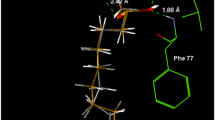
Abstract
Vernonia amygdalina, a widely consumed West African food herb, can be a boon in the discovery of safe anti-obesity agents given the extensive reports on its anti-obesity and antidiabetic potentials. The main aim of this study was to screen 78 Vernonia—Derived Phytocompounds (VDPs) against the active site regions of Human Pancreatic Lipase (HPL), Human Pancreatic Amylase and Human Glucosidase (HG) as drug targets associated with obesity in silico. Structure-based virtual screening helped to identify Luteolin 7-O-glucuronoside and Andrographidoid D2 as hit compounds with dual targeting tendency towards the HPL and HG. Analysis of the molecular dynamic simulation trajectory files of the ligand-receptor complexes as computed from the thermodynamic parameters plots showed not only increased flexibility and greater interaction potential of the active site residues of the receptor towards the VDPs as indicated by the root mean square fluctuation but also higher stability as indicated by the root mean square deviation, radius of gyration and number of hydrogen bonds. The cluster analysis further showed that the interactions with important residues were preserved in the dynamic environment. These observations were further verified from Molecular Mechanics Generalized Born Surface Area Analysis, which also showed that residual contributions to the binding free energies were mainly from catalytic residues at the active sites of the enzymes. The hit compounds also feature desirable physicochemical properties and drug-likeness. This study provides in silico evidence for the inhibitory potential of phytochemicals from Vernonia amygdalina against two target enzymes in obesity.
Graphical abstract















Similar content being viewed by others
References
Balaji M, Ganjayi MS, Hanuma Kumar GE, Parim BN, Mopuri R, Dasari S (2016) A review on possible therapeutic targets to contain obesity: the role of phytochemicals. Obes Res Clin Pract 10(4):363–80. https://doi.org/10.1016/j.orcp.2015.12.004
Adeloye D, Ige-Elegbede JO, Ezejimofor M, Owolabi EO, Ezeigwe N, Omoyele C, Mpazanje RG, Dewan MT, Agogo E, Gadanya MA, Alemu W, Harhay MO, Auta A, Adebiyi AO (2021) Estimating the prevalence of overweight and obesity in Nigeria in 2020: a systematic review and meta-analysis. Ann Med 53(1):495–507. https://doi.org/10.1080/07853890.2021.1897665
Forouzanfar MH, Alexander L, Anderson HR, Bachman VF et al (2015) Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 386(10010):2287–2323. https://doi.org/10.1016/S0140-6736(15)00128-2
Lillich FF, Imig JD, Proschak E (2021) Multi-target approaches in metabolic syndrome. Front Pharmacol. https://doi.org/10.3389/fphar.2020.554961
Ford ND, Patel SA, Narayan KM (2017) Obesity in low- and middle-income countries: burden, drivers, and emerging challenges. Annu Rev Public Health 38:145–164. https://doi.org/10.1146/annurev-publhealth-031816-044604
Steyn NP, McHiza ZJ (2014) Obesity and the nutrition transition in Sub-Saharan Africa. Ann N Y Acad Sci 1311:88–101. https://doi.org/10.1111/nyas.12433
Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG (2014) 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American heart association task force on practice guidelines and the obesity society. Circulation 129(25 Suppl 2):S102–S138
