Abstract
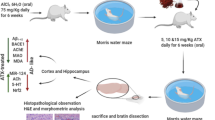
Alzheimer’s disease (AD) is a most common neurodegenerative disease. Sodium Tanshinone IIA Sulfonate (STS) has been reported to ameliorate AD pathology. However, the underlying mechanism is still unclear. In this study, AD transgenic mouse model (APP/PS1) was used to explore the potential mechanism of STS against AD. Morris water maze and Y-maze tests showed that administration of STS improved learning and memory abilities of APP/PS1 mice. STS reduced the levels of reactive oxygen species and malondialdehyde, while improved the activity of superoxide dismutase in both hippocampus and cortex in APP/PS1 mice. STS inhibited the activity of acetylcholinesterase, while improved the activity of choline acetyltransferase in APP/PS1 mice. In addition, STS elevated the protein expressions of neurotrophic factors and synapse-related proteins in both the hippocampus and cortex in APP/PS1 mice. At last, STS improved the protein expressions of glucose transporter 1 (GLUT1) and low-density lipoprotein receptor-related protein 1 (LRP1). These results indicated that the potential mechanism of STS on AD might be related to Aβ transportation function via GLUT1/LRP1 pathway.
Highlights
STS improves cognitive impairment of APP/PS1 mice.
STS ameliorates the oxidative stress damage and improves the cholinergic system.
STS protects against neuronal dysfunction and enhances the synaptic plasticity.
STS mediates the Aβ transportation of BMECs.







Similar content being viewed by others
Availability of data and materials
All original data and materials in the manuscript are available from the author.
Abbreviations
- AD:
-
Alzheimer's disease
- STS:
-
Sodium Tanshinone IIA Sulfonate
- Tan IIA:
-
Tanshinone IIA
- Danshen:
-
Salvia miltiorrhiza Bge
- ChAT:
-
Choline acetyltransferase
- AChE:
-
Acetylcholinesterase
- ROS:
-
Reactive oxygen species
- MDA:
-
Malondialdehyde
- SOD:
-
Superoxide dismutase
- PSD95:
-
Postsynaptic density 95
- PSD93:
-
Postsynaptic density 93
- SYP:
-
Synaptophysin
- NGF:
-
Nerve growth factor
- BDNF:
-
Brain-derived neurotrophic factor
- BMECs:
-
Brain microvascular endothelial cells
- GLUT1:
-
Glucose transporter
- LRP1:
-
Low-density lipoprotein receptor-related protein 1
- RAGE:
-
Receptor for advanced glycation end products
- APP/PS1:
-
APPswe/PSEN1dE9 double transgenic mice
- WT:
-
Wild-type mice
- BBB:
-
Blood brain barrier
- D-PBS:
-
Dulbecco’s PBS
References
Allen SJ, Watson JJ, Shoemark DK, Barua NU, Patel NK (2013) GDNF, NGF and BDNF as therapeutic options for neurodegeneration. Pharmacol Ther 138(2):155–175. https://doi.org/10.1016/j.pharmthera.2013.01.004
Arancibia S, Silhol M, Mouliц╗Re F, Meffre J, Hц╤Llinger I, Maurice T, & Tapia-Arancibia L, (2008) Protective effect of BDNF against beta-amyloid induced neurotoxicity in vitro and in vivo in rats. Neurobiol Dis 31(3):316–326. https://doi.org/10.1016/j.nbd.2008.05.012
Barage SH, Sonawane KD (2015) Amyloid cascade hypothesis: Pathogenesis and therapeutic strategies in Alzheimer’s disease. Neuropeptides 52:1–18. https://doi.org/10.1016/j.npep.2015.06.008
Cai Z, Liu N, Wang C, Qin B, Zhou Y, Xiao M, Chang L, Yan LJ, Zhao B (2016) Role of RAGE in Alzheimer’s Disease. Cell Mol Neurobiol 36(4):483–495. https://doi.org/10.1007/s10571-015-0233-3
Carvajal FJ, Inestrosa NC (2011) Interactions of AChE with Aн╡ Aggregates in Alzheimer’s Brain: Therapeutic Relevance of IDN 5706. Front Mol Neurosci 4:19. https://doi.org/10.3389/fnmol.2011.00019
