Abstract
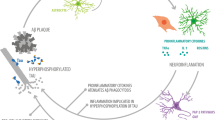
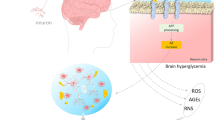
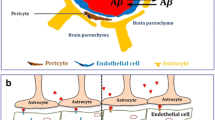
Alzheimer’s disease (AD) is a neurodegenerative disorder that leads to cognitive decline and memory loss. Insulin resistance in central nervous system (CNS) is a common feature in dementia. Defective insulin signaling is associated to higher levels of inflammation and to neuronal dysfunction. A reactive gliosis, a change that occurs in glial cells due to damage in CNS, seems to be one of the most important pro-inflammatory mechanisms in AD pathology. The first response to CNS injury is the migration of macrophages and microglia to the specific site of the injury. Oligodendrocytes are also recruited to to contribute with remyelination. The last component of a reactive gliosis is astrogliosis, which is the enhancement of astrocytes expression with concomitant changes in its morphology being the main cells of the glial scar. Here, we review the mechanisms by which a reactive gliosis can induce or contribute to the development and progression of AD.



Similar content being viewed by others
Data availability
All data generated during this study are included in this published article.
Code availability
Not applicable.
References
Alkadhi KA, Dao AT (2018) Exercise decreases BACE and APP levels in the hippocampus of a rat model of Alzheimer’s disease. Mol Cell Neurosci 86:25–29. https://doi.org/10.1016/j.mcn.2017.11.008
Barker AJ, Ullian EM (2008) New roles for astrocytes in developing synaptic circuits. Commun Integr Biol 1(2):207–211
Bélanger M, Allaman I, Magistretti PJ (2011) Brain energy metabolism: Focus on Astrocyte-neuron metabolic cooperation. Cell Metab 14:724–738. https://doi.org/10.1016/j.cmet.2011.08.016
Bennett MVL, Contreras JE, Bukauskas FF, Sáez JC (2003) New roles for astrocytes: gap junction hemichannels have something to communicate. Trends Neurosci 26:610–617. https://doi.org/10.1016/j.tins.2003.09.008
Bhat R, Crowe EP, Bitto A et al (2012) Astrocyte Senescence as a Component of Alzheimer’s Disease. PLoS ONE 7:1–10. https://doi.org/10.1371/journal.pone.0045069
Bollinger JL, Burns CMB, Wellman CL (2017) Differential Effects of Stress on Microglial Cell Activation in Male and Female Medial Prefrontal Cortex. Physiol Behav 176:139–148. https://doi.org/10.1016/j.bbi.2015.10.003.Differential
Carter SF, Herholz K, Rosa-Neto P et al (2019) Astrocyte Biomarkers in Alzheimer’s Disease. Trends Mol Med 25:77–95. https://doi.org/10.1016/j.molmed.2018.11.006
Cavalcante BRR, Improta-caria AC, de Melo VH, De Sousa RAL (2021) Exercise-linked consequences on epilepsy. Epilepsy Behav 121:1–6. https://doi.org/10.1016/j.yebeh.2021.108079
Chang KJ, Redmond SA, Chan JR (2016) Remodeling myelination: Implications for mechanisms of neural plasticity. Nat Neurosci 19:190–197. https://doi.org/10.1038/nn.4200
Chen K, Zheng Y, Wei J, an et al (2019) Exercise training improves motor skill learning via selective activation of mTOR. Sci Adv 5:1–12. https://doi.org/10.1126/sciadv.aaw1888
Chen Z, Trapp BD (2016) Microglia and neuroprotection. J Neurochem 136(Suppl):10–17. https://doi.org/10.1111/jnc.13062
Cherry JD, Olschowka JA, O’Banion MK (2014) Neuroinflammation and M2 microglia: The good, the bad, and the inflamed. J Neuroinflammation 11:1–15. https://doi.org/10.1186/1742-2094-11-98
De Sousa RAL (2021) Animal models of gestational diabetes : characteristics and consequences to the brain and behavior of the offspring. Metab Brain Dis 2014–2019
