Abstract
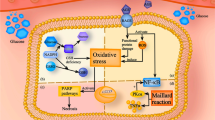
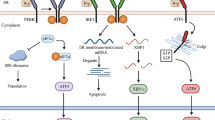
Endoplasmic stress response, the unfolded protein response (UPR), is a homeostatic signaling pathway comprising transmembrane sensors that get activated upon alterations in ER luminal environment. Studies suggest a relation between activated UPR pathways and several disease states such as Parkinson, Alzheimer, inflammatory bowel disease, tumor growth, and metabolic syndrome. Diabetic peripheral neuropathy (DPN), a common microvascular complication of diabetes-related chronic hyperglycemia, causes chronic pain, loss of sensation, foot ulcers, amputations, allodynia, hyperalgesia, paresthesia, and spontaneous pain. Factors like disrupted calcium signaling, dyslipidemia, hyperglycemia, inflammation, insulin signaling, and oxidative stress disturb the UPR sensor levels manifesting as DPN. We discuss new effective therapeutic alternatives for DPN that can be developed by targeting UPR pathways like synthetic ER stress inhibitors like 4-PhenylButyric acid (4-PBA), Sephin 1, Salubrinal and natural ER stress inhibitors like Tauroursodeoxycholic acid (TUDCA), Cordycepin, Proanthocyanidins, Crocin, Purple Rice extract and cyanidin and Caffeic Acid Phenethyl Ester (CAPE).




Similar content being viewed by others
Data availability
Not Applicable.
Code availability (software application or custom code)
Not Applicable.
References
Adams CJ, Kopp MC, Larburu N, Nowak PR, Ali MM (2019) Structure and molecular mechanism of ER stress signaling by the unfolded protein response signal activator IRE1. Front Mol Biosci 12(6):11
Andreassi C, Angelozzi C, Tiziano FD, Vitali T, De Vincenzi E, Boninsegna A, Villanova M, Bertini E, Pini A, Neri G, Brahe C (2004) Phenylbutyrate increases SMN expression in vitro: relevance for treatment of spinal muscular atrophy. Eur J Hum Genet 12(1):59–65
Bek MF, Bayer M, Müller B, Greiber S, Lang D, Schwab A, August C, Springer E, Rohrbach R, Huber TB, Benzing T (2006) Expression and function of C/EBP homology protein (GADD153) in podocytes. Am J Pathol 168(1):20–32
Boden G, Duan X, Homko C, Molina EJ, Song W, Perez O, Cheung P, Merali S (2008) Increase in endoplasmic reticulum stress–related proteins and genes in adipose tissue of obese, insulin-resistant individuals. Diabetes 57(9):2438–2444
Boyce M, Bryant KF, Jousse C, Long K, Harding HP, Scheuner D, Kaufman RJ, Ma D, Coen DM, Ron D, Yuan J (2005) A selective inhibitor of eIF2α dephosphorylation protects cells from ER stress. Science 307:935–939
Brussee V, Cunningham FA, Zochodne DW (2004) Direct insulin signaling of neurons reverses diabetic neuropathy. Diabetes 53(7):1824–1830
Cao SS, Kaufman RJ (2013) Targeting endoplasmic reticulum stress in metabolic disease. Expert Opin Ther Targets 17(4):437–448
Candeias, E, Sebastião I, Cardoso S, Carvalho C, Santos MS, Oliveira CR, Duarte AI (2018) Brain GLP-1/IGF-1 signaling and autophagy mediate exendin-4 protection against apoptosis in type 2 diabetic rats. Mol Neurobiol 55:4030–4050
Celik S, Erdogan S, Tuzcu M (2009) Caffeic acid phenethyl ester (CAPE) exhibits significant potential as an antidiabetic and liver-protective agent in streptozotocin-induced diabetic rats. Pharmacol Res 60(4):270–276
Cermenati G, Abbiati F, Cermenati S, Brioschi E, Volonterio A, Cavaletti G, Saez E, De Fabiani E, Crestani M, Garcia-Segura LM, Melcangi RC (2012) Diabetes-induced myelin abnormalities are associated with an altered lipid pattern: protective effects of LXR activation. J Lipid Res 53(2):300–310