Harvie M, Howell A (2017) Potential benefits and harms of intermittent energy restriction and intermittent fasting amongst obese, overweight and normal weight subjects-A narrative review of human and animal evidence. Behav Sci (Basel). https://doi.org/10.3390/bs7010004
Qi X (2018) Review of the clinical effect of orlistat. IOP Conf Series Mater Sci Eng 301:012063
Luo S, Gill H, Dias DA, Li M, Hung A, Nguyen LT, Lenon GB (2019) The inhibitory effects of an eight-herb formula (RCM-107) on pancreatic lipase: enzymatic, HPTLC profiling and in silico approaches. Heliyon 5(9):e02453. https://doi.org/10.1016/j.heliyon.2019.e02453
Lankatillake C, Luo S, Flavel M, Lenon GB, Gill H, Huynh T, Dias DA (2021) Screening natural product extracts for potential enzyme inhibitors: protocols, and the standardisation of the usage of blanks in α-amylase, α-glucosidase and lipase assays. Plant Methods 17(1):3. https://doi.org/10.1186/s13007-020-00702-5
Copeland RA, Harpel MR, Tummino PJ (2007) Targeting enzyme inhibitors in drug discovery. Expert Opin Ther Targets 11(7):967–78. https://doi.org/10.1517/14728222.11.7.967
Shi Y, Burn P (2004) Lipid metabolic enzymes: emerging drug targets for the treatment of obesity. Nat Rev Drug Discovery 3(8):695–710
Winkler FK, D’Arcy A, Hunziker W (1990) Structure of human pancreatic lipase. Nature 343(6260):771–4. https://doi.org/10.1038/343771a0
Lüthi-Peng Q, Märki HP, Hadváry P (1992) Identification of the active-site serine in human pancreatic lipase by chemical modification with tetrahydrolipstatin. FEBS Lett 299(1):111–5. https://doi.org/10.1016/0014-5793(92)80112-t
Cygler M, Schrag JD, Ergan F (1992) Advances in structural understanding of lipases. Biotechnol Genet Eng Rev 10:143–84. https://doi.org/10.1080/02648725.1992.10647887
Egloff M-P, Marguet F, Buono G, Verger R, Cambillau C, van Tilbeurgh H (1995) The 2.46. ANG. resolution structure of the pancreatic lipase-colipase complex inhibited by a C11 alkyl phosphonate. Biochemistry 34(9):2751–2762. https://doi.org/10.1021/bi00009a003
Bourne Y, Martinez C, Kerfelec B, Lombardo D, Chapus C, Cambillau C (1994) Horse pancreatic lipase. The crystal structure refined at 2.3 A resolution. J Mol Biol 238(5):709–32
Nguyen PTV, Huynh HA, Truong DV, Tran TD, Vo CT (2020) Exploring aurone derivatives as potential human pancreatic lipase inhibitors through molecular docking and molecular dynamics simulations. Molecules. https://doi.org/10.3390/molecules25204657
Kumar A, Chauhan S (2021) Pancreatic lipase inhibitors: the road voyaged and successes. Life Sci 271:119115. https://doi.org/10.1016/j.lfs.2021.119115
Ogunyemi OM, Gyebi AG, Adebayo JO, Oguntola JA, Olaiya CO (2020) Marsectohexol and other pregnane phytochemicals derived from Gongronema latifolium as α-amylase and α-glucosidase inhibitors: in vitro and molecular docking studies. SN Appl Sci 2(12):2119. https://doi.org/10.1007/s42452-020-03951-0
Nahoum V, Roux G, Anton V, Rougé P, Puigserver A, Bischoff H, Henrissat B, Payan F (2000) Crystal structures of human pancreatic alpha-amylase in complex with carbohydrate and proteinaceous inhibitors. Biochem J 346(Pt 1):201–208
Rydberg EH, Li C, Maurus R, Overall CM, Brayer GD, Withers SG (2002) Mechanistic analyses of catalysis in human pancreatic alpha-amylase: detailed kinetic and structural studies of mutants of three conserved carboxylic acids. Biochemistry 41(13):4492–502
Dhital S, Lin AH, Hamaker BR, Gidley MJ, Muniandy A (2013) Mammalian mucosal alpha-glucosidases coordinate with alpha-amylase in the initial starch hydrolysis stage to have a role in starch digestion beyond glucogenesis. PLoS One 8(4):e62546