Davies P, Maloney AJ (1976) Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet 2(8000):1403. https://doi.org/10.1016/s0140-6736(76)91936-x
Faiq MA, Wollstein G, Schuman JS, Chan KC (2019) Cholinergic nervous system and glaucoma: From basic science to clinical applications. Prog Retin Eye Res 72:100767. https://doi.org/10.1016/j.preteyeres.2019.06.003
Ferreira-Vieira TH, Guimaraes IM, Silva FR, Ribeiro FM (2016) Alzheimer’s disease: Targeting the Cholinergic System. Curr Neuropharmacol 14(1):101–115. https://doi.org/10.2174/1570159x13666150716165726
Gauthier S (2002) Advances in the pharmacotherapy of Alzheimer’s disease. CMAJ 166(5):616–623
Horwood N, Davies DC (1994) Immunolabelling of hippocampal microvessel glucose transporter protein is reduced in Alzheimer’s disease. Virchows Arch 425(1):69–72. https://doi.org/10.1007/bf00193951
Hsia AY, Masliah E, Mcconlogue L, Yu GQ, Tatsuno G, Hu K, Kholodenko D, Malenka RC, Nicoll RA, Mucke L (1999) Plaque-independent disruption of neural circuits in Alzheimer’s disease mouse models. Proc Natl Acad Sci U S A 96(6):3228–3233. https://doi.org/10.1073/pnas.96.6.3228
Hsieh H, Boehm J, Sato C, Iwatsubo T, Tomita T, Sisodia S, Malinow R (2006) AMPAR removal underlies Abeta-induced synaptic depression and dendritic spine loss. Neuron 52(5):831–843. https://doi.org/10.1016/j.neuron.2006.10.035
Ji B, Zhou F, Han L, Yang J, Fan H, Li S, Li J, Zhang X, Wang X, Chen X, Xu Y (2017) Sodium Tanshinone IIA Sulfonate Enhances Effectiveness Rt-PA Treatment in Acute Ischemic Stroke Patients Associated with Ameliorating Blood-Brain Barrier Damage. Transl Stroke Res 8(4):334–340. https://doi.org/10.1007/s12975-017-0526-6
Jones DP (2006) Redefining oxidative stress. Antioxid Redox Signal 8(9–10):1865–1879. https://doi.org/10.1089/ars.2006.8.1865
Kalaria RN, Harik SI (1989) Reduced glucose transporter at the blood-brain barrier and in cerebral cortex in Alzheimer disease. J Neurochem 53(4):1083–1088. https://doi.org/10.1111/j.1471-4159.1989.tb07399.x
Kryscio RJ, Abner EL, Caban-Holt A, Lovell M, Goodman P, Darke AK, Yee M, Crowley J, Schmitt FA (2017) Association of Antioxidant Supplement Use and Dementia in the Prevention of Alzheimer’s Disease by Vitamin E and Selenium Trial (PREADViSE). JAMA Neurol 74(5):567–573. https://doi.org/10.1001/jamaneurol.2016.5778
Lane CA, Hardy J, Schott JM (2018) Alzheimer’s disease. Eur J Neurol 25(1):59–70. https://doi.org/10.1111/ene.13439
Li D, Wang J, Sun D, Gong X, Jiang H, Shu J, Wang Z, Long Z, Chen Y, Zhang Z, Yuan L, Guan R, Liang X, Li Z, Yao H, Zhong N, Lu W (2018) Tanshinone IIAб═sulfonate protects against cigarette smoke-induced COPD and down-regulation of CFTR in mice. Sci Rep 8(1):376. https://doi.org/10.1038/s41598-017-18745-5
Liu HC, Liu HH (2013) Adverse reactions of tanshinone II(A) sodium sulfonate injection in treating 18 cases: an analysis of clinical features. Zhongguo Zhong Xi Yi Jie He Za Zhi 33(9):1287–1289
Liu XQ, Deng YX, Dai Z, Hu T, Cai WW, Liu HF, Li H, Zhu WL, Li BY, Wang Q, Zhang SJ (2020) Sodium tanshinone IIA sulfonate protects against Aн╡(1–42)-induced cellular toxicity by modulating Aн╡-degrading enzymes in HT22 cells. Int J Biol Macromol 151:47–55. https://doi.org/10.1016/j.ijbiomac.2020.02.040
Mamounas LA, Blue ME, Siuciak JA, Altar CA (1995) Brain-derived neurotrophic factor promotes the survival and sprouting of serotonergic axons in rat brain. J Neurosci 15(12):7929–7939. https://doi.org/10.1523/jneurosci.15-12-07929.1995