De Sousa RAL (2017) Brief report of the effects of the aerobic, resistance, and high-intensity interval training in type 2 diabetes mellitus individuals Diabetes mellitus. Int J Diabetes Dev Ctries 38:138–145. https://doi.org/10.1007/s13410-017-0582-1
De Sousa RAL, Caria ACI, De Jesus Silva FM et al (2020a) High-intensity resistance training induces changes in cognitive function, but not in locomotor activity or anxious behavior in rats induced to type 2 diabetes. Physiol Behav 223:1–7. https://doi.org/10.1016/j.physbeh.2020.112998
De Sousa RAL, Harmer AR, Freitas DA et al (2020b) An update on potential links between type 2 diabetes mellitus and Alzheimer’s disease. Mol Biol Rep 47:6347–6356. https://doi.org/10.1007/s11033-020-05693-z
De Sousa RAL, Improta-Caria AC (2022) Regulation of microRNAs in Alzheimer´s disease, type 2 diabetes, and aerobic exercise training. Metab Brain Dis. https://doi.org/10.1007/s11011-022-00903-y
De Sousa RAL, Improta-Caria AC, de Freitas Souza BS (2021c) Exercise–linked irisin: Consequences on mental and cardiovascular health in type 2 diabetes. Int J Mol Sci 22:1–15. https://doi.org/10.3390/ijms22042199
De Sousa RAL, Improta-caria AC, Aras-júnior R et al (2021a) Physical exercise effects on the brain during COVID-19 pandemic : links between mental and cardiovascular health. Neurol Sci 42:1325–1334
De Sousa RAL, Peixoto MFD, Leite HR et al (2020c) Neurological consequences of exercise during prenatal Zika virus exposure to mice pups. Int J Neurosci 21:1–11. https://doi.org/10.1080/00207454.2020.1860970
De Sousa RAL, Santos LG, Lopes PM, et al (2021b) Physical exercise consequences on memory in obesity : A systematic review. Obes Rev 1–10. https://doi.org/10.1111/obr.13298
Di Castro MA, Chuquet J, Liaudet N et al (2011) Local Ca2+ detection and modulation of synaptic release by astrocytes. Nat Neurosci 14:1276–1284. https://doi.org/10.1038/nn.2929
Exalto LG, Whitmer RA, Kappele LJ, Biessels GJ (2012) An update on type 2 diabetes, vascular dementia and Alzheimer’s disease. Exp Gerontol 47:858–864. https://doi.org/10.1016/j.exger.2012.07.014
Fawcett JW, Asher RA (1999) The glial scar and central nervous system repair. Brain Res Bull 49:377–391. https://doi.org/10.1016/S0361-9230(99)00072-6
Fiacco TA, Agulhon C, Mccarthy KD (2009) Sorting Out Astrocyte Physiology from Pharmacology. Annu Rev Pharmacol Toxicol 49:151–174. https://doi.org/10.1146/annurev.pharmtox.011008.145602
Fourgeaud L, Traves PG, Tufail Y et al (2016) TAM receptors regulate multiple features of microglial physiology. Nature 532:240–244. https://doi.org/10.1038/nature17630
Gao Y, Vidal-Itriago AA, Kalsbeek MJ et al (2017) Lipoprotein Lipase Maintains Microglial Innate Immunity in Obesity. Cell Rep 20:3034–3042. https://doi.org/10.1016/j.celrep.2017.09.008
Gehrmann J, Matsumoto Y, Kreutzberg GW (1995) REVIEWS Microglia : intrinsic immuneffector cell of the brain. Brain Res Rev 20:269–287
Hambardzumyan D, Gutmann DH, Kettenmann H (2015) The role of microglia and macrophages in glioma maintenance and progression. Nat Neurosci 19:20–27. https://doi.org/10.1038/nn.4185
Hasel P, Dando O, Jiwaji Z et al (2017) Neurons and neuronal activity control gene expression in astrocytes to regulate their development and metabolism. Nat Commun 8:15132. https://doi.org/10.1038/ncomms15132
Improta Caria AC, Nonaka CKV, Pereira CS et al (2018) Exercise Training-Induced Changes in MicroRNAs: Beneficial Regulatory Effects in Hypertension, Type 2 Diabetes, and Obesity. Int J Mol Sci 19:1–36. https://doi.org/10.3390/ijms19113608
Li Y, Du X-F, Liu C-S et al (2012) Reciprocal regulation between resting microglial dynamics and neuronal activity in vivo. Dev Cell 23:1189–1202. https://doi.org/10.1016/j.devcel.2012.10.027
