Abstract
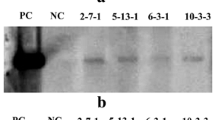
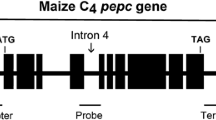
Pyruvate, orthophosphate dikinase (PPDK) is a key enzyme in the C4 photosynthetic pathway of maize. To improve the cold tolerance of the enzyme in maize, we designed two genomic sequence-based constructs in which the carboxy-terminal region of the enzyme was modified to mimic the amino acid sequence of the cold-tolerant PPDK of Flaveria brownii (Asteraceae). A large amount of PPDK was found to have accumulated in the leaves of many of the maize plants transformed with one of these constructs – that which introduced 17 amino acid substitutions without any alteration of the exon-intron structure – although there was a wide range of variation in the amount of PPDK among the separate plants. In contrast, the production was much less in maize transformed with the second construct in which a cDNA fragment for the same carboxy-terminal region was inserted. The specific activity of PPDK in the plants transformed with the gene with the amino acid substitutions was inversely correlated with the amount of enzyme in the leaves. In addition, the activity of the cold-tolerant recombinant enzyme was judged to be regulated by the PPDK regulatory protein, similar to that of the native PPDK. The cold tolerance of PPDK in crude leaf extracts was greatly improved in plants that produced a large amount of the engineered PPDK. The photosynthetic rate at 8°C increased significantly (by 23%, p<0.05), but there was no obvious effect at higher temperatures. These results support the hypothesis that PPDK is one of the limiting factors in the C4 photosynthesis of maize under cold conditions.
Similar content being viewed by others
Abbreviations
- PEPC:
-
Phosphoenolpyruvate carboxylase
- PPDK:
-
Pyruvate, orthophosphate dikinase
References
Aoyagi K, Bassham JA, Greene FC (1984) Pyruvate orthophosphate dikinase gene expression in developing wheat seeds. Plant Physiol 75:393–396
Ashton AR, Burnell JN, Hatch MD (1984) Regulation of C4 photosynthesis: inactivation of pyruvate, Pi dikinase by ADP-dependent phosphorylation and activation by phosphorolysis. Arch Biochem Biophys 230:492–503
Burnell JN (1990) A comparative study of the cold-sensitivity of pyruvate, Pi dikinase in Flaveria species. Plant Cell Physiol 31:295–297
Callis J, Fromm M, Walbot V (1987) Introns increase gene expression in cultured maize cells. Genes Dev 1:1183–1200
Christensen AH, Sharrock RA, Quail PH (1992) Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol Biol 18:675–689
Denecke J, Gosele J, Botterman J, Cornelissen M (1989) Quantitative analysis of transiently expressed gene in plant cells. Methods Mol Cell Biol 1:19–27
Du Y-C, Nose A, Wasano K (1999) Thermal characteristics of C4 photosynthetic enzymes from leaves of three sugarcane species differing in cold sensitivity. Plant Cell Physiol 40:298–304
Fukayama H, Tsuchida H, Agarie S, Nomura M, Onodera H, Ono K, Lee BH, Hirose S, Toki S, Ku MS, Makino A, Matsuoka M, Miyao M (2001) Significant accumulation of C4-specific pyruvate, orthophosphate dikinase in a C3 plant, rice. Plant Physiol 127:1136–1146
Glackin CA, Grula JW (1990) Organ-specific transcripts of different size and abundance derive from the same pyruvate, orthophosphate dikinase gene in maize. Proc Natl Acad Sci USA 87:3004–3008
Hatch MD (1979) Regulation of C4 carbon pathway photosynthesis: factors affecting cold mediated inactivation and reactivation of pyruvate orthophosphate dikinase EC-2.7.9.1. Aust J Plant Physiol 6:607–619
Ho SN, Hunt HD, Horton RM, Pullen JK, Pease LR (1989) Site-directed mutagenesis by overlap extension using the polymerase chain reaction. Gene 15:51–59
Imaizumi N, Ku MS, Ishihara K, Samejima M, Kaneko S, Matsuoka M (1997). Characterization of the gene for pyruvate, orthophosphate dikinase from rice, a C3 plant, and a comparison of structure and expression between C3 and C4 genes for this protein. Plant Mol Biol 34:701–716
Ishida Y, Saito H, Ohta S, Hiei Y, Komari T, Kumashiro T (1996) High efficiency transformation of maize (Zea mays L.) mediated by Agrobacterium tumefaciens. Nat Biotechnol 14:745–750
Ishida Y, Murai N, Kuraya Y, Ohta S, Saito H, Hiei Y, Komari T (2004) Improved co-transformation of maize with vectors carrying two separate T-DNAs mediated by Agrobacterium tumefaciens. Plant Biotechnol 21:57–63
Jenkins CL, Hatch MD (1985) Properties and reaction mechanism of C4 leaf pyruvate, Pi dikinase. Arch Biochem Biophys 239:53–62
Kawabe A, Miyashita NT (2003) Patterns of codon usage bias in three dicot and four monocot plant species. Genes Genet Syst 78:343–352
Komari T, Hiei Y, Saito Y, Murai N, Kumashiro T (1996) Vectors carrying two separate T-DNAs for co-transformation of higher plants mediated by Agrobacterium tumefaciens and segregation of transformants free from selection markers. Plant J 10:165–174
