Abstract
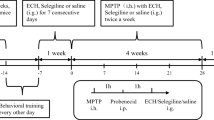
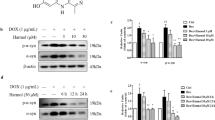
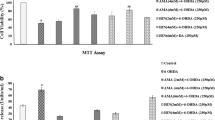
Cryptotanshinone (CTN) has shown its neuroprotective and anti-inflammatory qualities in non-genetic mouse model of Alzheimer’s disease. According to bioinformatics analysis, CTN and Signal Transducer and Activator of Transcription 3 (STAT3) may interact to form a drug-target network. This study was conducted to identify the role of CTN-STAT3 interaction in Parkinson’s disease (PD). PD model was established with MMP+-stimulated SH-SY5Y cells. After pre-treatment with CTN or co-treatment with CTN and STAT3 agonist, MTT assay was performed to observe cell viability; ELISA kit was used to measure the expression level of pro-inflammatory cytokines; DCFH-DA and corresponding assay kits were employed to determine the production of ROS, SOD, CAT and GSH-px; TUNEL assay and western blot were performed to detect cell apoptosis. STAT3 activity was also detected by western blot. Treatment with CTN alone had no impact on SH-SY5Y cell viability, but CTN pre-treatment effectively improved MPP+-induced loss of viability in SH-SY5Y cells. Moreover, pre-treatment with CTN inhibited MPP+-induced oxidative stress, apoptosis and STAT3 activity in SH-SY5Y cells, whereas this inhibitory effect was diminished after additional treatment with STAT3 agonist. CTN ameliorates MPP+-induced oxidative stress and apoptosis of SH-SY5Y neuroblastoma cells by inhibiting the expression of STAT3. Therefore, CTN could be a promising therapeutic agent, and STAT3 could be a potential target for PD treatment.





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Choi DJ, Kwon JK, Joe EH (2018) A Parkinson's disease gene, DJ-1, regulates astrogliosis through STAT3. Neurosci Lett 685:144–149. https://doi.org/10.1016/j.neulet.2018.08.025
De Virgilio A et al (2016) Parkinson's disease: autoimmunity and neuroinflammation. Autoimmun Rev 15:1005–1011. https://doi.org/10.1016/j.autrev.2016.07.022
Feng ST, Wang ZZ, Yuan YH, Wang XL, Sun HM, Chen NH, Zhang Y (2020) Dynamin-related protein 1: a protein critical for mitochondrial fission, mitophagy, and neuronal death in Parkinson's disease. Pharmacol Res 151:104553. https://doi.org/10.1016/j.phrs.2019.104553
Franco-Iborra S, Cuadros T, Parent A, Romero-Gimenez J, Vila M, Perier C (2018) Defective mitochondrial protein import contributes to complex I-induced mitochondrial dysfunction and neurodegeneration in Parkinson's disease. Cell Death Dis 9:1122. https://doi.org/10.1038/s41419-018-1154-0
Gelders G, Baekelandt V, Van der Perren A (2018) Linking Neuroinflammation and neurodegeneration in Parkinson's disease. J Immunol Res 2018:4784268. https://doi.org/10.1155/2018/4784268
Geng L, Liu W, Chen Y (2017) miR-124-3p attenuates MPP(+)-induced neuronal injury by targeting STAT3 in SH-SY5Y cells. Exp Biol Med (Maywood) 242:1757–1764. https://doi.org/10.1177/1535370217734492
Halliday GM et al (2005) Alpha-synuclein redistributes to neuromelanin lipid in the substantia nigra early in Parkinson's disease. Brain 128:2654–2664. https://doi.org/10.1093/brain/awh584
Han L, Xie YH, Wu R, Chen C, Zhang Y, Wang XP (2017) Traditional Chinese medicine for modern treatment of Parkinson's disease. Chin J Integr Med 23:635–640. https://doi.org/10.1007/s11655-016-2537-7
Hayes MT (2019) Parkinson's disease and parkinsonism. Am J Med 132:802–807. https://doi.org/10.1016/j.amjmed.2019.03.001
Huang J, Zeng F, Xu Q, Ma J (2020) Cryptotanshinone decreases granulosa cell apoptosis and restores ovarian function in mice with premature ovarian failure. Gen Physiol Biophys 39:277–283. https://doi.org/10.4149/gpb_2019059