Chen WJ, Wang LX, Wang YP, Chen Z, Liu XY, Liu XH, Liu LB (2012) Exendin-4 protects MIN6 cells from t-BHP-induced apoptosis via IRE1-JNK-caspase-3 signaling. Int J Endocrinol 18:2012
Chen CH, Shih PC, Lin HY, Wang PK, Pan PT, Chuang CW, Kao MC (2019) 4-Phenylbutyric acid protects against vasculitic peripheral neuropathy induced by ischaemia–reperfusion through attenuating endoplasmic reticulum stress. Inflammopharmacology 27(4):713–722
Chen Y, Podojil JR, Kunjamma RB, Jones J, Weiner M, Lin W, Miller SD, Popko B (2019) Sephin1, which prolongs the integrated stress response, is a promising therapeutic for multiple sclerosis. Brain 142(2):344–361
Cho JA, Zhang X, Miller GM, Lencer WI, Nery FC (2014) 4-Phenylbutyrate attenuates the ER stress response and cyclic AMP accumulation in DYT1 dystonia cell models. PLoS ONE 9(11):e110086
Choi JH, Choi AY, Yoon H, Choe W, Yoon K-S, Ha J et al (2010) Baicalein protects HT22 murine hippocampal neuronal cells against endoplasmic reticulum stress-induced apoptosis through inhibition of reactive oxygen species production and CHOP induction. Exp Mol Med 42(12):811–822
Das I, Krzyzosiak A, Schneider K, Wrabetz L, D’Antonio M, Barry N, Sigurdardottir A, Bertolotti A (2015) Preventing proteostasis diseases by selective inhibition of a phosphatase regulatory subunit. Science 348(6231):239–242
Del Signore SJ, Amante DJ, Kim J, Stack EC, Goodrich S, Cormier K, Smith K, Cudkowicz ME, Ferrante RJ (2009) Combined riluzole and sodium phenylbutyrate therapy in transgenic amyotrophic lateral sclerosis mice. Amyotroph Lateral Scler 10(2):85–94
Ding Y, Dai X, Zhang Z, Jiang Y, Ma X, Cai X, Li Y (2014) Proanthocyanidins protect against early diabetic peripheral neuropathy by modulating endoplasmic reticulum stress. J Nutr Biochem 25(7):765–772
Dong Y, Jing T, Meng Q, Liu C, Hu S, Ma Y, Liu Y, Lu J, Cheng Y, Wang D, Teng L (2014) Studies on the antidiabetic activities of Cordyceps militaris extract in diet-streptozotocin-induced diabetic Sprague-Dawley rats. BioMed Res Int 2014 Volume 14.
Doyle KM, Kennedy D, Gorman AM, Gupta S, Healy SJ, Samali A (2011) Unfolded proteins and endoplasmic reticulum stress in neurodegenerative disorders. J Cell Mol Med 15(10):2025–2039
Drummond IA, Lee AS, Resendez E Jr, Steinhardt RA (1987) Depletion of intracellular calcium stores by calcium ionophore A23187 induces the genes for glucose-regulated proteins in hamster fibroblasts. J Biol Chem 262(26):12801–12805
English AR, Voeltz GK (2013) Endoplasmic reticulum structure and interconnections with other organelles. Cold Spring Harb Perspect Biol 5(4):a013227
Farshid AA, Tamaddonfard E (2015) Histopathological and behavioral evaluations of the effects of crocin, safranal and insulin on diabetic peripheral neuropathy in rats. Avicenna J Phytomed 5(5):469
Ferdaoussi M, Abdelli S, Yang JY, Cornu M, Niederhauser G, Favre D, Widmann C, Regazzi R, Thorens B, Waeber G, Abderrahmani A (2008) Exendin-4 Protects β-Cells From Interleukin-1β–Induced Apoptosis by Interfering With the c-Jun NH2-Terminal Kinase Pathway. Diabetes 57(5):1205–1215
Fonseca SG, Urano F, Weir GC, Gromada J, Burcin M (2012) Wolfram syndrome 1 and adenylyl cyclase 8 interact at the plasma membrane to regulate insulin production and secretion. Nat Cell Biol 14(10):1105–1112
Fowler MJ (2011) Microvascular and macrovascular complications of diabetes. Clin Diabetes 29(3):116–122
