Abstract
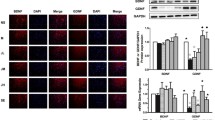
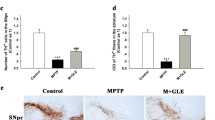
Parkinson’s disease (PD) is a neurodegenerative disease characterized by the pathological loss of nigrostriatal dopaminergic neurons, which causes an insufficient release of dopamine (DA) and then induces motor and nonmotor symptoms. Hyperoside (HYP) is a lignan component with anti-inflammatory, antioxidant, and neuroprotective effects. In this study, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and its active neurotoxic metabolite 1-methyl-4-phenylpyridinium ion (MPP+) were used to induce dopaminergic neurodegeneration. The results showed that HYP (100 µg/mL) reduced MPTP-mediated cytotoxicity of SH-SY5Y cells in vitro, and HYP [25 mg/(kg d)] alleviated MPTP-induced motor symptoms in vivo. HYP treatment reduced the contents of nitric oxide (NO), H2O2, and malondialdehyde (MDA), as well as the mitochondrial damage of dopaminergic neurons, both in vitro and in vivo. Meanwhile, HYP treatment elevated the levels of neurotrophic factors such as glial cell line–derived neurotrophic factor, brain-derived neurotrophic factor, and recombinant cerebral dopamine neurotrophic factor in vivo, but not in vitro. Finally, Akt signaling was activated after the administration of HYP in MPP+/MPTP-induced dopaminergic neurodegeneration. However, the blockage of the Akt pathway with Akt inhibitor did not abolish the neuroprotective effect of HYP on DA neurons. These results showed that HYP protected the dopaminergic neurons from the MPP+- and MPTP-induced injuries, which did not rely on the Akt pathway.








Similar content being viewed by others
Data availability
Data are available from the corresponding author upon request.
References
Adam-Vizi V (2005) Production of reactive oxygen species in brain mitochondria: contribution by electron transport chain and non-electron transport chain sources. Antioxid Redox Signal 7(9–10):1140–1149. https://doi.org/10.1089/ars.2005.7.1140
Akanji MA, Rotimi DE, Elebiyo TC, Awakan OJ, Adeyemi OS (2021) Redox Homeostasis and Prospects for Therapeutic Targeting in Neurodegenerative Disorders. Oxid Med Cell Longev 2021:9971885.https://doi.org/10.1155/2021/9971885
Cao J, Tang C, Gao M, Rui Y, Zhang J, Wang L, Wang Y, Xu B, Yan BC (2020) Hyperoside alleviates epilepsy-induced neuronal damage by enhancing antioxidant levels and reducing autophagy. J Ethnopharmacol 257:112884. https://doi.org/10.1016/j.jep.2020.112884
Chakraborty A, Brauer S, Diwan A (2020) A review of possible therapies for Parkinson’s disease. J Clin Neurosci 76:1–4. https://doi.org/10.1016/j.jocn.2020.03.047
Chang E, Wang J (2021) Brain-derived neurotrophic factor attenuates cognitive impairment and motor deficits in a mouse model of Parkinson’s disease. Brain Behav 11(8):e2251. https://doi.org/10.1002/brb3.2251
Charvin D, Medori R, Hauser RA, Rascol O (2018) Therapeutic strategies for Parkinson disease: beyond dopaminergic drugs. Nat Rev Drug Discov 17(11):804–822. https://doi.org/10.1038/nrd.2018.136
Chen T, Wang J, Liu M, Zhang LY, Liao H (2015) Screening of natural compounds with neuronal differentiation promoting effects in a cell-based model. Chin J Nat Med 13(8):602–608. https://doi.org/10.1016/s1875-5364(15)30056-x
Daina A, Michielin O, Zoete V (2019) SwissTargetPrediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res 47(W1):W357–w364. https://doi.org/10.1093/nar/gkz382
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39(6):889–909. https://doi.org/10.1016/s0896-6273(03)00568-3