Mooradian AD, Chung HC, Shah GN (1997) GLUT-1 expression in the cerebra of patients with Alzheimer’s disease. Neurobiol Aging 18(5):469–474. https://doi.org/10.1016/s0197-4580(97)00111-5
Mucke L, Masliah E, Yu GQ, Mallory M, Rockenstein EM, Tatsuno G, Hu K, Kholodenko D, Johnson-Wood K, Mcconlogue L (2000) High-level neuronal expression of abeta 1–42 in wild-type human amyloid protein precursor transgenic mice: synaptotoxicity without plaque formation. J Neurosci 20(11):4050–4058. https://doi.org/10.1523/jneurosci.20-11-04050.2000
Navone SE, Marfia G, Invernici G, Cristini S, Nava S, Balbi S, Sangiorgi S, Ciusani E, Bosutti A, Alessandri G, Slevin M, Parati EA (2013) Isolation and expansion of human and mouse brain microvascular endothelial cells. Nat Protoc 8(9):1680–1693. https://doi.org/10.1038/nprot.2013.107
Newcomer JW, Farber NB, & Olney JW (2000) NMDA receptor function, memory, and brain aging. Dialogues Clin Neurosci 2(3): 219–232. https://doi.org/10.31887/DCNS.2000.2.3/jnewcomer
Osgood D, Miller MC, Messier AA, Gonzalez L, Silverberg GD (2017) Aging alters mRNA expression of amyloid transporter genes at the blood-brain barrier. Neurobiol Aging 57:178–185. https://doi.org/10.1016/j.neurobiolaging.2017.05.011
Schott JM, Aisen PS, Cummings JL, Howard RJ, Fox NC (2019) Unsuccessful trials of therapies for Alzheimer’s disease. Lancet 393(10166):29. https://doi.org/10.1016/s0140-6736(18)31896-8
Selkoe DJ (2001) Alzheimer’s disease: genes, proteins, and therapy. Physiol Rev 81(2):741–766. https://doi.org/10.1152/physrev.2001.81.2.741
Sevigny J, Chiao P, Bussiц╗Re T, Weinreb PH, Williams L, Maier M, Dunstan R, Salloway S, Chen T, Ling Y, O’gorman J, Qian F, Arastu M, Li M, Chollate S, Brennan MS, Quintero-Monzon O, Scannevin RH, Arnold HM, Engber T, Rhodes K, Ferrero J, Hang Y, Mikulskis A, Grimm J, Hock C, Nitsch RM, & Sandrock A, (2016) The antibody aducanumab reduces Aн╡ plaques in Alzheimer’s disease. Nature 537(7618):50–56. https://doi.org/10.1038/nature19323
Shang Q, Xu H, Huang L (2012) Tanshinone IIA: A Promising Natural Cardioprotective Agent. Evid Based Complement Alternat Med 2012:716459. https://doi.org/10.1155/2012/716459
Simpson IA, Chundu KR, Davies-Hill T, Honer WG, Davies P (1994) Decreased concentrations of GLUT1 and GLUT3 glucose transporters in the brains of patients with Alzheimer’s disease. Ann Neurol 35(5):546–551. https://doi.org/10.1002/ana.410350507
Sweeney MD, Sagare AP, Zlokovic BV (2018) Blood-brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat Rev Neurol 14(3):133–150. https://doi.org/10.1038/nrneurol.2017.188
Tarasoff-Conway JM, Carare RO, Osorio RS, Glodzik L, Butler T, Fieremans E, Axel L, Rusinek H, Nicholson C, Zlokovic BV, Frangione B, Blennow K, Mц╘Nard J, Zetterberg H, Wisniewski T, & De Leon MJ, (2015) Clearance systems in the brain-implications for Alzheimer disease. Nat Rev Neurol 11(8):457–470. https://doi.org/10.1038/nrneurol.2015.119
Uttara B, Singh AV, Zamboni P, Mahajan RT (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7(1):65–74. https://doi.org/10.2174/157015909787602823
Wang XC, Xu YM, Li HY, Wu CY, Xu TT, Luo NC, Zhang SJ, Wang Q, Quan SJ (2018) Jiao-Tai-Wan Improves Cognitive Dysfunctions through Cholinergic Pathway in Scopolamine-Treated Mice. Biomed Res Int 2018:3538763. https://doi.org/10.1155/2018/3538763
Whitehouse PJ, Price DL, Struble RG, Clark AW, Coyle JT, Delon MR (1982) Alzheimer’s disease and senile dementia: loss of neurons in the basal forebrain. Science 215(4537):1237–1239. https://doi.org/10.1126/science.7058341