Fujiwara M, Yamamoto H, Miyagi T, Seki T, Tanaka S, Hide I, Sakai N (2013) Effects of the chemical chaperone 4-phenylbutylate on the function of the serotonin transporter (SERT) expressed in COS-7 cells. J Pharmacol Sci 2013:12194FP
Gao B, Zhang XY, Han R, Zhang TT, Chen C, Qin ZH, Sheng R (2013) The endoplasmic reticulum stress inhibitor salubrinal inhibits the activation of autophagy and neuroprotection induced by brain ischemic preconditioning. Acta Pharmacol Sin 34(5):657–666
Gardian G, Browne SE, Choi DK, Klivenyi P, Gregorio J, Kubilus JK, Ryu H, Langley B, Ratan RR, Ferrante RJ, Beal MF (2005) Neuroprotective effects of phenylbutyrate in the N171–82Q transgenic mouse model of Huntington’s disease. J Biol Chem 280(1):556–563
Görlach A, Klappa P, Kietzmann DT (2006) The endoplasmic reticulum: folding, calcium homeostasis, signaling, and redox control. Antioxid Redox Signal 8(9–10):1391–1418
Gregor MF, Yang L, Fabbrini E, Mohammed BS, Eagon JC, Hotamisligil GS, Klein S (2009) Endoplasmic reticulum stress is reduced in tissues of obese subjects after weight loss. Diabetes 58(3):693–700
Gu YY, Wang H, Wang S, Gao H, Qiu MC (2015) Effects of Cordyceps sinensis on the Expressions of NF-κB and TGF-β1 in Myocardium of Diabetic Rats. Evid Based Complement Alternat Med 1:2015
Han J, Kaufman RJ (2016) The role of ER stress in lipid metabolism and lipotoxicity. J Lipid Res 57(8):1329–1338
Harding HP, Zhang Y, Ron D (1999) Protein translation and folding are coupled by an endoplasmic-reticulum-resident kinase. Nature 397(6716):271–274
Hinder LM, Vincent AM, Burant CF, Pennathur S, Feldman EL (2012) Bioenergetics in diabetic neuropathy: what we need to know. J Peripher Nerv Syst 17:10–14
Huang TJ, Sayers NM, Fernyhough P, Verkhratsky A (2002) Diabetes-induced alterations in calcium homeostasis in sensory neurones of streptozotocin-diabetic rats are restricted to lumbar ganglia and are prevented by neurotrophin-3. Diabetologia 45(4):560–570
Huang X, Chen Y, Zhang H, Ma Q, Zhang YW, Xu H (2012) Salubrinal attenuates β-amyloid-induced neuronal death and microglial activation by inhibition of the NF-κB pathway. Neurobiol Aging 33(5):1007–e9
Inden M, Kitamura Y, Takeuchi H, Yanagida T, Takata K, Kobayashi Y, Taniguchi T, Yoshimoto K, Kaneko M, Okuma Y, Taira T (2007) Neurodegeneration of mouse nigrostriatal dopaminergic system induced by repeated oral administration of rotenone is prevented by 4-phenylbutyrate, a chemical chaperone. J Neurochem 101(6):1491–1504
Jia J, Xiao Y, Wang W, Qing L, Xu Y, Song H et al (2013) Differential mechanisms underlying neuroprotection of hydrogen sulfide donors against oxidative stress. Neurochem Int 62(8):1072–1078
Jin ML, Park SY, Kim YH, Oh JI, Lee SJ, Park G (2014) The neuroprotective effects of cordycepin inhibit glutamate-induced oxidative and ER stress-associated apoptosis in hippocampal HT22 cells. Neurotoxicology 1(41):102–111
Juster-Switlyk K, Smith AG (2016) Updates in diabetic peripheral neuropathy. F1000Research, 5
Kadowaki H, Nishitoh H (2013) Signaling pathways from the endoplasmic reticulum and their roles in disease. Genes 4(3):306–333
Khalaf K, Tornese P, Cocco A, Albanese A (2022) Tauroursodeoxycholic acid: a potential therapeutic tool in neurodegenerative diseases. Transl Neurodegener 11(1):1–7
Kim B, McLean LL, Philip SS, Feldman EL (2011) Hyperinsulinemia induces insulin resistance in dorsal root ganglion neurons. Endocrinology 152(10):3638–3647
Kim JS, Kim H, Yi C-o, Shin HJ, Han JW, Roh GS (2014) Salubrinal, ER stress inhibitor, attenuates kainic acid-induced hippocampal cell death. J Neural Transm 121(10):1233–43