Davis AP, Grondin CJ, Johnson RJ, Sciaky D, Wiegers J, Wiegers TC, Mattingly CJ (2021) Comparative toxicogenomics database (CTD): update 2021. Nucleic Acids Res 49(D1):D1138–d1143. https://doi.org/10.1093/nar/gkaa891
Dorman CW, Krug HE, Frizelle SP, Funkenbusch S, Mahowald ML (2014) A comparison of DigiGait™ and TreadScan™ imaging systems: assessment of pain using gait analysis in murine monoarthritis. J Pain Res 7:25–35. https://doi.org/10.2147/jpr.S52195
Floor E, Wetzel MG (1998) Increased protein oxidation in human substantia nigra pars compacta in comparison with basal ganglia and prefrontal cortex measured with an improved dinitrophenylhydrazine assay. J Neurochem 70(1):268–275. https://doi.org/10.1046/j.1471-4159.1998.70010268.x
González-Rodríguez P, Zampese E, Stout KA, Guzman JN, Ilijic E, Yang B, Tkatch T, Stavarache MA, Wokosin DL, Gao L et al (2021) Disruption of mitochondrial complex I induces progressive parkinsonism. Nature 599(7886):650–656. https://doi.org/10.1038/s41586-021-04059-0
Hattori N, Kitada T, Matsumine H, Asakawa S, Yamamura Y, Yoshino H, Kobayashi T, Yokochi M, Wang M, Yoritaka A et al (1998) Molecular genetic analysis of a novel parkin gene in japanese families with autosomal recessive juvenile parkinsonism: evidence for variable homozygous deletions in the parkin gene in affected individuals. Ann Neurol 44(6):935–941. https://doi.org/10.1002/ana.410440612
Jiao X, Sherman BT, Huang da W, Stephens R, Baseler MW, Lane HC, Lempicki RA (2012) DAVID-WS: a stateful web service to facilitate gene/protein list analysis. Bioinformatics 28(13):1805–1806. https://doi.org/10.1093/bioinformatics/bts251
Kalia LV, Lang AE (2015) Parkinson’s disease. Lancet 386(9996):896–912. https://doi.org/10.1016/s0140-6736(14)61393-3
Kim S, Chen J, Cheng T, Gindulyte A, He J, He S, Li Q, Shoemaker BA, Thiessen PA, Yu B et al (2019) PubChem 2019 update: improved access to chemical data. Nucleic Acids Res 47(D1):D1102–d1109. https://doi.org/10.1093/nar/gky1033
Kwon SH, Lee SR, Park YJ, Ra M, Lee Y, Pang C, Kim KH (2019) Suppression of 6-Hydroxydopamine-Induced oxidative stress by Hyperoside Via activation of Nrf2/HO-1 signaling in dopaminergic neurons. Int J Mol Sci 20(23). https://doi.org/10.3390/ijms20235832
Li H, Yahaya BH, Ng WH, Yusoff NM, Lin J (2019) Conditioned medium of human menstrual blood-derived endometrial stem cells protects against MPP(+)-Induced cytotoxicity in vitro. Front Mol Neurosci 12:80. https://doi.org/10.3389/fnmol.2019.00080
Li L, Xu W, Yi C, Cheng Y, Xin H, Xue H, Li CK, Fang X, Yang L, Chen C et al (2021) Astragalus polysaccharide has a protective effect on hematopoiesis in an irradiated mouse model and decreases apoptosis in megakaryocytes. Mol Med Rep 23(1). https://doi.org/10.3892/mmr.2020.11653
Li YR, Fan HJ, Chai Z, Sun MY, Liu JC, Xiao WS, Yu JZ, Li YH, Zhang B, Xiao BG et al (2020a) Neuroprotective and anti-inflammatory effects of Wuzi Yanzong Pills on Parkinson’s disease mice [J]. China J Tradit Chin Med Pharm 35(07):3623–3626 ((in Chinese) CNKI: SUN: BXYY.0.2020-07-096)
Li YR, Fan HJ, Chai Z, Lu J, Sun MY, Qiang M, Xiao WS, Yu JZ, Li YH, Zhang B et al (2020b) Effects of Wuzi Yanzong Pills on behavior and oxidative stress in Parkinson’s disease mice [J]. China J Tradit Chin Med Pharm 35(06):2795–2799 ((in Chinese) CNKI: SUN: BXYY.0.2020-06-027)
Liu RL, Xiong QJ, Shu Q, Wu WN, Cheng J, Fu H, Wang F, Chen JG, Hu ZL (2012) Hyperoside protects cortical neurons from oxygen-glucose deprivation-reperfusion induced injury via nitric oxide signal pathway. Brain Res 1469:164–173. https://doi.org/10.1016/j.brainres.2012.06.044
