Abstract
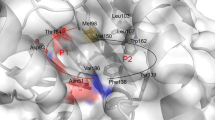
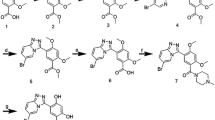
Heat shock protein 90 (HSP90) is a promising anticancer drug target, which could be employed to construct HSP90 inhibitors-based drug conjugates for selective tumor therapy. Herein, a series of 4-(1H-1,2,3-triazol-1-yl)benzamides were rationally designed, synthesized as HSP90 inhibitors, and their structures were characterized by 1H NMR, 13C NMR, and HR-MS. Preliminary HSP90 binding assay showed that compounds 6b, 6l, 6m, 6n, 6t, and 6u exhibited significant HSP90α binding affinity. Among these selected compounds, 6u displayed the most potent anti-proliferative activities and particularly in Capan-1 cell line. Molecular modeling studies also confirmed possible mode of interaction between 6u and the binding sites of HSP90 by hydrogen bond and hydrophobic interactions. Above all, these encouraging data indicated that 6u could be used as a HSP90 inhibitor for further study and helped the recognition of the 4-(1H-1,2,3-triazol-1-yl)benzamide motif as a new scaffold for HSP90 inhibitors.
Graphical abstract






Similar content being viewed by others
References
Lv C, Zhang X, Liu Y, Zhang T, Chen H, Zang J, Zhao G (2021) Redesign of protein nanocages: the way from 0D, 1D, 2D to 3D assembly. Chem Soc Rev 50(6):3957–3989
Bellone ML, Muñoz Camero C, Chini MG, Dal Piaz F, Hernandez V, Bifulco G, Braca A (2021) Limonoids from Guarea guidonia and Cedrela odorata: Heat Shock Protein 90 (Hsp90) Modulator Properties of Chisomicine D. J Nat Prod 84(3):724–737
Serwetnyk MA, Blagg BS (2021) The disruption of protein-protein interactions with co-chaperones and client substrates as a strategy towards Hsp90 inhibition. Acta Pharm Sin B 11(6):1446–1468
Dymock BW, Barril X, Brough PA, Cansfield JE, Massey A, McDonald E, Drysdale MJ (2005) Novel, potent small-molecule inhibitors of the molecular chaperone Hsp90 discovered through structure-based design. J Med Chem 48(13):4212–4215
Lopez A, Dahiya V, Delhommel F, Freiburger L, Stehle R, Asami S, Sattler M (2021) Client binding shifts the populations of dynamic Hsp90 conformations through an allosteric network. Sci Adv 7(51):7295
Walter S, Buchner J (2002) Molecular chaperones—cellular machines for protein folding. Angew Chem Int Ed 41(7):1098–1113
Song J, Herrmann JM, Becker T (2021) Quality control of the mitochondrial proteome. Nat Rev Mol Cell Biol 22(1):54–70
Lin S, Zhang L, Zhang X, Yu Z, Huang X, Xu J, Wu L (2020) Synthesis of novel dual target inhibitors of PARP and HSP90 and their antitumor activities. Bioorg Med Chem 28(9):115434
Nguyen TT, Shang E, Shu C, Kim S, Mela A, Humala N, Siegelin MD (2021) Aurora kinase A inhibition reverses the Warburg effect and elicits unique metabolic vulnerabilities in glioblastoma. Nat Commun 12(1):1–19
Xu M, Zhao C, Zhu B, Wang L, Zhou H, Yan D, Xu J (2021) Discovering high potent Hsp90 inhibitors as antinasopharyngeal carcinoma agents through fragment assembling approach. J Med Chem 64(4):2010–2023
Luo S, Gao X, Ding J, Liu C, Du C, Hou C, Lou B (2019) Transcriptome sequencing reveals the traits of spermatogenesis and testicular development in large yellow croaker (Larimichthys crocea). Genes 10(12):958
Costa TE, Raghavendra NM, Penido C (2020) Natural heat shock protein 90 inhibitors in cancer and inflammation. Eur J Med Chem 189:112063
Ferraro M, D’Annessa I, Moroni E, Morra G, Paladino A, Rinaldi S, Colombo G (2018) Allosteric modulators of HSP90 and HSP70: dynamics meets function through structure-based drug design. J Med Chem 62(1):60–87
Zhu S, Li Y, Huang Y, Zhang M, Gu X, He Y, Lu W (2020) Optimized HSP90 mediated fluorescent probes for cancer-specific bioimaging. J Mater Chem B 8(9):1878–1896