Kostyuk E, Voitenko N, Kruglikov I, Shmigol A, Shishkin V, Efimov A, Kostyuk P (2001) Diabetes-induced changes in calcium homeostasis and the effects of calcium channel blockers in rat and mice nociceptive neurons. Diabetologia 44(10):1302–1309
Kubota K, Niinuma Y, Kaneko M, Okuma Y, Sugai M, Omura T, Uesugi M, Uehara T, Hosoi T, Nomura Y (2006) Suppressive effects of 4-phenylbutyrate on the aggregation of Pael receptors and endoplasmic reticulum stress. J Neurochem 97(5):1259–1268
Laybutt DR, Preston AM, Åkerfeldt MC, Kench JG, Busch AK, Biankin AV, Biden TJ (2007) Endoplasmic reticulum stress contributes to beta cell apoptosis in type 2 diabetes. Diabetologia 50(4):752–763
Li Y, Maher P, Schubert D (1997) Requirement for cGMP in nerve cell death caused by glutathione depletion. J Cell Biol 139(5):1317–1324
Li Z, Zhou Z, Huang G, Hu F, Xiang Y, He L (2013) Exendin-4 protects mitochondria from reactive oxygen species induced apoptosis in pancreatic Beta cells. PLoS ONE 8(10):e76172. https://doi.org/10.1371/journal.pone.0076172
Lin CC, Yue EQ, Liang Z, Yuen Lee SM, Zhang Z, Man Hoi, MP (2021) Crocin inhibited amyloid-beta (Aβ) generation via promoting non-amyloidogenic APP processing and suppressed ER stress UPR signaling in N2a/APP cells. bioRxiv, 2021-09
Liu WJ, Jin HY, Lee KA, Xie SH, Baek HS, Park TS (2011) Neuroprotective effect of the glucagon-like peptide-1 receptor agonist, synthetic exendin-4, in streptozotocin-induced diabetic rats. Br J Pharmacol 164(5):1410–1420
Liu H, Cao D, Liu H, Liu X, Mai W, Lan H, Huo W, Zheng Q (2016) The herbal medicine cordyceps sinensis protects pancreatic beta cells from streptozotocin-induced endoplasmic reticulum stress. Can J Diabetes 40(4):329–335
Liu CL, Li X, Hu GL, Li RJ, He YY, Zhong W, Wang LL (2012) Salubrinal protects against tunicamycin and hypoxia induced cardiomyocyte apoptosis via the PERK-eIF2α signaling pathway. J Geriatr Cardiol JGC 9(3):258
Lupachyk S, Watcho P, Obrosov AA, Stavniichuk R, Obrosova IG (2013a) Endoplasmic reticulum stress contributes to prediabetic peripheral neuropathy. Exp Neurol 1(247):342–348
Lupachyk S, Watcho P, Stavniichuk R, Shevalye H, Obrosova IG (2013b) Endoplasmic reticulum stress plays a key role in the pathogenesis of diabetic peripheral neuropathy. Diabetes 62(3):944–952
Malhotra JD, Kaufman RJ (2007) The endoplasmic reticulum and the unfolded protein response. Semin Cell Dev Biol 18:716–731
Mantuano E, Henry K, Yamauchi T, Hiramatsu N, Yamauchi K, Orita S, Takahashi K, Lin JH, Gonias SL, Campana WM (2011) The unfolded protein response is a major mechanism by which LRP1 regulates Schwann cell survival after injury. J Neurosci 31(38):13376–13385
Mayer CM, Belsham DD (2010) Palmitate attenuates insulin signaling and induces endoplasmic reticulum stress and apoptosis in hypothalamic neurons: rescue of resistance and apoptosis through adenosine 5′ monophosphate-activated protein kinase activation. Endocrinology 151(2):576–585
McCullough KD, Martindale JL, Klotz LO, Aw TY, Holbrook NJ (2001) Gadd153 sensitizes cells to endoplasmic reticulum stress by down-regulating Bcl2 and perturbing the cellular redox state. Mol Cell Biol 21(4):1249–1259
Olatunji OJ, Feng Y, Olatunji OO, Tang J, Ouyang Z, Su Z (2016) Cordycepin protects PC12 cells against 6-hydroxydopamine induced neurotoxicity via its antioxidant properties. Biomed Pharmacother 81:7–14