Long HZ, Cheng Y, Zhou ZW, Luo HY, Wen DD, Gao LC (2021) PI3K/AKT Signal Pathway: a target of Natural Products in the Prevention and Treatment of Alzheimer’s Disease and Parkinson’s Disease. Front Pharmacol 12:648636. https://doi.org/10.3389/fphar.2021.648636
Lorigados Pedre L, Pavón Fuentes N, Alvarez González L, McRae A, Serrano Sánchez T, Blanco Lescano L, Macías González R (2002) Nerve growth factor levels in Parkinson disease and experimental parkinsonian rats. Brain Res 952(1):122–127. https://doi.org/10.1016/s0006-8993(02)03222-5
Marchetti B (2020) Nrf2/Wnt resilience orchestrates rejuvenation of glia-neuron dialogue in Parkinson’s disease. Redox Biol 36:101664. https://doi.org/10.1016/j.redox.2020.101664
Massaquoi MS, Liguore WA, Churchill MJ, Moore C, Melrose HL, Meshul CK (2020) Gait deficits and loss of Striatal Tyrosine Hydroxlase/Trk-B are restored following 7,8-Dihydroxyflavone treatment in a Progressive MPTP Mouse Model of Parkinson’s Disease. Neuroscience 433:53–71. https://doi.org/10.1016/j.neuroscience.2020.02.046
McIntyre CC, Anderson RW (2016) Deep brain stimulation mechanisms: the control of network activity via neurochemistry modulation. J Neurochem 139(Suppl 1):338–345. https://doi.org/10.1111/jnc.13649
Mustapha M, Mat Taib CN (2021) MPTP-induced mouse model of Parkinson’s disease: a promising direction of therapeutic strategies. Bosn J Basic Med Sci 21(4):422–433. https://doi.org/10.17305/bjbms.2020.5181
Nasrolahi A, Mahmoudi J, Akbarzadeh A, Karimipour M, Sadigh-Eteghad S, Salehi R, Farhoudi M (2018) Neurotrophic factors hold promise for the future of Parkinson’s disease treatment: is there a light at the end of the tunnel? Rev Neurosci 29(5):475–489. https://doi.org/10.1515/revneuro-2017-0040
Parain K, Murer MG, Yan Q, Faucheux B, Agid Y, Hirsch E, Raisman-Vozari R (1999) Reduced expression of brain-derived neurotrophic factor protein in Parkinson’s disease substantia nigra. NeuroReport 10(3):557–561. https://doi.org/10.1097/00001756-199902250-00021
Perier C, Vila M (2012) Mitochondrial biology and Parkinson’s disease. Cold Spring Harb Perspect Med 2(2):a009332. https://doi.org/10.1101/cshperspect.a009332
Pöyhönen S, Er S, Domanskyi A, Airavaara M (2019) Effects of neurotrophic factors in glial cells in the Central Nervous System: expression and Properties in Neurodegeneration and Injury. Front Physiol 10:486. https://doi.org/10.3389/fphys.2019.00486
Puspita L, Chung SY, Shim JW (2017) Oxidative stress and cellular pathologies in Parkinson’s disease. Mol Brain 10(1):53. https://doi.org/10.1186/s13041-017-0340-9
Qian H, Kang X, Hu J, Zhang D, Liang Z, Meng F, Zhang X, Xue Y, Maimon R, Dowdy SF et al (2020) Reversing a model of Parkinson’s disease with in situ converted nigral neurons. Nature 582(7813):550–556. https://doi.org/10.1038/s41586-020-2388-4
Redmond DE Jr, Weiss S, Elsworth JD, Roth RH, Wakeman DR, Bjugstad KB, Collier TJ, Blanchard BC, Teng YD, Synder EY et al (2010) Cellular repair in the parkinsonian nonhuman primate brain. Rejuvenation Res 13(2–3):188–194. https://doi.org/10.1089/rej.2009.0960
Robea MA, Balmus IM, Ciobica A, Strungaru S, Plavan G, Gorgan LD, Savuca A, Nicoara M (2020) Parkinson’s Disease-Induced Zebrafish Models: Focussing on Oxidative Stress Implications and Sleep Processes. Oxid Med Cell Longev 2020:1370837.https://doi.org/10.1155/2020/1370837
Schapira AH, Cooper JM, Dexter D, Clark JB, Jenner P, Marsden CD (1990) Mitochondrial complex I deficiency in Parkinson’s disease. J Neurochem 54(3):823–827. https://doi.org/10.1111/j.1471-4159.1990.tb02325.x
