Abstract
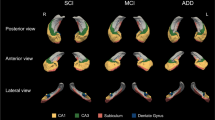
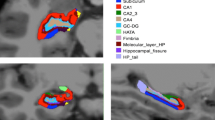
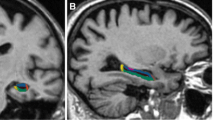
Hippocampal brain regions are strongly implicated in Niemann Pick type C disease (NPC), but little is known regarding distinct subregions of the hippocampal complex and whether these are equally or differentially affected. To address this gap, we compared volumes of five hippocampal subfields between NPC and healthy individuals using MRI. To this end, 9 adult-onset NPC cases and 9 age- and gender-matched controls underwent a 3 T T1-weighted MRI scan. Gray matter volumes of the cornu ammonis (CA1, CA2 and CA3), dentate gyrus (DG), subiculum, entorhinal cortex and hippocampal-amygdalar transition area were calculated by integrating MRI-based image intensities with microscopically defined cytoarchitectonic probabilities. Compared to healthy controls, NPC patients showed smaller volumes of the CA1-3 and DG regions bilaterally, with the greatest difference localized to the left DG (Cohen’s d = 1.993, p = 0.008). No significant associations were shown between hippocampal subfield volumes and key clinical features of NPC, including disease duration, symptom severity and psychosis. The pattern of hippocampal subregional atrophy in NPC differs from those seen in other dementias, which may indicate unique cytoarchitectural vulnerabilities in this earlier-onset disorder. Future MRI studies of hippocampal subfields may clarify its potential as a biomarker of neurodegeneration in NPC.


Similar content being viewed by others
Data availability
The generated and analyzed data during this study are available from the corresponding author on reasonable request.
References
Amunts K et al (2005) Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: intersubject variability and probability maps. Anat Embryol (berl) 210:343–352. https://doi.org/10.1007/s00429-005-0025-5
Ashburner J (2007) A fast diffeomorphic image registration algorithm. Neuroimage 38:95–113. https://doi.org/10.1016/j.neuroimage.2007.07.007
Ashburner J, Friston KJ (2000) Voxel-based morphometry–the methods. Neuroimage 11:805–821. https://doi.org/10.1006/nimg.2000.0582
Bartsch T et al (2015) Selective neuronal vulnerability of human hippocampal CA1 neurons: lesion evolution, temporal course, and pattern of hippocampal damage in diffusion-weighted MR imaging. J Cereb Blood Flow Metab 35:1836–1845. https://doi.org/10.1038/jcbfm.2015.137
Benjamini Y, Yekutieli D (2001) The control of the false discovery rate in multiple testing under dependency. Ann Statist 29:1165–1188. https://doi.org/10.1214/aos/1013699998
Bonnot O, Klünemann H-H, Velten C, Torres Martin JV, Walterfang M (2019) Systematic review of psychiatric signs in Niemann-Pick disease type C. World J Biol Psychiatry 20:320–332. https://doi.org/10.1080/15622975.2018.1441548
Bora E et al (2011) Neuroanatomical abnormalities in schizophrenia: a multimodal voxelwise meta-analysis and meta-regression analysis. Schizophr Res 127:46–57. https://doi.org/10.1016/j.schres.2010.12.020
Braak H, Alafuzoff I, Arzberger T, Kretzschmar H, Del Tredici K (2006) Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol 112:389–404. https://doi.org/10.1007/s00401-006-0127-z
Bu B, Li J, Davies P, Vincent I (2002) Deregulation of cdk5, hyperphosphorylation, and cytoskeletal pathology in the Niemann-Pick type C murine model. J Neurosci 22:6515–6525
Byun K et al (2011) Alteration of the CNS pathway to the hippocampus in a mouse model of Niemann-Pick, type C disease. J Chem Neuroanat 42:39–44. https://doi.org/10.1016/j.jchemneu.2011.04.003
Causevic M et al (2018) BACE1-cleavage of Sez6 and Sez6L is elevated in Niemann-Pick type C disease mouse brains. PLoS ONE 13:e0200344. https://doi.org/10.1371/journal.pone.0200344
Distl R, Treiber-Held S, Albert F, Meske V, Harzer K, Ohm TG (2003) Cholesterol storage and tau pathology in Niemann-Pick type C disease in the brain. J Pathol 200:104–111. https://doi.org/10.1002/path.1320
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K, Zilles K (2005) A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage 25:1325–1335. https://doi.org/10.1016/j.neuroimage.2004.12.034
Eratne D et al (2019) Cerebrospinal fluid neurofilament light chain is elevated in Niemann–Pick type C compared to psychiatric disorders and healthy controls and may be a marker of treatment response Australian & New Zealand. J Psychiatry:0004867419893431. https://doi.org/10.1177/0004867419893431
Evans TE et al (2018) Subregional volumes of the hippocampus in relation to cognitive function and risk of dementia. Neuroimage 178:129–135. https://doi.org/10.1016/j.neuroimage.2018.05.041