Oyadomari S, Koizumi A, Takeda K, Gotoh T, Akira S, Araki E, Mori M (2002) Targeted disruption of the Chop gene delays endoplasmic reticulum stress–mediated diabetes. J Clin Investig 109(4):525–532
Ozcan U, Cao Q, Yilmaz E, Lee AH, Iwakoshi NN, Ozdelen E, Tuncman G, Gorgun C, Glimcher LH, Hotamisligil GS (2004) Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306(5695):457–461
Ozcan U, Yilmaz E, Ozcan L, Furuhashi M, Vaillancourt E, Smith RO, Görgün CZ, Hotamisligil GS (2006) Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science 313(5790):1137–1140
Padilla A, Descorbeth M, Almeyda AL, Payne K, De Leon M (2011) Hyperglycemia magnifies Schwann cell dysfunction and cell death triggered by PA-induced lipotoxicity. Brain Res 25(1370):64–79
Pavitt GD, Ron D (2012) New insights into translational regulation in the endoplasmic reticulum unfolded protein response. Cold Spring Harb Perspect Biol 4(6):a012278
Pérez-Arancibia R, Rivas A, Hetz C (2018) (off) Targeting UPR signaling: the race toward intervening ER proteostasis. Expert Opin Ther Targets 22(2):97–100
Plitman E, Nakajima S, de la Fuente-Sandoval C, Gerretsen P, Chakravarty MM, Kobylianskii J, Graff-Guerrero A (2014) Glutamate-mediated excitotoxicity in schizophrenia: a review. Eur Neuropsychopharmacol 24(10):1591–1605
Qi X, Hosoi T, Okuma Y, Kaneko M, Nomura Y (2004) Sodium 4-phenylbutyrate protects against cerebral ischemic injury. Mol Pharmacol 66(4):899–908
Saeedi P, Petersohn I, Salpea P, Malanda B, Karuranga S, Unwin N, Colagiuri S, Guariguata L, Motala AA, Ogurtsova K, Shaw JE (2019) Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas. Diabetes Res Clin Pract 1(157):107843
Shimoda H, Aitani M, Tanaka J, Hitoe S (2015) Purple rice extract exhibits preventive activities on experimental diabetes models and human subjects. J Rice Res 3(2):137
Soliman E, Behairy SF, El-Maraghy NN, Elshazly SM (2019) PPAR-γ agonist, pioglitazone, reduced oxidative and endoplasmic reticulum stress associated with L-NAME-induced hypertension in rats. Life Sci 15(239):117047
Song B, Scheuner D, Ron D, Pennathur S, Kaufman RJ (2008) Chop deletion reduces oxidative stress, improves β cell function, and promotes cell survival in multiple mouse models of diabetes. J Clin Investig 118(10):3378–3389
Sun J, Ren J, Hu X, Hou Y, Yang Y (2021) Therapeutic effects of Chinese herbal medicines and their extracts on diabetes. Biomed Pharmacother 1(142):111977
Thummayot S, Tocharus C, Pinkaew D, Viwatpinyo K, Sringarm K, Tocharus J (2014) Neuroprotective effect of purple rice extract and its constituent against amyloid beta-induced neuronal cell death in SK-N-SH cells. Neurotoxicology 1(45):149–158
Tuli HS, Sandhu SS, Sharma A (2014) Pharmacological and therapeutic potential of Cordyceps with special reference to Cordycepin. 3 Biotech 4(1):1–12
Turan D, Abdik H, Sahin F, Abdik EA (2020) Evaluation of the neuroprotective potential of caffeic acid phenethyl ester in a cellular model of Parkinson’s disease. Eur J Pharmacol 15(883):173342
Vernon PJ, Tang D (2013) Eat-me: autophagy, phagocytosis, and reactive oxygen species signaling. Antioxid Redox Signal 18(6):677–691
Veves A, Backonja M, Malik RA (2008) Painful diabetic neuropathy: epidemiology, natural history, early diagnosis, and treatment options. Pain Med 9(6):660–674
Vincent AM, Russell JW, Low P, Feldman EL (2004) Oxidative stress in the pathogenesis of diabetic neuropathy. Endocr Rev 25(4):612–628
Vincent AM, Mclean LL, Backus C, Feldman EL (2005) Short-term hyperglycemia produces oxidative damage and apoptosis in neurons. FASEB J 19(6):1–24