Schapira AH, Cooper JM, Dexter D, Jenner P, Clark JB, Marsden CD (1989) Mitochondrial complex I deficiency in Parkinson’s disease. Lancet 1(8649):1269. https://doi.org/10.1016/s0140-6736(89)92366-0
Smoot ME, Ono K, Ruscheinski J, Wang PL, Ideker T (2011) Cytoscape 2.8: new features for data integration and network visualization. Bioinformatics 27(3):431–432. https://doi.org/10.1093/bioinformatics/btq675
Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork P et al (2019) STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res 47(D1):D607–d613. https://doi.org/10.1093/nar/gky1131
Valente EM, Abou-Sleiman PM, Caputo V, Muqit MM, Harvey K, Gispert S, Ali Z, Del Turco D, Bentivoglio AR, Healy DG et al (2004) Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science 304(5674):1158–1160. https://doi.org/10.1126/science.1096284
Wang X, Michaelis EK (2010) Selective neuronal vulnerability to oxidative stress in the brain. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2010.00012
Yang Y, Li S, Qu Y, Wang X, An W, Li Z, Han Z, Qin L (2020) Nitrate partially inhibits lipopolysaccharide-induced inflammation by maintaining mitochondrial function. J Int Med Res 48(2):300060520902605. https://doi.org/10.1177/0300060520902605
Zeng KW, Wang XM, Ko H, Kwon HC, Cha JW, Yang HO (2011) Hyperoside protects primary rat cortical neurons from neurotoxicity induced by amyloid β-protein via the PI3K/Akt/Bad/Bcl(XL)-regulated mitochondrial apoptotic pathway. Eur J Pharmacol 672(1–3):45–55. https://doi.org/10.1016/j.ejphar.2011.09.177
Acknowledgements
We would like to deeply appreciate all authors who performed all eligible studies which have been included in the present paper.
Funding
This study was supported by the National Natural Science Foundation of China (Nos. 81703978 and 81102552), the Central Government Guided Local Funding Projects for Science and Technology Development (No.YDZX20201400001483), the Outstanding Youth Talents Program of Shanxi Province (No. [2019]35), the Natural Science Foundation of Shanxi Province (No.20210302124293), the Research Project supported by Shanxi Scholarship Council of China (No.2021 − 142), the Key science and technology R&D project of Jinzhong (No. Y213004), the Young Scientist Cultivation Program Project, Shanxi University of Chinese Medicine (No.2021PY-QN-03),and the special fund for Science and Technology Innovation Team of Shanxi University of Chinese Medicine (No.2022TD1013).
Author information
Authors and Affiliations
Contributions
Zhi Chai, Cun-Gen Ma, Bo Zhang, Zhen-Yu Li, and Bao-Guo Xiao provided overall guidance for designing the experiment, model establishment, and manuscript writing. Xing-Jie Xu drafted the manuscript and performed experiments in vitro together with Tao Pan and Hui-Jie Fan. Xu Wang contributed to the animal experiments and manuscript writing. Jie-Zhong Yu and Hai-Fei Zhang contributed to the animal experiments and supervised the data analysis. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
The study was approved by the Animal Care Committee of Shanxi University of Chinese Medicine (2021DW268, Jinzhong, China) and complied with the laboratory animal guidelines of the International Council of Laboratory Animals.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, XJ., Pan, T., Fan, HJ. et al. Neuroprotective effect of hyperoside in MPP+/MPTP -induced dopaminergic neurodegeneration. Metab Brain Dis 38, 1035–1050 (2023). https://doi.org/10.1007/s11011-022-01153-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01153-8