Nyambe-Silavwe H, Villa-Rodriguez JA, Ifie I, Holmes M, Aydin E, Jensen JM, Williamson G (2015) Inhibition of human α-amylase by dietary polyphenols. J Funct Foods 19:723–732
Atangwho IJ, Ebong PE, Eyong EU, Asmawi MZ, Ahmad M (2012) Synergistic antidiabetic activity of Vernonia amygdalina and Azadirachta indica: biochemical effects and possible mechanism. J Ethnopharmacol 141(3):878–887. https://doi.org/10.1016/j.jep.2012.03.041
Atangwho IJ, Edet EE, Uti DE, Obi AU, Asmawi MZ, Ahmad M (2012) Biochemical and histological impact of Vernonia amygdalina supplemented diet in obese rats. Saudi J Biol Sci 19(3):385–392. https://doi.org/10.1016/j.sjbs.2012.05.003
Wang J, Song H, Wu X, Zhang S, Gao X, Li F, Zhu X, Chen Q (2018) Steroidal saponins from vernonia amygdalina del and their biological activity. Mole A J Synthet Chem Nat Prod Chem 23(3):579. https://doi.org/10.3390/molecules23030579
Erukainure OL, Chukwuma CI, Sanni O, Matsabisa MG, Islam MS (2019) Histochemistry, phenolic content, antioxidant, and anti-diabetic activities of Vernonia amygdalina leaf extract. J Food Biochem 43(2):e12737. https://doi.org/10.1111/jfbc.12737
Adaramoye O, Ogungbenro B, Anyaegbu O, Fafunso M (2008) Protective effects of extracts of Vernonia amygdalina, Hibiscus sabdariffa and vitamin C against radiation-induced liver damage in rats. J Radiat Res 49(2):123–31. https://doi.org/10.1269/jrr.07062
Olaiya CO, Choudhary MI, Ogunyemi OM, Nwauzoma AB (2013) Nutraceuticals from bitter leaf (Vernonia amygdalina Del.) protects against cadmium chloride induced hypertension in albino rats. Nat Sci 11(6):136–145
Ch’ng YS, Loh YC, Tan CS, Ahmad M, Asmawi MZ, Wan Omar WM, Yam MF (2017) Vasorelaxant properties of Vernonia amygdalina ethanol extract and its possible mechanism. Pharm Biol 55(1):2083–2094. https://doi.org/10.1080/13880209.2017.1357735
Momoh MA, Muhamed U, Agboke AA, Akpabio EI, Osonwa UE (2012) Immunological effect of aqueous extract of Vernonia amygdalina and a known immune booster called immunace(®) and their admixtures on HIV/AIDS clients: a comparative study. Asian Pacific J Tropic Biomed 2(3):181–184. https://doi.org/10.1016/S2221-1691(12)60038-0
Erasto P, Grierson DS, Afolayan AJ (2007) Antioxidant constituents in vernonia amygdalina. Leaves. Pharm Biol 45(3):195–199
Igile GO, Oleszek W, Jurzysta M, Burda S, Fafunso M, Fasanmade AA (1994) Flavonoids from Vernonia amygdalina and their antioxidant activities. J Agric Food Chem 42(11):2445–2448. https://doi.org/10.1021/jf00047a015
Sinisi A, Millán E, Abay SM, Habluetzel A, Appendino G, Muñoz E, Taglialatela-Scafati O (2015) Poly-electrophilic sesquiterpene lactones from vernonia amygdalina: new members and differences in their mechanism of thiol trapping and in bioactivity. J Nat Prod 78(7):1618–23
Jisaka M, Ohigashi H, Takagaki T, Nozaki H, Tada T, Hirota M, Irie R, Huffman MA, Nishida T, Kaji M, Koshimizu K (1992) Bitter steroid glucosides, vernoniosides A1, A2, and A3, and related B1 from a possible medicinal plant, Vernonia amygdalina, used by wild chimpanzees. Tetrahedron 48(4):625–632. https://doi.org/10.1016/S0040-4020(01)88123-0
Igile G, Olenszek W, Jurzysta M, Aquino R, de Tommasi N, Pizza C (1995) Vemoniosides D and E, two novel saponins from vernonia amygdalina. J Nat Prod 58(9):1438–1443
Quasie O, Zhang Y-M, Zhang H-J, Luo J, Kong L-Y (2016) Four new steroid saponins with highly oxidized side chains from the leaves of Vernonia amygdalina. Phytochem Lett 15:16–20