Jin HJ, Li CG (2013) Tanshinone IIA and Cryptotanshinone prevent mitochondrial dysfunction in hypoxia-induced H9c2 cells: association to mitochondrial ROS, intracellular nitric oxide, and calcium levels Evid based complement. Alternat Med 2013:610694. https://doi.org/10.1155/2013/610694
Lee JE et al (2020) Neuroprotective effects of Cryptotanshinone in a direct reprogramming model of Parkinson's disease. Molecules 25. https://doi.org/10.3390/molecules25163602
Li S, Le W (2021) Parkinson's disease in traditional Chinese medicine. Lancet Neurol 20:262. https://doi.org/10.1016/S1474-4422(19)30224-8
Li J et al (2020a) Cryptotanshinone attenuates allergic airway inflammation through negative regulation of NF-kappaB and p38 MAPK. Biosci Biotechnol Biochem 84:268–278. https://doi.org/10.1080/09168451.2019.1687280
Li Y et al (2020b) Danshen formula granule and salvianic acid a alleviate ethanol-induced neurotoxicity. J Nat Med 74:399–408. https://doi.org/10.1007/s11418-019-01379-4
Liu Y, Wang H, Gao J, Wen Z, Peng L (2021) Cryptotanshinone ameliorates the pathogenicity of Streptococcus suis by targeting suilysin and inflammation. J Appl Microbiol 130:736–744. https://doi.org/10.1111/jam.14810
Ma H et al (2019) Echinacoside selectively rescues complex I inhibition-induced mitochondrial respiratory impairment via enhancing complex II activity. Neurochem Int 125:136–143. https://doi.org/10.1016/j.neuint.2019.02.012
Mahesh R, Jung HW, Kim GW, Kim YS, Park YK (2012) Cryptotanshinone from Salviae miltiorrhizae radix inhibits sodium-nitroprusside-induced apoptosis in neuro-2a cells. Phytother Res 26:1211–1219. https://doi.org/10.1002/ptr.3705
Maione F et al (2018) Down regulation of pro-inflammatory pathways by tanshinone IIA and cryptotanshinone in a non-genetic mouse model of Alzheimer's disease. Pharmacol Res 129:482–490. https://doi.org/10.1016/j.phrs.2017.11.018
O'Callaghan JP, Kelly KA, VanGilder RL, Sofroniew MV, Miller DB (2014) Early activation of STAT3 regulates reactive astrogliosis induced by diverse forms of neurotoxicity. PLoS One 9:e102003. https://doi.org/10.1371/journal.pone.0102003
Poewe W et al (2017) Parkinson disease Nat Rev Dis Primers 3:17013. https://doi.org/10.1038/nrdp.2017.13
Raza C, Anjum R, Shakeel NUA (2019) Parkinson's disease: mechanisms, translational models and management strategies. Life Sci 226:77–90. https://doi.org/10.1016/j.lfs.2019.03.057
Reeve A, Simcox E, Turnbull D (2014) Ageing and Parkinson's disease: why is advancing age the biggest risk factor? Ageing Res Rev 14:19–30. https://doi.org/10.1016/j.arr.2014.01.004
Reich SG, Savitt JM (2019) Parkinson's disease. Med Clin North Am 103:337–350. https://doi.org/10.1016/j.mcna.2018.10.014
Risiglione P et al (2020) High-resolution Respirometry reveals MPP(+) mitochondrial toxicity mechanism in a cellular model of Parkinson's disease. Int J Mol Sci 21. https://doi.org/10.3390/ijms21217809
Saraf R et al (2020) In silico modeling of the induction of apoptosis by Cryptotanshinone in osteosarcoma cell lines. IEEE/ACM Trans Comput Biol Bioinform. https://doi.org/10.1109/TCBB.2020.3037318
Schapira AH, Jenner P (2011) Etiology and pathogenesis of Parkinson's disease. Mov Disord 26:1049–1055. https://doi.org/10.1002/mds.23732
Shi G et al (2021) Effect of Cryptotanshinone on measures of rat cardiomyocyte oxidative stress and gene activation associated with apoptosis. Cardiorenal Med 11:18–26. https://doi.org/10.1159/000507184
