Abstract
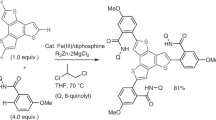
A novel, simple and efficient protocol for the Fe-catalyzed, intermolecular cross-dehydrogenative coupling of pyrimidine bearing 4-thiazolidinones systems and terminal alkynes was established. This methodology offers a high yielding, straightforward, and one-pot approach towards the synthesis of alkynylated 4-thiazolidinones without prior activation of C(sp3)–H and C(sp)–H. The results of control experiments indicated that this conversion might proceed via a radical process.
Graphical abstract





Similar content being viewed by others
References
Verma A, Saraf SK (2008) 4-Thiazolidinone–a biologically active scaffold. Eur J Med Chem 43(5):897–905
Tripathi AC, Gupta S, Fatima GN, Sonar PK, Verma A, Saraf SK (2014) 4-Thiazolidinones: the advances continue…. Eur J Med Chem 72:52–77
Jain AK, Vaidya A, Ravichandran V, Kashaw SK, Agrawal RK (2012) Recent developments and biological activities of thiazolidinone derivatives: a review. Bioorg Med Chem 20(11):3378–3395
Momose Y, Meguro K, Ikeda H, Hatanaka C, Oi S, Sohda T (1991) Studies on antidiabetic agents. X. Synthesis and biological activities of pioglitazone and related compounds. Chem Pharma Bull. 39(6):1440–1445
Cheng P, Guo W, Chen P, Liu Y, Du X, Li C (2016) The enantioselective construction of chiral spirooxindole-based 4-thiazolidinone via asymmetric catalytic formal [3+ 2] annulation using a bifunctional catalyst. Chem Comm 52(16):3418–3421
Mehdi RBA, Shaaban KA, Rebai IK, Smaoui S, Bejar S, Mellouli L (2009) Five naturally bioactive molecules including two rhamnopyranoside derivatives isolated from the Streptomyces sp. strain TN58. Nat Prod Res 23(12):1095–1107
Fischer W, Bodewei R, Satzinger G (1992) Anticonvulsant and sodium channel blocking effects of ralitoline in different screening models. Naunyn-Schmiedeb Arch Pharmacol 346(4):442–452
Sharples KR, Hawkes TR, Mitchell G, Edwards LS, Langford MP, Langton DW, Rogers KM, Townson JK, Wang Y (1998) A novel thiazolidinone herbicide is a potent inhibitor of glucose incorporation into cell wall material. Pesti Sci 54(4):368–376
Trost BM, Li CJ (eds) (2015) Modern alkyne chemistry: catalytic and atom-economic transformations. Wiley
Cera G, Haven T, Ackermann L (2017) Iron-catalyzed C−H alkynylation through triazole assistance: expedient access to bioactive heterocycles. Chem Eur J 23(15):3577–3582
Brand JP, Waser J (2012) Electrophilic alkynylation: the dark side of acetylene chemistry. Chem Soc Rev 41(11):4165–4179
Amir M, Javed SA, Kumar H (2007) Pyrimidine as antiinflammatory agent: a review. Ind J Pharm Sci 69(3):337
Selvam TP, James CR, Dniandev PV, Valzita SK (2015) A mini review of pyrimidine and fused pyrimidine marketed drugs. Res Pharm 2(4):1–9
Bhat AR, Dongre RS, Naikoo GA, Hassan IU, Ara T (2017) Proficient synthesis of bioactive annulated pyrimidine derivatives: a review. J Taibah Univ Sci 11(6):1047–1069
Watson JD, Crick FHC (1953) Molecular structure of nucleic acids: a structure for deoxyribose nucleic acid. Nature 171(4356):737–738
Bérubé G (2016) An overview of molecular hybrids in drug discovery. Expert Opin Drug Discov 11(3):281–305
Meunier B (2008) Hybrid molecules with a dual mode of action: dream or reality? Acc Chem Res 41(1):69–77
Jung ME, Ku JM, Du L, Hu H, Gatti RA (2011) Synthesis and evaluation of compounds that induce readthrough of premature termination codons. Bioorg Med Chem Lett 21(19):5842–5848
Gopalakrishnan M, Thanusu J, Kanagarajan V (2009) Design, synthesis, spectral analysis and in vitro microbiological evaluation of 2-phenyl-3-(4, 6-diarylpyrimidin-2-yl) thiazolidin-4-ones. J Enzyme Inhib Med Chem 24(5):1088–1094
