Abstract
Abstract
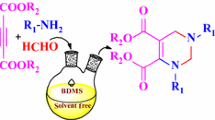
Efficient one-pot three-component reactions of pyrazolones with arylaldehydes and triethyl phosphite were carried out in the presence of ethylenediammonium diacetate as catalyst to synthesize biologically interesting pyrazolylphosphonate derivatives. This methodology offers several significant advantages such as environmentally benign character, the use of a mild catalyst, high yields, and ease of handling. The synthesized compounds were screened for their antioxidant and antibacterial activities. The result showed that compound 4d \((\hbox {IC}_{50 }= 3.40\,\upmu \hbox {M})\) exhibited a strong free radical scavenger toward DPPH free radicals compared with standard BHT \((\hbox {IC}_{50 } = 29.23\,\upmu \hbox {M})\). In addition, compounds 4e and 4p showed potent antibacterial activities against Gram-negative bacteria of E. coli and compound 4o exhibited a potent activity against Gram-positive bacteria of S. aureus compared with standard Ampicillin.
Graphical Abstract




Similar content being viewed by others
References
Moonen K, Lauryn I, Stevens CV (2004) Synthetic methods for azaheterocyclic phosphonates and their biological activity. Chem Rev 104:6177–6215. doi:10.1021/cr030451c
Schug KA, Lindner W (2005) Noncovalent binding between guanidinium and anionic groups: focus on biological- and synthetic-based arginine/guanidinium interactions with phosph[on]ate and sulf[on]ate residues. Chem Rev 105:67–113. doi:10.1021/cr040603j
Allen MC, Fuhrer W, Tuck B, Wade R, Wood JM (1989) Renin inhibitors. Synthesis of transition-state analogue inhibitors containing phosphorus acid derivatives at the scissile bond. J Med Chem 32:1652–1661. doi:10.1021/jm00127a041
Patel DV, Rielly-Gauvin K, Ryono DE (1990) Preparation of peptidic \(\upalpha \)-hydroxy phosphonates a new class of transition state analog renin inhibitors. Tetrahedron Lett 31:5587–5590. doi:10.1016/S0040-4039(00)97903-6
Stowasser B, Budt K-H, Jian-Qi L, Peyman A, Ruppert D (1992) New hybrid transition state analog inhibitors of HIV protease with peripheric C2-symmetry. Tetrahedron Lett 33:6625–6628. doi:10.1016/S0040-4039(00)61002-X
Kafarski P, Lejczak B (1991) Biological activity of aminophosphonic acids. Phosphorus Sulfur Silicon Relat Elem 63:193–215. doi:10.1080/10426509108029443
Baylis EK, Campbell CD, Dingwall JG (1984) 1-Aminoalkylphosphonous acids. Part 1. Isosteres of the protein amino acids. J Chem Soc Perkin Trans 1:2845–2853. doi:10.1039/P19840002845
Atherton FR, Hassall CH, Lambert RW (1986) Synthesis and structure–activity relationships of antibacterial phosphonopeptides incorporating (l-aminoethy1)phosphonic acid and (aminomethy1)phosphonic acid. J Med Chem 29:29–40. doi:10.1021/jm00151a005
Maryanoff BE, Reitz AB (1989) The Wittig olefination reaction and modifications involving phosphoryl-stabilized carbanions. Stereochemistry, mechanism, and selected synthetic aspects. Chem Rev 89:863–927. doi:10.1021/cr00094a007
Pudovik AN, Konovalova IV (1979) Addition reactions of esters of phosphorus(III) acids with unsaturated systems. Synthesis 2:81–96. doi:10.1055/s-1979-28566
