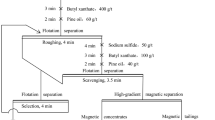
Results from studies of the use of reverse cationic flotation and an effective new reagent-based method for the beneficiation of waste products (Fe ~25% and SiO2 ~54%) formed in processing iron quartzites from the Mihailovskii deposit show that the hematite concentrate which is obtained has an iron content of roughly 60% and contains less than 5% SiO2 (without preliminary dressing). The use of high-gradient magnetic separation for beneficiation of the same waste products yielded iron concentrate with excellent processing properties. It is found that a technology which combines magnetic separation and flotation is the most promising approach to the concentration of materials that contain hematite.
Similar content being viewed by others
References
D. R. Kaplunov and V. A. Yukov, “Feasibility of the commercial use of a combination geotechnology in working KMA deposits,” Fiz. Tekh. Probl. Razrab. Polezn. Iskop., No. 2, 77–83 (2007).
http://www.metallinvest.ru/rus/factorys/gornorudnii-divizion/mgok/MGOK_now/.
H. Sis and S. Chander, “Reagents used in the flotation of phosphate ores: a critical review,” Minerals Eng., 16, 577–585 (2003).
P. K. Weissenborn, L. J. Warren, and J. G. Dunn, “Selective flotation of ultrafine iron ore. 1. Mechanism of adsorption of starch onto hematite,” Colloids and Surfaces A: Physicochem. Eng. Aspects, 99, 11–27 (1995).
I. Dogu and A. I. Arol, “Separation of dark-colored minerals from feldspar by selective flocculation using starch,” Powder Technol., No. 139, 258–263 (2004).
I. O. Filippov, I. V. Filippova, and V. V. Severov, “The use of collectors mixture in the reverse cationic flotation of magnetite ore: The role of Fe-bearing silicates,” Minerals Eng., 23, No. 2, 91–98 (2010).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Metallurg, No. 5, pp. 30–33, May, 2010.
Rights and permissions
About this article
Cite this article
Filippov, L.O., Filippova, I.V. & Severov, V.V. New technology for producing hematite concentrate from wastes generated in the processing of iron quartzites. Metallurgist 54, 268–272 (2010). https://doi.org/10.1007/s11015-010-9291-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11015-010-9291-4