Abstract
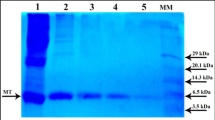
Metallothioneins are rather ubiquitous metal-binding proteins induced by stressing or physiological stimuli. Two major metallothionein isoforms have been identified in mussel: MT10 and MT20. Nevertheless the high sequence homology, the two isoforms exhibit different expression and inducibility in vivo. We cloned and produced in Escherichia coli the MT20 isoform from Mytilus galloprovincilis. cDNA was subcloned into pGEX-6P.1 vector, in frame with a sequence encoding a glutathione-S-transferase (GST) tail. Recombinant protein was purified to electrophoretic homogeneity by affinity chromatography. After enzymatic cleavage of the GST tail the MT moiety was recovered with a final yield of about 5 mg of protein per litre of bacterial culture. The metal-binding ability of MT20 was assessed by absorption spectroscopy upon addition of cadmium equivalents and the metal release was checked as a function of the environment pH. Moreover the protein was analysed for the propensity to polymerization, typical of this class of protein, before and after exposure to reducing and alkylating agents.







Similar content being viewed by others
Abbreviations
- DTNB:
-
5′,5′-dithio-bis(2-nitrobenzoic acid)
- DTT:
-
1,4 dithio-dl-threitol
- GST:
-
glutathione S-transferase
- IA:
-
iodoacetamide
- IPTG:
-
isopropyl-β-d-thiogalactopyranoside
- MT:
-
metallothionein
- NEM:
-
N-ethylmaleimide
Reference
Hamer DH (1986) Metallothionein. Annu Rev Biochem 55:913–951
Coyle P, Philcox JC, Carey LC, Rofe AM (2002) Metallothionein:the multipurpose protein. Cell Mol Life Sci 59:627–647
Kägi JHR (1993) Evolution, structure, and chemical activity of class I Metallothioneins: An overview. In: Suzuki KT et al (eds) Metallothionein III. Birkhauser Verlag, Basel/Switzerland. pp. 29–55
Braun W, Vasak M, Robbins AH, Stout CD, Wagner G, Kägi JH, Wuthrich K (1992) Comparison of the NMR solution structure and the x-ray crystal structure of rat metallothionein-2. Proc Natl Acad USA 89:10124–10128
Geret F, Cosson RP (2002) Induction of specific isoforms of metallothionein in mussel tissues after exposure to cadmium or mercury. Arch Environ Contam Toxicol 42:36–42
Frazier JM (1986) Cadmium-binding proteins in the mussel, Mytilus edulis. Environ Health Perspect 65:39–43
Mackay EA, Overnell J, Dunbar B, Davidson I, Hunziker PE, Kägi JHR, Fothergill JE (1993) Complete amino acid sequences of five dimeric and four monomeric forms of metallothionein from the edible mussel Mytilus edulis. Eur J Biochem 218:183–194
Barsyte D, White KN, Lovejoy DA (1999) Cloning and characterization of metallothionein cDNAs in the mussel Mytilus edulis L. digestive gland, Comp. Biochem. Physiol. C. Pharmacol. Toxicol. Endocrinol 122:287–296
Lemoine S, Bigot Y, Sellos D, Cosson RP, Laulier M, (2000) Metallothionein Isoforms in Mytilus edulis (Mollusca, Bivalvia): Complementary DNA Characterization and Quantification of Expression in Different Organs after Exposure to Cadmium, Zinc, and Copper. Mar Biotechnol (NY) 2:195–203
Dondero F, Piacentini L, Banni M, Rebelo M, Burlando B, Viarengo A (2005) Quantitative PCR analysis of two molluscan metallothionein genes unveils differential expression and regulation. Gene 345:259–270
Dallinger R, Berger B, Hunziker PE, Birchler N, Hauer CN, Kägi JHR, (1993) Purification and primary structure of snail metallothionein. Similarity of the N-terminal sequence with histones H4 and H2A. Eur J Biochem 216:739–746
Simens DC, Bebianno MJ, Moura JJ (2003) Isolation and characterisation of metallothionein from the clam Ruditapes decussates. Aquatic Toxicol 63:307–318
Vergani L, Canneva F, Ghisellini P, Nicolini C (2002) Expression, purification, and structural characterization of Human Histone H4. Protein Expr Purif 24:420–428
Needleman SB, Wunsch CD (1970) A general method applicable to the search for similarities in the amino acid sequence of two proteins. J Mol Biol 48:443–453
Dagert M, Ehrlich SD (1979) Prolonged incubation in calcium chloride improves the competence of Escherichia coli cells. Gene 6:23–28
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Schägger H, von Jagow G (1987) Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem 166:368–379
Vergani L, Grattarola M, Borghi C, Dondero F, Viarengo A (2005) Fish and molluscan metallothioneins. FEBS J 272:6014–6023
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilising the principle of protein-dye binding. Anal Biochem 72:142–146
Bühler RHO, Kägi JHR (1979) Spectroscopic properties of zinc-metallothionein. Exp Suppl 34:211–220
Viarengo A, Ponzano E, Dondero F, Fabbri R (1997) A simple spectrophotometric method for metallothionein evaluation in marine organism: an application to mediterranean and antarctic molluscs. Marine Env Res 44:69–87
Kägi JHR (1991) Overview of metallothionein. Methods Enzymol 205:613–626
Vergani L, Grattarola M, Dondero F, Viarengo A (2003) Expression, purification, and characterization of metallothionein-A from rainbow trout. Protein Expr Purif 27:338–345
Zangger K, Shen G, Oz G, Otvos JD, Armitage IM (2001) Oxidative dimerization in metallothionein is a result of intermolecular disulphide bonds between cysteines in the alpha-domain. Biochem J 15:353–360
Means GE, Feeney RE (1971) Chemical modification of Proteins, Holden-Day, Inc San Francisco pp 271–220
Acknowledgments
We would like to extend our gratitude to Prof. Gabriella Gallo for her scientific collaboration, to Drs. Cristina Borghi and Cristina Lanza for the technical assistance. This research was supported by the University of Genova Project for year 2002. This work was also granted by the 5th UE Framework Program project Biological Effects of Environmental Pollution in marine coastal ecosystems (BEEP). (Contract N°.EVK3–2000–00543).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grattarola, M., Carloni, M., Dondero, F. et al. Expression, purification and preliminary characterization of mussel (Mytilus galloprovincialis) metallothionein MT20. Mol Biol Rep 33, 265–272 (2006). https://doi.org/10.1007/s11033-006-9009-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-006-9009-7