Patel JJ, Morja MI, Chikhalia KH (2020) An efficient synthesis of designed 4-thiazolidinone fused pyrimidine derivatives as potent antimicrobial agents. J Heterocycl Chem 57(10):3531–3543
Magano J, Dunetz JR (2011) Large-scale applications of transition metal-catalyzed couplings for the synthesis of pharmaceuticals. Chem Rev 111(3):2177–2250
Beletskaya IP, Ananikov VP (2011) Transition-metal-catalyzed C−S, C−Se, and C−Te bond formation via cross-coupling and atom-economic addition reactions. Chem Rev 111(3):1596–1636
Chen PH, Billett BA, Tsukamoto T, Dong G (2017) Cut and sew” transformations via transition-metal-catalyzed carbon–carbon bond activation. ACS Catal 7(2):1340–1360
Chandra D, Dhiman AK, Parmar D, Sharma U (2020) Alkylation, alkenylation, and alkynylation of heterocyclic compounds through group 9 (Co, Rh, Ir) metal-catalyzed CH activation. Catal Rev. https://doi.org/10.1080/01614940.2020.1839849
Hu L, Dietl MC, Han C, Rudolph M, Rominger F, Hashmi ASK (2021) Au–Ag bimetallic catalysis: 3-alkynyl benzofurans from phenols via tandem C−H alkynylation/oxy-alkynylation. Angew Chem Int Ed 60(19):10637–10642
Shaikh MUM, Mudaliar SS, Chikhalia KH (2016) An efficient alkynylation of 4-thiazolidinone with terminal alkyne under C–H functionalisation. RSC Adv 6(56):50780–50785
Shaikh MM, Patel AP, Chikhalia KH (2019) Copper catalysed cross-dehydrogenative coupling (CDC) reaction of 4-thiazolidinone with terminal alkyne. Tetrahedron 75(4):475–485
Patel A, Patel J, Chikhalia KH (2017) Palladium-catalyzed acid-free Fujiwara-Moritani alkenylation of 4-thiazolidinones. Mol Divers 21(4):1011–1020
Chauhan PM, Morja MI, Asamdi M, Chikhalia KH (2020) Copper catalyzed cyanomethylation reaction of 4-thiazolidinone. Tetrahedron Lett 61(50):152601
Morja MI, Patel JJ, Chauhan PM, Chikhalia KH (2020) An efficient synthesis of strained thio-bridged compounds via Pd (0) catalyzed intramolecular Csp2 (aryl)-Csp3 (alkyl) cross dehydrohalogenative coupling reaction. Tetrahedron 76(34):131348
Morja MI, Chauhan PM, Chikhalia KH (2021) Palladium-catalyzed novel C (formyl)-C (aryl)/C (aryl)-N (amine) coupling sequence between 2-(methylamino) nicotinaldehyde and 3-bromo-2-chlorothiophene: an efficient construction of 4-pyridone fused hybrid scaffolds. ChemistrySelect 6(20):5014–5020
Morja MI, Chauhan PM, Chikhalia KH (2021) Iron catalyzed alkynylation of thiohydantoins with terminal alkyne via cross-dehydrogenative coupling (CDC). Tetrahedron Lett 77:153148
Gao J, Song QW, He LN, Yang ZZ, Dou XY (2012) Efficient iron (iii)-catalyzed three-component coupling reaction of alkynes, CH2Cl2 and amines to propargylamines. Chem Commun 48(14):2024–2026
Volla CM, Vogel P (2009) Chemoselective C−H bond activation: ligand and solvent free iron-catalyzed oxidative C−C cross-coupling of tertiary amines with terminal alkynes. React Scope Mech Org Lett 11(8):1701–1704
Patil SS, Jadhav RP, Patil SV, Bobade VD (2011) Ligand and solvent-free iron catalyzed oxidative alkynylation of azoles with terminal alkynes. Tetrahedron Lett 52(43):5617–5619
Acknowledgements
Authors are thankful to IISER-Pune and IIT-Bombay for the spectral analysis. Authors express their gratitude to Head of Chemistry Department, Veer Narmad South Gujarat University, Surat, to provide necessary laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Morja, M.I., Chikhalia, K.H. Iron-catalyzed intermolecular cross-dehydrogenative C(sp3)–H/C(sp)–H coupling of pyrimidine bearing 4-thiazolidinones with terminal alkynes. Mol Divers 26, 3037–3045 (2022). https://doi.org/10.1007/s11030-021-10363-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-021-10363-8