Ku MS, Kano-Murakami Y, Matsuoka M (1996) Evolution and expression of C4 photosynthesis genes. Plant Physiol 111:949–957
Ku MS, Agarie S, Nomura M, Fukayama H, Tsuchida H, Ono K, Hirose S, Toki S, Miyao M, Matsuoka M (1999) High-level expression of maize phosphoenolpyruvate carboxylase in transgenic rice plants. Nat Biotechnol 17:76–80
Matsuoka M (1990) Structure, genetic mapping, and expression of the gene for pyruvate, orthophosphate dikinase from maize. J Biol Chem 265:16772–16777
Matsuoka M (1995) The gene for pyruvate, orthophosphate dikinase in C4 plants: structure, regulation and evolution. Plant Cell Physiol 36:1–7
McElroy D, Zhang W, Cao J, Wu R (1990) Isolation of an efficient actin promoter for use in rice transformation. Plant Cell 2:163–171
Murray EE, Lotzer J, Eberle M (1989) Codon usage in plant genes. Nucleic Acids Res 17:477–498
Naidu SL, Moose SP, Al-Shoaibi AK, Raines CA, Long SP (2003) Cold tolerance of C4 photosynthesis in Miscanthus × giganteus: adaptation in amounts and sequence of C4 photosynthetic enzymes. Plant Physiol 132:1688–1697
Ohta S, Usami S, Ueki J, Kumashiro T, Komari T, Burnell JN (1996) Identification of the amino acid residues responsible for cold tolerance in Flaveria brownii pyruvate, orthophosphate dikinase. FEBS Lett 396:152–156
Ohta S, Ishida Y, Usami S (2004) Expression of cold-tolerant pyruvate, orthophosphate dikinase cDNA, and heterotetramer formation in transgenic maize plants. Transgenic Res 13:475–485
Rosche E, Westhoff P (1995) Genomic structure and expression of the pyruvate, orthophosphate dikinase gene of the dicotyledonous C4 plant Flaveria trinervia (Asteraceae). Plant Mol Biol 29:663–678
Sheen J (1991) Molecular mechanisms underlying the differential expression of maize pyruvate, orthophosphate dikinase genes. Plant Cell 3:225–245
Shirahashi K, Hayakawa S, Sugiyama T (1978) Cold lability of pyruvate,orthophosphate dikinase in the maize leaf. Plant Physiol 62:826–830
Simon J-P (1996) Molecular forms and kinetic properties of pyruvate, Pi dikinase from two populations of␣barnyard grass (Echinochloa crus-galli) from sites␣of contrasting climates. Aust J Plant Physiol 23:191–199
Sugiyama T (1973) Purification, molecular, and catalytic properties of pyruvate phosphate dikinase from the maize leaf. Biochemistry 12:2862–2868
Sugiyama T, Boku K (1976) Differing sensitivity of pyruvate orthophosphate dikinase to low temperature in maize cultivars. Plant Cell Physiol 17:851–854
Sugiyama T, Hirayama Y (1983) Correlation of activities of phosphoenolpyruvate carboxylase and pyruvate,orthophosphate dikinase with biomass in maize seedlings. Plant Cell Physiol 24:783–787
Tanaka A, Mita S, Ohta S, Kyozuka J, Shimamoto K, Nakamura K (1990) Enhancement of foreign gene expression by a dicot intron in rice but not in tobacco is correlated with an increased level of mRNA and an efficient splicing of the intron. Nucleic Acids Res 18:6767–6770
Usami S, Ohta S, Komari T, Burnell JN (1995) Cold stability of pyruvate, orthophosphate dikinase of Flaveria brownii. Plant Mol Biol 27:969–980
Usuda H (1984) Variations in the photosynthesis rate and activity of photosynthetic enzymes in maize leaf tissue of different ages. Plant Cell Physiol 25:1297–1301
Usuda H, Ku MSB, Edwards GE (1984a) Activation of NADP-malate dehydrogenase, pyruvate, Pi dikinase, and fructose 1,6-bisphosphatase in relation to photosynthetic rate in maize. Plant Physiol 76:238–243
Usuda H, Ku MSB, Edwards GE (1984b) Rates of photosynthesis relative to activity of photosynthetic enzymes, chlorophyll and soluble protein content among ten 4-carbon pathway species. Aust J Plant Physiol 11:509–517
Vasil V, Clancy M, Ferl RJ, Vasil KI, Hannah LC (1989) Increased gene expression by the first intron of maize shrunken-1 locus in grass species. Plant Physiol 91:1575–1579
Wang M-B, Waterhouse PM (1997) A rapid and simple method of assaying plants transformed with hygromycin and PPT resistance genes. Plant Mol Biol Rep 15:209–215
Ward DA (1987) The temperature acclimation of photosynthetic response to CO2 in Zea mays and its relationship to the activities of photosynthetic enzymes and the CO2-concentrating mechanism of C4 photosynthesis. Plant Cell Environ 10:407–411
Acknowledgements
We are grateful to Dr. M. Matsuoka for kindly providing the genomic clone of maize PPDK. We also thank Dr. T. Komari for critical reading of the manuscript, and Ms. M. Tabayashi-Kuramoto, Ms. A.␣Yamashita, and Ms E. Usami for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ohta, S., Ishida, Y. & Usami, S. High-Level Expression of Cold-Tolerant Pyruvate, Orthophosphate Dikinase from a Genomic Clone with Site-Directed Mutations in Transgenic Maize. Mol Breeding 18, 29–38 (2006). https://doi.org/10.1007/s11032-006-9011-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-006-9011-8