Erasto P, Grierson DS, Afolayan AJ (2007) Evaluation of antioxidant activity and the fatty acid profile of the leaves of Vernonia amygdalina growing in South Africa. Food Chem 104(2):636–642
Oyeyemi IT, Akinlabi AA, Adewumi A, Aleshinloye AO, Oyeyemi OT (2018) Vernonia amygdalina: a folkloric herb with anthelminthic properties. Beni-Suef Univ J Basic Appl Sci 7(1):43–49
Alara OR, Abdurahman NH, Ukaegbu CI, Kabbashi NA (2019) Extraction and characterization of bioactive compounds in Vernonia amygdalina leaf ethanolic extract comparing Soxhlet and microwave-assisted extraction techniques. J Taibah Univ Sci 13(1):414–422
Sales PM, Souza PM, Simeoni LA, Silveira D (2012) Alpha-Amylase inhibitors: a review of raw material and isolated compounds from plant source. J Pharm Pharm Sci 15(1):141–83
Saini P, Sharma A, Tuli HS, Kashyap D, Kumar Mishra N, Debnath P (2016) In silico comparative analysis of natural metabolites to alpha glucosidase inhibitors. J Biol Chem Sci 3:226–232
Uzunova VV, Quareshy M, Del Genio CI, Napier RM (2016) Tomographic docking suggests the mechanism of auxin receptor TIR1 selectivity. Open Biol 6(10):160139
Stark JL, Powers R (2012) Application of NMR and molecular docking in structure-based drug discovery. Top Curr Chem 326:1–34. https://doi.org/10.1007/128_2011_213
Xie ZR, Hwang MJ (2015) Methods for predicting protein-ligand binding sites. Methods Mol Biol 1215:383–398
O’Boyle NM, Banck M, James CA, Morley C, Vandermeersch T, Hutchison GR (2011) Open babel: an open chemical toolbox. J Cheminform 3:33
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31(2):455–461
Jo S, Kim T, Iyer VG, Im WJJ et al (2008) CHARMM-GUI: a web-based graphical user interface for CHARMM. J Comput Chem 29(11):1859–1865
Brooks BR, Brooks CL III, Mackerell AD Jr, Nilsson L, Petrella RJ, Roux B, Won Y, Archontis G, Bartels C, Boresch SJ (2009) CHARMM: the biomolecular simulation program. J Comput Chem 30(10):1545–1614. https://doi.org/10.1002/jcc.21287
Lee J, Cheng X, Swails JM, Yeom MS, Eastman PK, Lemkul JA, Wei S, Buckner J, Jeong JC, Qi YJ (2016) computation, CHARMM-GUI input generator for NAMD, GROMACS, AMBER, OpenMM, and CHARMM/OpenMM simulations using the CHARMM36 additive force field. J Chem Theory Comput 12(1):405–413. https://doi.org/10.1021/acs.jctc.5b00935
Miller BR III, McGee TD Jr, Swails JM, Homeyer N, Gohlke H, Roitberg AE (2012) MMPBSA. py: an efficient program for end-state free energy calculations. J Chem Theory Comput 8(9):3314–3321. https://doi.org/10.1021/ct300418h
Case DSC DA, Cheatham TE III, Darden TA, Duke RE, Giese TJ, Gohlke H, Goetz AWD, Greene NH, Izadi S, Kovalenko A, Lee TS, LeGrand S, Li P, Lin C, Liu J, Luchko T, Luo R, Mermelstein D KMM, Monard G, Nguyen H, Omelyan I, Onufriev A, Pan F, Qi R, Roe DR, Roitberg A CS, Simmerling CL, Botello-Smith WM, Swails J, Walker RC, Wang J, Wolf RM, Wu X LX, York DM, Kollman PA, AMBER 2017, University of California, San Francisco. 2017
Daina A, Michielin O, Zoete V (2017) SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep 7:42717
Banerjee P, Eckert AO, Schrey AK, Preissner R (2018) ProTox-II: a webserver for the prediction of toxicity of chemicals. Nucleic Acids Res 46(W1):W257-w263
Anh HLT, Vinh LB, Lien LT, Cuong PV, Arai M, Ha TP, Lin HN, Dat TTH, Cuong LCV, Kim YH (2021) In vitro study on α-amylase and α-glucosidase inhibitory activities of a new stigmastane-type steroid saponin from the leaves of Vernonia amygdalina. Nat Prod Res 35(5):873–879