Winkler EA, Nishida Y, Sagare AP, Rege SV, Bell RD, Perlmutter D, Sengillo JD, Hillman S, Kong P, Nelson AR, Sullivan JS, Zhao Z, Meiselman HJ, Wendy RB, Soto J, Abel ED, Makshanoff J, Zuniga E, De Vivo DC, Zlokovic BV (2015) GLUT1 reductions exacerbate Alzheimer’s disease vasculo-neuronal dysfunction and degeneration. Nat Neurosci 18(4):521–530. https://doi.org/10.1038/nn.3966
Xu J, Zhang P, Chen Y, Xu Y, Luan P, Zhu Y, Zhang J (2021) Sodium tanshinone IIA sulfonate ameliorates cerebral ischemic injury through regulation of angiogenesis. Exp Ther Med 22(4):1122. https://doi.org/10.3892/etm.2021.10556
Xu QQ, Xu YJ, Yang C, Tang Y, Li L, Cai HB, Hou BN, Chen HF, Wang Q, Shi XG, Zhang SJ (2016) Sodium Tanshinone IIA Sulfonate Attenuates Scopolamine-Induced Cognitive Dysfunctions via Improving Cholinergic System. Biomed Res Int 2016:9852536. https://doi.org/10.1155/2016/9852536
Xu YM, Wang XC, Xu TT, Li HY, Hei SY, Luo NC, Wang H, Zhao W, Fang SH, Chen YB, Guan L, Fang YQ, Zhang SJ, Wang Q, Liang WX (2019) Kai Xin San ameliorates scopolamine-induced cognitive dysfunction. Neural Regen Res 14(5):794–804. https://doi.org/10.4103/1673-5374.249227
Zhang DP, Lu XY, He SC, Li WY, Ao R, Leung FC, Zhang ZM, Chen QB, Zhang SJ (2020) Sodium tanshinone IIA sulfonate protects against Aн╡-induced cell toxicity through regulating Aн╡ process. J Cell Mol Med 24(6):3328–3335. https://doi.org/10.1111/jcmm.15006
Zhou ZY, Huang B, Li S, Huang XH, Tang JY, Kwan YW, Hoi PM, Lee SM (2018) Sodium tanshinone IIA sulfonate promotes endothelial integrity via regulating VE-cadherin dynamics and RhoA/ROCK-mediated cellular contractility and prevents atorvastatin-induced intracerebral hemorrhage in zebrafish. Toxicol Appl Pharmacol 350:32–42. https://doi.org/10.1016/j.taap.2018.04.037
Zhou ZY, Zhao WR, Zhang J, Chen XL, Tang JY (2019) Sodium tanshinone IIA sulfonate: A review of pharmacological activity and pharmacokinetics. Biomed Pharmacother 118:109362. https://doi.org/10.1016/j.biopha.2019.109362
Acknowledgements
This work was supported by Guangdong Provincial Key Laboratory of Research on Emergency in TCM (2017B030314176); The Natural Science Foundation of Guangdong (2018A0303130053, 2021A1515011478).
Author information
Authors and Affiliations
Contributions
Hui-Han Ma, Can Wan and Lu-Di Zhang conducted the experiment. Hui-Han Ma, Can Wan and Shi-Jie Zhang contributed to initial data analysis and interpretation, and drafted the initial manuscript. Rong-Rong Zhang, Dong Peng and Li-Jun Qiao helped revising the manuscript. Shi-Jie Zhang designed the study. Shi-Jie Zhang, Ye-Feng Cai and Hong-Qiang Huang supervised all the aspects of the study, critically reviewed and revised the manuscript, and approved the final manuscript as submitted.
Corresponding authors
Ethics declarations
Consent to participate and publication
All authors declare consent to participate and consent for publication.
Competing interests
The authors declare that they have no conflicts of interest.
Ethics approval
All of the animal experiments were approved by the Animal Ethics Committee of Guangzhou University of Chinese Medicine, in accordance with the guide for the animal experiments, clinical studies, and biodiversity rights (00,142,833).
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ma, HH., Wan, C., Zhang, LD. et al. Sodium tanshinone IIA sulfonate improves cognitive impairment via regulating Aβ transportation in AD transgenic mouse model. Metab Brain Dis 37, 989–1001 (2022). https://doi.org/10.1007/s11011-022-00911-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-00911-y