Simoni D, Invidiata FP, Manferdini M, Lampronti I, Rondanin R, Roberti M, Pollini GP (1998) Tetramethylguanidine (TMG)-catalyzed addition of dialkyl phosphites to \(\upalpha \),\(\upbeta \)-unsaturated carbonyl compounds, alkenenitriles, aldehydes, ketones and imines. Tetrahedron Lett 39:7615–7618. doi:10.1016/S0040-4039(98)01656-6
Hosseini-Sarvari M, Etemad S (2008) Nanosized zinc oxide as a catalyst for the rapid and green synthesis of \(\upbeta \)-phosphono malonates. Tetrahedron 64:5519–5523. doi:10.1016/j.tet.2008.03.095
Green K (1989) Trimethylaluminum promoted conjugate additions of dimethyl phosphite to \(\upalpha \),\(\upbeta \)-unsaturated esters and ketones. Tetrahedron Lett 30:4807–4810. doi:10.1016/S0040-4039(01)80513-X
Hindersinn RR, Ludington RS (1965) The reaction of trialkyl phosphites with maleate esters. J Org Chem 30:4020–4025. doi:10.1021/jo01023a008
Stockland RA Jr, Taylor RI, Thompson LE, Patel PB (2005) Microwave-assisted regioselective addition of P(O)-H bonds to alkenes without added solvent or catalyst. Org Lett 7:851–853. doi:10.1021/ol0474047
Shulyupin MO, Kazankova MA, Beletskaya IP (2002) Catalytic hydrophosphination of styrenes. Org Lett 4:761–763. doi:10.1021/ol017238s
Xu Q, Han L-B (2006) Palladium-catalyzed asymmetric hydrophosphorylation of norbornenes. Org Lett 8:2099–2101. doi:10.1021/ol060568y
Semenzin D, Etemad-Moghadam G, Albouy D, Diallo O, Koenig M (1997) Dual radical/polar pudovik reaction: application field of new activation methods. J Org Chem 62:2414–2422. doi:10.1021/jo9622441
Han L-B, Zhao C-Q (2005) Stereospecific addition of H-P bond to alkenes: a simple method for the preparation of (RP)-phenylphosphinates. J Org Chem 70:10121–10123. doi:10.1021/jo051582b
Kolla SR, Lee YR (2012) Efficient one-pot synthesis of \(\upbeta \)-phosphono malonates and 2-amino-4\(H\)-chromen-4-ylphosphonate derivatives by ethylenediamine diacetate-catalyzed three-component reactions. Tetrahedron 68:226–237. doi:10.1016/j.tet.2011.10.060
Lin R, Chiu G, Yu Y, Connolly PJ, Li S, Lu Y, Adams M, Fuentes-Pesquera AR, Emanuel SL, Greenberger LM (2007) Design, synthesis, and evaluation of 3,4-disubstituted pyrazole analogues as anti-tumor CDK inhibitors. Bioorg Med Chem Lett 17:4557–4561. doi:10.1016/j.bmcl.2007.05.092
Farag AM, Mayhoub AS, Barakat SE, Bayomi AH (2008) Synthesis of new \(N\)-phenylpyrazole derivatives with potent antimicrobial activity. Bioorg Med Chem 16:4569–4578. doi:10.1016/j.bmc.2008.02.043
Farghaly AM, Soliman FSG, El Semary MMA, Rostom SAF (2001) Polysubstituted pyrazoles, part 4: synthesis, antimicrobial and antiinflammatory activity of some pyrazoles. Pharmazie 56:28–32
Barcelo M, Ravina E, Masaguer CF, Dominuez E, Areias FM, Brea J, Loza MI (2007) Synthesis and binding affinity of new pyrazole and isoxazole derivatives as potential atypical antipsychotics. Bioorg Med Chem Lett 17:4873–4877. doi:10.1016/j.bmcl.2007.06.045
Seki K, Isegawa J, Fukuda M, Ohki M (1984) Studies on hypolipidemic agents. II. Synthesis and pharmacological properties of alkylpyrazole derivatives. Chem Pharm Bull 32:1568–1577
Cottineau B, Toto P, Marot C, Pipaud A, Chenault J (2002) Synthesis and hypoglycemic evaluation of substituted pyrazole-4-carboxylic acids. Bioorg Med Chem Lett 12:2105–2108. doi:10.1016/S0960-894X(02)00380-3