Suresh PS, Singh PP, Padwad YS, Sharma U (2021) Steroidal saponins from Trillium govanianum as α-amylase, α-glucosidase, and dipeptidyl peptidase IV inhibitory agents. J Pharm Pharmacol 73(4):487–495
Marrelli M, Conforti F, Araniti F, Statti GA (2016) Effects of saponins on lipid metabolism: a review of potential health benefits in the treatment of obesity. Mole (Basel, Switzerland) 21(10):1404
Maqsood M, Ahmed D, Atique I, Malik W (2017) Lipase inhibitory activity of Lagenaria siceraria fruit as a strategy to treat obesity. Asian Pacific J Tropic Med 10(3):305–310
Jeepipalli SPK, Du B, Sabitaliyevich UY, Xu B (2020) New insights into potential nutritional effects of dietary saponins in protecting against the development of obesity. Food Chemistry 318:126474
Roig-Zamboni V, Cobucci-Ponzano B, Iacono R, Ferrara MC, Germany S, Bourne Y, Parenti G, Moracci M, Sulzenbacher G (2017) Structure of human lysosomal acid α-glucosidase–a guide for the treatment of Pompe disease. Nat Commun 8(1):1111
Hughes JD, Blagg J, Price DA, Bailey S, DeCrescenzo GA, Devraj RV, Ellsworth E, Fobian YM, Gibbs ME, Gilles RW (2008) Physiochemical drug properties associated with in vivo toxicological outcomes. Bioorgan Med Chem Lett 18(17):4872–4875
Lipinski C, Lombardo F, Dominy BW, Feeney PJ (2001) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev 46(1–3):3
Veber DF, Johnson SR, Cheng H-Y, Smith BR, Ward KW, Kopple KD (2002) Molecular properties that influence the oral bioavailability of drug candidates. J Med Chem 45(12):2615–2623
Ghose AK, Viswanadhan VN, Wendoloski JJ (1999) A knowledge-based approach in designing combinatorial or medicinal chemistry libraries for drug discovery: a qualitative and quantitative characterization of known drug databases. J Comb Chem 1(1):55–68
Egan WJ, Merz KM, Baldwin JJ (2000) Prediction of drug absorption using multivariate statistics. J Med Chem 43(21):3867–3877
Martin YC (2005) A bioavailability score. J Med Chem 48(9):3164–3170
Lin JH, Yamazaki M (2003) Role of P-glycoprotein in pharmacokinetics. Clin Pharmacokinet 42(1):59–98
Kratz JM, Grienke U, Scheel O, Mann SA, Rollinger JM (2017) Natural products modulating the hERG channel: heartaches and hope. Nat Prod Rep 34(8):957–980
Raschi E, Vasina V, Poluzzi E, De Ponti F (2008) The hERG K+ channel: target and antitarget strategies in drug development. Pharmacol Res 57(3):181–195
Shen J, Cheng F, Xu Y, Li W, Tang Y (2010) Estimation of ADME properties with substructure pattern recognition. J Chem Inf Model 50(6):1034–1041
Acknowledgements
The authors acknowledge with thanks resources offered by the PhytoBioNet platform. The MDS and MM-GBSA calculations are done on the Bibliotheca Alexandrina HPC facility, Alexandria, Egypt. We appreciate and thank Taif University for the financial support for Taif University Researchers Supporting Project (TURSP-2020/09), Taif University, Taif, Saudi Arabia.
Funding
This study was supported by the Taif University Researchers Supporting Project (TURSP-2020/09), Taif University, Taif, Saudi Arabia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest is reported by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ogunyemi, O.M., Gyebi, G.A., Ibrahim, I.M. et al. Identification of promising multi-targeting inhibitors of obesity from Vernonia amygdalina through computational analysis. Mol Divers 27, 1–25 (2023). https://doi.org/10.1007/s11030-022-10397-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-022-10397-6