Vincent AM, Hayes JM, McLean LL, Vivekanandan-Giri A, Pennathur S, Feldman EL (2009a) Dyslipidemia-induced neuropathy in mice: the role of oxLDL/LOX-1. Diabetes 58(10):2376–2385
Vincent AM, Hinder LM, Pop-Busui R, Feldman EL (2009b) Hyperlipidemia: a new therapeutic target for diabetic neuropathy. J Peripher Nerv Syst 14(4):257–267
Wang M, Kaufman RJ (2016) Protein misfolding in the endoplasmic reticulum as a conduit to human disease. Nature 529(7586):326–335
Wang Y, Vera L, Fischer WH, Montminy M (2009) The CREB coactivator CRTC2 links hepatic ER stress and fasting gluconeogenesis. Nature 460(7254):534–537
Wu CT, Sheu ML, Tsai KS, Chiang CK, Liu SH (2011) Salubrinal, an eIF2α dephosphorylation inhibitor, enhances cisplatin-induced oxidative stress and nephrotoxicity in a mouse model. Free Radical Biol Med 51(3):671–680
Yap MK, Misuan N (2019) Exendin-4 from Heloderma suspectum venom: From discovery to its latest application as type II diabetes combatant. Basic Clin Pharmacol Toxicol 124(5):513–527
Ylitalo KR, Sowers M, Heeringa S (2011) Peripheral vascular disease and peripheral neuropathy in individuals with cardiometabolic clustering and obesity: National Health and Nutrition Examination Survey 2001–2004. Diabetes Care 34(7):1642–1647
Zhang GF, Zhang Y, Zhao G (2015) Crocin protects PC12 cells against MPP+-induced injury through inhibition of mitochondrial dysfunction and ER stress. Neurochem Int 1(89):101–110
Zherebitskaya E, Schapansky J, Akude E, Smith DR, Van der Ploeg R, Solovyova N, Verkhratsky A, Fernyhough P (2011) Sensory neurons derived from diabetic rats have diminished internal Ca2+ stores linked to impaired re-uptake by the endoplasmic reticulum. ASN Neuro 4(1):AN20110038
Zhou F, Zhang W, Zhou J, Li M, Zhong F, Zhang Y, Liu Y, Wang Y (2017) Involvement of endoplasmic reticulum stress in formalin-induced pain is attenuated by 4-phenylbutyric acid. J Pain Res 10:653
Author information
Authors and Affiliations
Contributions
Study Conception and design-Shivangi Patel; Material preparation and data collection- Shivangi Patel, Arnika Pangarkar, Sakshi Mahajan. The first draft of the manuscript was written by Shivangi Patel and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval (include appropriate approvals or waivers)
Not Applicable.
Consent to participate (include appropriate statements)
Not Applicable.
Consent for publication (include appropriate statements)
Yes.
Conflicts of interest/competing interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Numerous factors such as disrupted calcium signaling, dyslipidemia, hyperglycemia, inflammation, insulin signaling, and oxidative stress alter the levels of ER stress response signaling molecules, further leading to diabetic peripheral neuropathy (DPN).
• In this review, we suggest that synthetic ER stress inhibitors such as 4-phenylbutyric acid, sephin-1, salubrinal and natural ER stress inhibitors such as tauroursodeoxycholic acid, cordycepin, proanthocyanidins, crocin, purple rice extracts, and caffeic acid phenethyl ester might serve as potential treatment options for DPN by interacting with the ER stress response signaling chaperones.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patel, S., Pangarkar, A., Mahajan, S. et al. Therapeutic potential of endoplasmic reticulum stress inhibitors in the treatment of diabetic peripheral neuropathy. Metab Brain Dis 38, 1841–1856 (2023). https://doi.org/10.1007/s11011-023-01239-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-023-01239-x