First M et al (2002) Structured clinical interview for DSM-IV-TR axis I disorders, research version, patient edition. SCID-I/P, New York, pp 94-1
Geberhiwot T et al (2018) Consensus clinical management guidelines for Niemann-Pick disease type C. Orphanet J Rare Dis 13:50. https://doi.org/10.1186/s13023-018-0785-7
Good CD, Johnsrude IS, Ashburner J, Henson RN, Friston KJ, Frackowiak RS (2001) A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage 14:21–36. https://doi.org/10.1006/nimg.2001.0786
Heitz C, Epelbaum S, Nadjar Y (2017) Cognitive impairment profile in adult patients with Niemann pick type C disease. Orphanet J Rare Dis 12:166. https://doi.org/10.1186/s13023-017-0714-1
Henneman WJP et al (2009) Baseline CSF p-tau levels independently predict progression of hippocampal atrophy in Alzheimer disease. Neurology 73:935–940. https://doi.org/10.1212/WNL.0b013e3181b879ac
Hochberg Y, Benjamini Y (1990) More powerful procedures for multiple significance testing. Stat Med 9:811–818. https://doi.org/10.1002/sim.4780090710
Iturriaga C, Pineda M, Fernandez-Valero EM, Vanier MT, Coll MJ (2006) Niemann-Pick C disease in Spain: clinical spectrum and development of a disability scale. J Neurol Sci 249:1–6. https://doi.org/10.1016/j.jns.2006.05.054
Kesner RP (2018) An analysis of dentate gyrus function (an update). Behav Brain Res 354:84–91. https://doi.org/10.1016/j.bbr.2017.07.033
Kurth F, Cherbuin N, Luders E (2015a) Reduced age-related degeneration of the hippocampal subiculum in long-term meditators. Psychiatry Res 232:214–218. https://doi.org/10.1016/j.pscychresns.2015.03.008
Kurth F, Luders E, Gaser C (2015b) Voxel-based Morphometry. In: Toga A (ed) Brain mapping: an encyclopedic reference, vol 1. Academic Press, London, pp 345–349
Kurth F, Cherbuin N, Luders E (2017a) The impact of aging on subregions of the hippocampal complex in healthy adults. NeuroImage 163.https://doi.org/10.1016/j.neuroimage.2017.09.016
Kurth F, Cherbuin N, Luders E (2017b) The impact of aging on subregions of the hippocampal complex in healthy adults. Neuroimage 163:296–300. https://doi.org/10.1016/j.neuroimage.2017.09.016
Kurth F, Jancke L, Luders E (2017c) Sexual dimorphism of Broca’s region: More gray matter in female brains in Brodmann areas 44 and 45. J Neurosci Res 95:626–632. https://doi.org/10.1002/jnr.23898
Kurth F, Cherbuin N, Luders E (2018a) Age but no sex effects on subareas of the amygdala. Hum Brain Mapp. https://doi.org/10.1002/hbm.24481
Kurth F, Jancke L, Luders E (2018b) Integrating cytoarchitectonic probabilities with MRI-based signal intensities to calculate regional volumes of interest. In: Spalletta G, Piras F, Gili T (eds) Brain morphometry. Neuromethods, vol 136. Humana Press, New York. https://doi.org/10.1007/978-1-4939-7647-8_8
Low A, Foo H, Yong TT, Tan LCS, Kandiah N (2019) Hippocampal subfield atrophy of CA1 and subicular structures predict progression to dementia in idiopathic Parkinson’s disease. J Neurol Neurosurg Psychiatry 90:681–687. https://doi.org/10.1136/jnnp-2018-319592
Luders E, Kurth F, Toga AW, Narr KL, Gaser C (2013) Meditation effects within the hippocampal complex revealed by voxel-based morphometry and cytoarchitectonic probabilistic mapping. Front Psychol 4:398. https://doi.org/10.3389/fpsyg.2013.00398
Mak E et al (2019) Beta amyloid deposition maps onto hippocampal and subiculum atrophy in dementia with Lewy bodies. Neurobiol Aging 73:74–81. https://doi.org/10.1016/j.neurobiolaging.2018.09.004
Mattsson N et al (2012) Amyloid-beta metabolism in Niemann-Pick C disease models and patients. Metab Brain Dis 27:573–585. https://doi.org/10.1007/s11011-012-9332-8
Muller-Ehrenberg L, Riphagen JM, Verhey FRJ, Sack AT, Jacobs HIL (2018) Alzheimer’s disease biomarkers have distinct associations with specific hippocampal subfield volumes. J Alzheimers Dis 66:811–823. https://doi.org/10.3233/jad-180676
Nakahara S, Matsumoto M, van Erp TGM (2018) Hippocampal subregion abnormalities in schizophrenia: a systematic review of structural and physiological imaging studies. Neuropsychopharmacology Reports 38:156–166. https://doi.org/10.1002/npr2.12031
Patterson M (1993) Niemann-Pick Disease Type C. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Stephens K, Amemiya A (eds) GeneReviews(®). University of Washington, Seattle Copyright © 1993–2020, University of Washington, Seattle. GeneReviews is a registered trademark of the University of Washington, Seattle. All rights reserved., Seattle