Lian Q, Dheen ST, Liao D, Tay SSW (2004) Enhanced Inflammatory Response in Neural Tubes of Embryos Derived from Diabetic Mice Exposed to a Teratogen. J Neurosci Res 75:554–564. https://doi.org/10.1002/jnr.20006
Matias I, Morgado J, Gomes FCA (2019) Astrocyte Heterogeneity: Impact to Brain Aging and Disease. Front Aging Neurosci 11:1–18. https://doi.org/10.3389/fnagi.2019.00059
McCullough LD, Mirza MA, Xu Y et al (2016) Stroke sensitivity in the aged: Sex chromosome complement vs gonadal hormones. Aging (Albany NY) 8:1432–1441. https://doi.org/10.18632/aging.100997
Nichols MR, St-Pierre M-K, Wendeln A-C, et al (2019) Inflammatory Mechanisms in Neurodegeneration. J Neurochem 0–3. https://doi.org/10.1111/jnc.14674
Perry VH, Nicoll JAR, Holmes C (2010) Microglia in neurodegenerative disease. Nat Rev Neurol 6:193–201. https://doi.org/10.1038/nrneurol.2010.17
Prinz M, Priller J (2014) Microglia and brain macrophages in the molecular age: from origin to neuropsychiatric disease. Nat Rev Neurosci 15:300–312. https://doi.org/10.1038/nrn3722
Roth AD, Ramírez G, Alarcón R, Von Bernhardi R (2005) Oligodendrocytes damage in Alzheimer’s disease: Beta amyloid toxicity and inflammation. Biol Res 38:381–387. https://doi.org/10.4067/S0716-97602005000400011
Salter MW, Stevens B (2017) Microglia emerge as central players in brain disease. Nat Med 23:1018–1027. https://doi.org/10.1038/nm.4397
Soreq L, Rose J, Soreq E et al (2017) Major Shifts in Glial Regional Identity Are a Transcriptional Hallmark of Human Brain Aging. Cell Rep 18:557–570. https://doi.org/10.1016/j.celrep.2016.12.011
Streit WJ (2006) Microglial senescence: does the brain’s immune system have an expiration date? Trends Neurosci 29:506–510. https://doi.org/10.1016/j.tins.2006.07.001
Tapia-Rojas C, Aranguiz F, Varela-Nallar L, Inestrosa NC (2016) Voluntary Running Attenuates Memory Loss, Decreases Neuropathological Changes and Induces Neurogenesis in a Mouse Model of Alzheimer’s Disease. Brain Pathol 26:62–74. https://doi.org/10.1111/bpa.12255
Tomassoni D, Nwankwo IE, Gabrielli MG et al (2013) Astrogliosis in the brain of obese Zucker rat: a model of metabolic syndrome. Neurosci Lett 543:136–141. https://doi.org/10.1016/j.neulet.2013.03.025
Wang K, Li H, Wang H, et al (2018) Irisin exerts neuroprotective effects on cultured neurons by regulating astrocytes. Mediators Inflamm 1–7. https://doi.org/10.1155/2018/9070341
Yang G, Cancino GI, Zahr SK et al (2016) A Glo1-Methylglyoxal Pathway that Is Perturbed in Maternal Diabetes Regulates Embryonic and Adult Neural Stem Cell Pools in Murine Offspring. Cell Rep 17:1022–1036. https://doi.org/10.1016/j.celrep.2016.09.067
Zhang J, Guo Y, Wang Y et al (2018) Long-term treadmill exercise attenuates Aβ burdens and astrocyte activation in APP/PS1 mouse model of Alzheimer’s disease. Neurosci Lett 666:70–77. https://doi.org/10.1016/j.neulet.2017.12.025
Zhang PF, Hu H, Tan L, Yu JT (2021) Microglia Biomarkers in Alzheimer’s Disease. Mol Neurobiol 58:3388–3404. https://doi.org/10.1007/s12035-021-02348-3
Funding
This work was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil—Finance Code 001.
Author information
Authors and Affiliations
Contributions
Ricardo Augusto Leoni De Sousa had the idea for the article, performed the literature search and data analysis, and critically revised the work.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
De Sousa, R.A.L. Reactive gliosis in Alzheimer’s disease: a crucial role for cognitive impairment and memory loss. Metab Brain Dis 37, 851–857 (2022). https://doi.org/10.1007/s11011-022-00953-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-00953-2