogenase. In: Van D (ed) Practical clinical enzymology. Nostrand Co, London, pp 83–93
Okinaka S, Kumagai H, Ebashi S, Sugita H, Momoi H, Toyokura Y, Fujie Y (1961) Serum creatine phosphokinase. Activity in progressive muscular dystrophy and neuromuscular diseases. Arch Neurol 4:520–525
Yuan CS (2002) Methods and composition for assaying analytes. US Patent US 6.376.210 B.1
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. doi:10.1016/0003-2697(79)90738-3
Ellman GL (1959) Tissue sulfydril groups. Arch Biochem Biophys 82:70–77. doi:10.1016/0003-9861(59)90090-6
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Misra HP, Fridovich I (1972) The role of superoxide anion in the auto oxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Takahara S, Hamilton HB, Neel JV, Kobara TY, Ogura Y, Nishimura ET (1960) Hypocatalasemia—a new genetic carried state. J Clin Invest 39:610–619. doi:10.1172/JCI104075
Farvin KHS, Anandan R, Kumar SHS, Shiny KS, Sankar TV, Thankappan TK (2004) Effect of squalene on tissue defense system in isoproterenol-induced myocardial infarction in rats. Pharmacol Res 50:231–236. doi:10.1016/j.phrs.2004.03.004
Rajadurai M, Stanely Mainzen Prince P (2007) Preventive effect of naringin on cardiac markers, electrocardiographic patterns and lysosomal hydrolases in normal and isoproterenol-induced myocardial infarction in Wistar rats. Toxicology 230:178–188. doi:10.1016/j.tox.2006.11.053
Kharbanda KK, Mailliard ME, Baldwin CR, Beckenhauer HC, Sorrell MF, Tuma DJ (2007) Betaine attenuates alcoholic steatosis by restoring phosphatidylcholine generation via the phosphatidylethanolamine methyltransferase pathway. J Hepatol 46:314–321. doi:10.1016/j.jhep.2006.08.024
Junnila M, Rahko T, Sukura A, Lindberg LA (2000) Reduction of carbon tetrachloride-induced hepatotoxic effects by oral administration of betaine in male Han-Wistar rats: a morphometric histological study. Vet Pathol 37:231–238. doi:10.1354/vp.37-3-231
Nasir K, Tsai M, Rosen BD, Fernandes V, Bluemke DA, Folsom AR, Lima JA (2007) Elevated homocysteine is associated with reduced regional left ventricular function: the multi-ethnic study of atherosclerosis. Circulation 115:180–187. doi:10.1161/CIRCULATIONAHA.106.633750
Hagar HH (2002) Folic acid and vitamin B (12) supplementation attenuates isoprenaline-induced myocardial infarction in experimental hyperhomocysteinemic rats. Pharmacol Res 46:213–219. doi:10.1016/S1043-6618(02)00095-6
Tawakol A, Forgione MA, Stuehlinger M, Alpert NM, Cooke JP, Loscalzo J, Fischman AJ, Creager MA, Gewirtz H (2002) Homocysteine impairs coronary microvascular dilator function in humans. J Am Coll Cardiol 40:1051–1058. doi:10.1016/S0735-1097(02)02069-7
Shai I, Stampfer MJ, Mab J, Manson JE, Hankinson SE, Cannuscio C, Selhub J, Curhanc G, Rimma EB (2004) Homocysteine as a risk factor for coronary heart diseases and its association with inflammatory biomarkers, lipids and dietary factors. Atherosclerosis 177:375–381. doi:10.1016/j.atherosclerosis.2004.07.020
van Aken BE, Jansen J, van Deventer SJ, Reitsma PH (2000) Elevated levels of homocysteine increase IL-6 production in monocytic Mono Mac 6 cells. Blood Coagul Fibrinolysis 11:159–164
Silverman MD, Tumuluri RJ, Davis M, Lopez G, Rosenbaum JT, Lelkes PI (2002) Homocysteine upregulates vascular cell adhesion molecule-1 expression in cultured human aortic endothelial cells and enhances monocyte adhesion. Arterioscler Thromb Vasc Biol 22:587–592. doi:10.1161/01.ATV.0000014221.30108.08
Prontera C, Martelli N, Evangelista V, D’Urbano E, Manarini S, Recchiuti A, Dragani A, Passeri C, Davi G, Romano M (2007) Homocysteine modulates the CD40/CD40L system. J Am Coll Cardiol 49:2182–2190. doi:10.1016/j.jacc.2007.02.044
Geng B, Chang L, Du JB, Tang CS (2005) A new strategy to treat hyperhomocysteinemia. Beijing Da Xue Xue Bao 37:215–219
Sathish V, Ebenezar KK, Devaki T (2003) Synergistic effect of Nicorandil and Amlodipine on tissue defense system during experimental myocardial infarction in rats. Mol Cell Biochem 243:133–138. doi:10.1023/A:1021612230000
Haidara MA, Yassin HZ, Rateb M, Ammar H, Zorkani MA (2006) Role of oxidative stress in development of cardiovascular complications in diabetes mellitus. Curr Vasc Pharmacol 4:215–227. doi:10.2174/157016106777698469
Del Monte U (2005) Swelling of hepatocytes injured by oxidative stress suggests pathological changes related to macromolecular crowding. Med Hypotheses 64:818–825. doi:10.1016/j.mehy.2004.08.028
Kanbak G, Akyuz F, Inal M (2001) Preventive effect of betaine on ethanol-induced membrane lipid composition and membrane ATPases. Arch Toxicol 75:59–61. doi:10.1007/s002040000179
Balkan J, Oztezcan S, Kucuk M, Cevikbas U, Kocak-Toker N, Uysal M (2004) The effect of betaine treatment on triglyceride levels and oxidative stress in the liver of ethanol-treated guinea pigs. Exp Toxicol Pathol 55:505–509. doi:10.1078/0940-2993-00347
Kumar SHS, Anandan R, Devaki T, Kumar MS (2001) Cardioprotective effects of Picrorrhiza kurroa against isoprenaline-induced myocardial stress in rats. Fitoterapia 72:402–405. doi:10.1016/S0367-326X(01)00264-7
Anandan R, Nair PG, Mathew S (2004) Anti-ulcerogenic effect of chitin and chitosan on mucosal antioxidant defence system in HCl-ethanol-induced ulcer in rats. J Pharm Pharmacol 56:265–269. doi:10.1211/0022357023079
Anandan R, Devi KP, Devaki T, Govindaraj P (1998) Preventive effects of Picrochiza kurroa on d-galactosamine-induced hepatitis in rats. J Clin Biochem Nutr 25:87–95
Kalra J, Lautner K, Massey L, Prasad K (1988) Oxygen free radicals induced release of lysosomal enzymes in vitro. Mol Cell Biochem 84:233–238. doi:10.1007/BF00421058
Saravanan G, Prakash J (2004) Effect of garlic (Allium sativum) on lipid peroxidation in experimental myocardial infarction in rats. J Ethnopharmacol 94:155–158. doi:10.1016/j.jep.2004.04.029
Barak AJ, Beckenhauer HC, Badakhsh S, Tuma DJ (1997) The effect of betaine in reversing alcoholic steatosis. Alcohol Clin Exp Res 21:1100–1102
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ganesan, B., Buddhan, S., Anandan, R. et al. Antioxidant defense of betaine against isoprenaline-induced myocardial infarction in rats. Mol Biol Rep 37, 1319–1327 (2010). https://doi.org/10.1007/s11033-009-9508-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-009-9508-4