Larsen JS, Zahran MA, Pedersen EB, Nielsen C (1999) Synthesis of triazenopyrazole derivatives as potential inhibitors of HIV-1. Monatsh Chem 130:1167–1173. doi:10.1007/PL00010295
Elguero J, Goya P, Jagerovic N, Silva AMS (2002) Pyrazoles as drugs: facts and fantasies. Targets Heterocycl Syst 6:52–98
Corte JR, Fang T, Pinto DJP, Han W, Hu Z, Jiang X-J, Li Y-L, Rendina AR, Luettgen JM, Wong PC, et al. (2006). Structure-activity relationships of anthranilamide-based factor Xa inhibitors containing piperidinone and pyridinone P4 moieties. In: 232nd ACS national meeting, San Francisco
Rosinger C, Van Den Berg J, Geraats B (2006) Safened herbicides for Liliiflorae. PCT Int. Appl, WO 2006007982
Natsume B, Kyomura N, Kikutake K, Fukuchi T (1991). Preparation of pyrazolecarboxamides as pesticides. Eur Pat Appl EP 462573
Londershausen M (1996) Approaches to new parasiticides. Pestic Sci 48:269–292. doi:10.1002/(SICI)1096-9063(199612)
Fahmy SM, Badran AH, Elnagdi MH (1980) Synthesis of some new azopyrazole dyes. J Chem Technol Biotechnol 30:390–395. doi:10.1002/jctb.503300147
Fouque D, About-Jaudet E, Collignon N (1995) \(\upalpha \)-Pyrazolyl-alkylphosphonates. Part II. A simple and efficient synthesis of diethyl 1-(pyrazol-4-yl)-alkylphosphonates. Synth Commun 25:3443–3455. doi:10.1080/00397919508013868
Khidre MD, Abou-Yousef HM, Refat M, Mahran H (2002) Organophosphorus chemistry 34. The reaction of alkyl phosphites and hexamethylphosphorus triamide with 1-phenyl-3-methyl-4-ylidene-5-pyrazolones. Phosphorus Sulfur Silicon Relat Elem 177:647–658. doi:10.1080/10426500210260
Tolstikova LL, Bel’skikh AV, Shainyan BA (2011) Protonation and alkylation of organophosphorus compounds with trifluoromethanesulfonic acid derivatives. Russ J Gen Chem 81:377–383. doi:10.1134/S1070363211030054
Boobalan R, Chen C (2013) Catalytic enantioselective hydrophosphonylation of aldehydes using the iron complex of a camphor-based tridentate schiff base [FeCl(SBAIB-d)]\(_{2}\). Adv Synth Catal 355:3443–3450. doi:10.1002/adsc.201300653
Cefarelli G, D’Abrosca B, Fiorentino A, Izzo A, Mastellone C, Pacifico S, Piscopo V (2006) Free-radical-scavenging and antioxidant activities of secondary metabolites from reddened Cv. Annurca Apple Fruits. J Agric Food Chem 54:803–809. doi:10.1021/jf052632g
Campos MG, Webby RF, Markham KR, Mitchell KA, da Cunha AP (2003) Age-induced diminution of free radical scavenging capacity in bee pollens and the contribution of constituent flavonoids. J Agric Food Chem 51:742–745. doi:10.1021/jf0206466
Bauer AW, Kirby WM, Sherris JC, Turck M (1966) Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 45:493–496
Acknowledgments
This work was supported by Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2014R1A6A1031189).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kang, S.R., Lee, Y.R. Efficient one-pot protocol for diverse pyrazolylphosphonates by multi-component reactions: their antioxidant and antibacterial activities. Mol Divers 19, 293–304 (2015). https://doi.org/10.1007/s11030-015-9568-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-015-9568-4