Sriram K, Benkovic SA, Hebert MA, Miller DB, O'Callaghan JP (2004) Induction of gp130-related cytokines and activation of JAK2/STAT3 pathway in astrocytes precedes up-regulation of glial fibrillary acidic protein in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of neurodegeneration: key signaling pathway for astrogliosis in vivo? J Biol Chem 279:19936–19947. https://doi.org/10.1074/jbc.M309304200
Szeto JY, Lewis SJ (2016) Current treatment options for Alzheimer's disease and Parkinson's disease dementia. Curr Neuropharmacol 14:326–338. https://doi.org/10.2174/1570159x14666151208112754
Vasta R et al (2017) Side effects induced by the acute levodopa challenge in Parkinson's disease and atypical parkinsonisms. PLoS One 12:e0172145. https://doi.org/10.1371/journal.pone.0172145
Wang N, Dong X, Shi D, Li N, Zhang Q (2020a) Cryptotanshinone ameliorates placental oxidative stress and inflammation in mice with gestational diabetes mellitus. Arch Pharm Res 43:755–764. https://doi.org/10.1007/s12272-020-01242-1
Wang T et al (2020b) Neuroprotective effects of Danshensu on rotenone-induced Parkinson's disease models in vitro and in vivo BMC complement. Med Ther 20:20. https://doi.org/10.1186/s12906-019-2738-7
Wu YH, Wu YR, Li B, Yan ZY (2020) Cryptotanshinone: a review of its pharmacology activities and molecular mechanisms. Fitoterapia 145:104633. https://doi.org/10.1016/j.fitote.2020.104633
Xicoy H, Brouwers JF, Kalnytska O, Wieringa B, Martens GJM (2020) Lipid analysis of the 6-Hydroxydopamine-treated SH-SY5Y cell model for Parkinson's disease. Mol Neurobiol 57:848–859. https://doi.org/10.1007/s12035-019-01733-3
Yuen CW, Murugaiyah V, Najimudin N, Azzam G (2021) Danshen (Salvia miltiorrhiza) water extract shows potential neuroprotective effects in Caenorhabditis elegans. J Ethnopharmacol 266:113418. https://doi.org/10.1016/j.jep.2020.113418
Zhang Y et al (2019) Cryptotanshinone protects against pulmonary fibrosis through inhibiting Smad and STAT3 signaling pathways. Pharmacol Res 147:104307. https://doi.org/10.1016/j.phrs.2019.104307
Zhang Y et al (2020) Cryptotanshinone ameliorates cardiac injury and cardiomyocyte apoptosis in rats with coronary microembolization. Drug Dev Res. https://doi.org/10.1002/ddr.21777
Zhao H et al (2019) Cryptotanshinone attenuates oxygen-glucose deprivation/ recovery-induced injury in an in vitro model of neurovascular unit. Front Neurol 10:381. https://doi.org/10.3389/fneur.2019.00381
Zhou Y, Wang X, Ying W, Wu D, Zhong P (2019) Cryptotanshinone attenuates inflammatory response of microglial cells via the Nrf2/HO-1 pathway. Front Neurosci 13:852. https://doi.org/10.3389/fnins.2019.00852
Zhu R, Wang W, Yang S (2019) Cryptotanshinone inhibits hypoxia/reoxygenation-induced oxidative stress and apoptosis in renal tubular epithelial cells. J Cell Biochem 120:13354–13360. https://doi.org/10.1002/jcb.28609
Funding
This work was supported by the Class A second-class scientific research project of the Health Commission of Inner Mongolia Autonomous Region (Grant No. 201302134).
Author information
Authors and Affiliations
Contributions
QW and YL designed the research, performed the experiments, drafted and revised the manuscript. QW searched the literature and analyzed the data. YL guided the experiments. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Q., Liu, Y. Cryptotanshinone ameliorates MPP+-induced oxidative stress and apoptosis of SH-SY5Y neuroblastoma cells: the role of STAT3 in Parkinson’s disease. Metab Brain Dis 37, 1477–1485 (2022). https://doi.org/10.1007/s11011-022-00905-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-00905-w