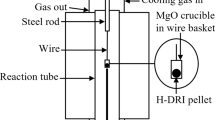
Experimental data are given for a study of dephosphorization and desulfurization of acid oxide melts. Results are provided for determination of phosphorus distribution between phases within acid melts. It is shown that diffusion mass transfer is a limiting stage for phosphorus removal into a gas phase. A two-stage preparation scheme is proposed for iron preparation from ore materials: melting a charge with low basicity (less than 0.9) without adding reducing agents, then oxide melt reduction with preparation of finished metal. The fundamental possibility of preparing iron with a low phosphorus and sulfur content by the suggested scheme is confirmed by experiment.






Similar content being viewed by others
Notes
With higher basicity values, phosphorus removal into a gas phase is difficult due to the effect of calcium cations, and with lower basicity, due to greater melt viscosity, and as a consequence difficult mass transfer.
References
Yu. Yu. Zubkov, Dephosphorization of Highly Alloyed Melts with the Aim of Drawing into Production Waste Metal and Slag with Increased Phosphorus Content: Diss. Cand., Techn. Sci., 05.16.02, Moscow (2008).
Suk Min-Oh, Jo Sung-Koo, Kim Seon-Hyo, et al., “x-Ray observation of phosphorus vaporisation from steelmaking slag and suppression method of phosphorus reversion in liquid iron,” Metal. Mater. Trans. B., 37B, 99-1-7 (2006).
Takayuki Futatsuka, Kiyoteru Shitogiden, Takahiro Miki, et al., “Dissolution behavior of nutrition elements from steelmaking slag into seawater,” ISIJ Int., 44, 753–761 (2004).
V. N. Makushin, Theoretical and Experimental Study of Phosphorus Removal from Stainless Steel with Minimum Alloying Losses: Diss. Cand., Techn. Sci., 05.16.02, Moscow (1995).
A. E. Semin, Dephosphorization and thorough Decarburization of Highly Alloyed Melts under Low Oxidation Capacity Conditions: Diss. Doct., Techn. Sci., 05.16.02, Moscow (1996).
V. A. Romenets, V. S. Valavin, A. B. Usachev, et al., Romelt Process, Ruka i Metally, Moscow (2005).
M. V. Genkin and S. V. Shvetsov, Patent 2373141 RF, IPC С01В25/00, “Method for treating phosphate raw material and device for accomplishing it,” subm. 05.15.2008, publ. 11.20.2009.
V. A. Ershov and S. D. Pimenov, Phosphorus Electrothermy, Khimiya, St. Petersburg (1996).
Ya. B. Dantsis, V. A. Ershov (ed.), G. M. Zhilov, et al., Electrochemical Processes of Chemical Technology: Textbook, Khimiya, Leningrad (1984).
A. V. Dubachev, Study of Refining Chromium Alloys by Means of Gas and Slag Treatment: Diss. Cand., Techn. Sci., 05.16.02, Moscow (2012).
D. I. Rakitin, Dephosphorization in the Production of Chromium-Containing Ferroalloys: Diss. Cand., Techn. Sci., 05.16.02, Moscow (1996).
D. Ya. Povolotskii, V. E. Roshchin, M. A. Ryss, et al., Steel and Ferroalloy Electrometallurgy: Textbook, Metallurgiya, Moscow (1974).
Ken-ichi Shimauchi, Shin-ya Kitamura, and Hiroyuki Shibata, “Distribution of P2O5 between solid dicalcium silicate and liquid phases in CaO–SiO2–Fe2O3 system,” ISIJ Int. 49, No. 45, 505–511 (2009).
Kazuyo Yokoyama, Hironari Kubo, Kazuhiro Mori, et al., “Separation and recovery of phosphorus from steelmaking slags with the aid of a strong magnetic field,” ISIJ Int., 47, No. 10, 1541–1548 (2007).
Jae Hong Shin and Joo Hyun Park, “Effect of atmosphere and slag composition on the evolution of PH3 gas during cooling of reducing dephosphorization slags,” ISIJ Int., 53, No. 3, 385–387 (2013).
Jiang Diao, Bing Xie, Yonghong Wang, and Xu Guo, “Recovery of phosphorus from dephosphorization slag produced by duplex high phosphorus hot metal refining,” ISIJ Int., 52, No. 6, 955–959 (2012).
L. Yu, Y. C. Dong, G. Z. Ye, and S. Du, “Concentrating of vanadium oxide in vanadium rich phase(s) by addition of SiO2 in converter slag,” ISIJ Int., 34, No. 2, 131–137 (2007).
I. A. Krasnyanskaya and G. S. Podgoretskii, “Experimental study of the mechanism of phosphorus removal from oxide melts of the system CaO–SiO2–MgO–Al2O3–P2O5 into a gas phase,” Izv. Vyssh. Uchebn. Zaved., Chern. Met., No. 5, 41–46 (2014).
I. A. Krasnyaskaya, G. S. Podgoretskii, and S. N. Paderin, “Kinetic analysis of dephosphorization of synthetic oxide melts,” Izv. Vyssh. Uchebn. Zaved., Chern. Met., No. 7, 5–9 (2014).
K. E. Wicks and F. E. Blok, Thermodynamic Properties of 65 Elements, Their Oxides, Halogenides, Carbides, and Nitrides [Russian translation], Metallurgiya, Moscow (1965).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Metallurg, No. 10, pp. 20–25, October, 2015.
Rights and permissions
About this article
Cite this article
Krasnyanskaya, I.A., Podgorodetskii, G.S. & Paderin, S.N. Dephosphorization and Desulfurization of Acid Oxide Melts and Produced Iron. Metallurgist 59, 887–894 (2016). https://doi.org/10.1007/s11015-016-0189-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11015-016-0189-7