Bridges AJ, Mehta RK, Shukla S, Schipper MJ, Lawrence TS, Nyati MK (2021) Drug-development, dose-selection, rational combinations from bench-to-bedside: are there any lessons worth revisiting. Oncotarget 12(11):1032
Crowe LB, Hughes PF, Alcorta DA, Osada T, Smith AP, Totzke J, Haystead TA (2017) A fluorescent Hsp90 probe demonstrates the unique association between extracellular Hsp90 and malignancy in vivo. ACS Chem Biol 12(4):1047–1055
Proia DA, Smith DL, Zhang J, Jimenez JP, Sang J, Ogawa LS, Ying W (2015) HSP90 Inhibitor–SN-38 Conjugate strategy for targeted delivery of topoisomerase I inhibitor to tumors. Mol Cancer Ther 14(11):2422–2432
Bobrov E, Skobeleva N, Restifo D, Beglyarova N, Cai KQ, Handorf E, Astsaturov I (2017) Targeted delivery of chemotherapy using HSP90 inhibitor drug conjugates is highly active against pancreatic cancer models. Oncotarget 8(3):4399
Coats S, Williams M, Kebble B, Dixit R, Tseng L, Yao NS, Soria JC (2019) Antibody–drug conjugates: future directions in clinical and translational strategies to improve the therapeutic index. Clin Cancer Res 25(18):5441–5448
Zhu S, Lu Y, Jin J, Yu J, Lu W (2021) An HSP90 inhibitor based fluorescent probe for selective tumor targeting. Dyes Pigm 196:109783
Zhu S, Shen Q, Gao Y, Wang L, Fang Y, Chen Y, Lu W (2020) Design, synthesis, and biological evaluation of HSP90 inhibitor–SN38 conjugates for targeted drug accumulation. J Med Chem 63(10):5421–5441
Taddei M, Ferrini S, Giannotti L, Corsi M, Manetti F, Giannini G, Cabri W (2014) Synthesis and evaluation of new Hsp90 inhibitors based on a 1, 4, 5-trisubstituted 1, 2, 3-triazole scaffold. J Med Chem 57(6):2258–2274
Peterson LB, Blagg BS (2010) Click chemistry to probe Hsp90: synthesis and evaluation of a series of triazole-containing novobiocin analogues. Bioorg Med Chem Lett 20(13):3957–3960
Terracciano S, Chini MG, Dal Piaz F, Vassallo A, Riccio R, Bruno I, Bifulco G (2013) Dimeric and trimeric triazole based molecules as a new class of Hsp90 molecular chaperone inhibitors. Eur J Med Chem 65:464–476
Park SY, Oh YJ, Lho Y, Jeong JH, Liu KH, Song J, Seo YH (2018) Design, synthesis, and biological evaluation of a series of resorcinol-based N-benzyl benzamide derivatives as potent Hsp90 inhibitors. Eur J Med Chem 143:390–401
Liu YM, Tu HJ, Wu CH, Lai MJ, Yu SC, Chao MW, Liou JP (2021) Ring-opening of five-membered heterocycles conjugated 4-isopropylresorcinol scaffold-based benzamides as HSP90 inhibitors suppressing tumor growth in vitro and in vivo. Eur J Med Chem 219:113428
Uno T, Kawai Y, Yamashita S, Oshiumi H, Yoshimura C, Mizutani T, Kitade M (2018) Discovery of 3-Ethyl-4-(3-isopropyl-4-(4-(1-methyl-1 H-pyrazol-4-yl)-1 H-imidazol-1-yl)-1 H-pyrazolo [3,4-b] pyridin-1-yl) benzamide (TAS-116) as a potent, selective, and orally available HSP90 inhibitor. J Med Chem 62(2):531–551
Xu XL, Bao QC, Jia JM, Liu F, Guo XK, Zhang MY, Sun HP (2016) CPUY201112, a novel synthetic small-molecule compound and inhibitor of heat shock protein Hsp90, induces p53-mediated apoptosis in MCF-7 cells. Sci Rep 6(1):1–16
Jiang F, Guo AP, Xu JC, You QD, Xu XL (2018) Discovery of a Potent Grp94 Selective inhibitor with anti-inflammatory efficacy in a mouse model of ulcerative colitis. J Med Chem 61(21):9513–9533
Acknowledgements
This research was funded by the Fundamental Research Funds for the Central Universities and the National Natural Science Foundation of China, Grant Numbers 22077034 and 82104000.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, T., Zhu, S. & Lu, W. Design, synthesis, and biological evaluation of 4-(1H-1,2,3-triazol-1-yl)benzamides as HSP90 inhibitors. Mol Divers 27, 239–248 (2023). https://doi.org/10.1007/s11030-022-10423-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-022-10423-7