Rego T et al (2019) Psychiatric and cognitive symptoms associated with Niemann-Pick type C disease: neurobiology and management. CNS Drugs 33:125–142. https://doi.org/10.1007/s40263-018-0599-0
Sevin M, Lesca G, Baumann N, Millat G, Lyon-Caen O, Vanier MT, Sedel F (2007) The adult form of Niemann-Pick disease type C. Brain 130:120–133. https://doi.org/10.1093/brain/awl260
Stampfer M et al (2013) Niemann-Pick disease type C clinical database: cognitive and coordination deficits are early disease indicators. Orphanet J Rare Dis 8:35. https://doi.org/10.1186/1750-1172-8-35
Stav AL et al (2016) Hippocampal subfield atrophy in relation to cerebrospinal fluid biomarkers and cognition in early Parkinson’s disease: a cross-sectional study. NPJ Parkinsons Dis 2:15030. https://doi.org/10.1038/npjparkd.2015.30
Steve TA, Jirsch JD, Gross DW (2014) Quantification of subfield pathology in hippocampal sclerosis: A systematic review and meta-analysis. Epilepsy Res 108:1279–1285. https://doi.org/10.1016/j.eplepsyres.2014.07.003
Tavitian A, Song W, Schipper HM (2019) Dentate Gyrus Immaturity in Schizophrenia. Neuroscientist 25:528–547. https://doi.org/10.1177/1073858418824072
Treiber-Held S, Distl R, Meske V, Albert F, Ohm TG (2003) Spatial and temporal distribution of intracellular free cholesterol in brains of a Niemann-Pick type C mouse model showing hyperphosphorylated tau protein. Implications for Alzheimer’s disease. J Pathol 200:95–103. https://doi.org/10.1002/path.1345
Villemagne VL, Velakoulis D, Dore V, Bozinoski S, Masters CL, Rowe CC, Walterfang M (2019) Imaging of tau deposits in adults with Niemann-Pick type C disease: a case-control study. Eur J Nucl Med Mol Imaging 46:1132–1138. https://doi.org/10.1007/s00259-019-4273-7
Walterfang M et al (2010) White and gray matter alterations in adults with Niemann-Pick disease type C: a cross-sectional study. Neurology 75:49–56. https://doi.org/10.1212/WNL.0b013e3181e6210e
Walterfang M et al (2011) Size and shape of the corpus callosum in adult Niemann-Pick type C reflects state and trait illness variables. AJNR Am J Neuroradiol 32:1340–1346. https://doi.org/10.3174/ajnr.A2490
Walterfang M et al (2013) Subcortical volumetric reductions in adult Niemann-Pick Disease Type C: a cross-sectional study. Am J Neuroradiol 34:1334. https://doi.org/10.3174/ajnr.A3356
Walterfang M et al (2020) Imaging of neuroinflammation in adult Niemann-Pick type C disease: A cross-sectional study. Neurology 94:e1716–e1725. https://doi.org/10.1212/wnl.0000000000009287
Zervas M, Dobrenis K, Walkley SU (2001) Neurons in Niemann-Pick disease type C accumulate gangliosides as well as unesterified cholesterol and undergo dendritic and axonal alterations. J Neuropathol Exp Neurol 60:49–64. https://doi.org/10.1093/jnen/60.1.49
Zhang M, Wang X, Jiang F, Wang W, Vincent I, Bu B (2010) Mitotic Epitopes are Incorporated into Age-dependent Neurofibrillary Tangles in Niemann-Pick Disease Type C. Brain Pathol 20:367–377. https://doi.org/10.1111/j.1750-3639.2009.00286.x
Zhou S, Davidson C, McGlynn R, Stephney G, Dobrenis K, Vanier MT, Walkley SU (2011) Endosomal/lysosomal processing of gangliosides affects neuronal cholesterol sequestration in Niemann-Pick disease type C. Am J Pathol 179:890–902. https://doi.org/10.1016/j.ajpath.2011.04.017
Funding
The authors declared no specific grant for this research from any funding agency in public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
PW was involved in the conception and coordination of the study, as well as interpretation of the data. MW led the conception and coordination of the study. Image pre-processing and statistical analyses were conducted by EL and FK. PW led the manuscript preparation and writing of first draft along with EL, FK and MW. VC, MDB, CP and DV were involved in the conception, design and execution of the study, interpretation of data, review and critique of the draft. All authors approved of the manuscript’s final version.
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the Austin Health and Melbourne Health ethics committees (approval number 2012.066), Victoria, Australia, and with the Australian National Statement on Ethical Conduct in Human Research (2007).
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Conflicts of interest
None to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wibawa, P., Kurth, F., Luders, E. et al. Differential involvement of hippocampal subfields in Niemann-Pick type C disease: a case–control study. Metab Brain Dis 36, 2071–2078 (2021). https://doi.org/10.1007/s11011-021-00782-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00782-9