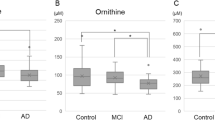
Abstract
Mild cognitive impairment (MCI) is transition phase between cognitive decline and dementia. The current study aims to investigate altered metabolic pattern in plasma of MCI for potential biomarkers. MCI (N = 50) and healthy controls (HC, N = 50) age group 55–75 years were screened based on Mini Mental State Examination Test (MMSE) and diffusion tensor imaging (DTI imaging). The MMSE score of MCI was significantly lower (25.74 ± 1.83) compared to healthy control subjects (29 ± 1). The MCI patients exhibit significant changes in white matter integrity in the right frontal lobe, right temporal lobe, left frontal lobe, forcep major, fornix, corpus callosum. Further, the plasma samples of twenty seven MCI patients (N = 27) and twenty HC subjects (N = 20; having no significant differences in any demographics) were analyzed using 1H NMR based metabolomics approach. Consistent with many previous reports, the levels of several plasma metabolites were found to be elevated in MCI patients compared to healthy controls. Further univariate and multivariate ROC curve analyses provided three plasma metabolites as a diagnostic panel of biomarker for MCI; which are lysine, glycine, and glutamine. Overall, the results of this study will help to improve the diagnostic and prognostic strategies of MCI in addition to improving our understanding about disease pathogenesis. We believe that the over-nutritional metabolic phenotype of MCI needs to be targeted for developing future dietary interventions so that the progression of MCI can be limited.
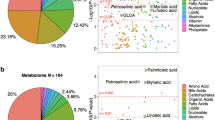
Graphical abstract
Metabolic derangements associated with Mild Cognitive Impairment





Similar content being viewed by others
Data availability
All relevant data are within the manuscript and its Supporting Information files. The raw NMR spectral data will be provided on request.
Abbreviations
- AD:
-
Alzheimer’s disease.
- HC:
-
Healthy control.
- NMR:
-
Nuclear Magnetic Resonance.
- MCI:
-
Mild Cognitive Impairment.
- ROC:
-
Receiver operating characteristic curve.
- VIP:
-
Variable importance for the projection.
- APOE:
-
apolipoprotein E.
- CSF:
-
Cerebrospinal fluid.
- AUROC:
-
Area under ROC curve.
- ESM:
-
Electronic Supplementary Material.
- CI:
-
Confidence interval.
- TCA:
-
Tricarboxylic acid.
- PUFAs:
-
Polyunsaturated fatty acids.
- NAG:
-
N-acetyl-glycoproteins.
- FA:
-
Fractional Anisotropy.
- MMSE:
-
Mini-Mental State Examination.
- DTI:
-
Diffusion tensor imaging.
- GPC:
-
Glycerophosphocholine.
- LDL:
-
Low-density lipoproteins.
- VLDL:
-
Very Low-density lipoproteins.
- CPMG:
-
Carr–Purcell–Meiboom–Gill.
- PCA:
-
Principal component analysis.
- PLS-DA:
-
Projection to least-squares discriminant analysis.
- OPLS-DA:
-
PLS-DA with Orthogonal Signal Correction(OSC).
- PTR:
-
Phenylalanine to tyrosine ratio.
- HTR:
-
Histidine to tyrosine ratio.
- EQR:
-
Glutamate to Glutamine ratio
- BTR:
-
Branched-chain amino acid to Tyrosine ratio.
- LPR:
-
Lactate to Pyruvate ratio.
- CT:
-
Computerized tomography.
- GPC:
-
Glycerophosphocholine.
- OSC:
-
Orthogonal Signal Correction.
- PS-1/PS-2:
-
Presenilin-1/Presenilin-2.
- ADC:
-
Apparent diffusion coefficient.
- 1D/2D:
-
One/two dimensional.
- TCA:
-
Tricarboxylic acid.
References
Barthel H, Gertz HJ, Dresel S, Peters O, Bartenstein P, Buerger K, Hiemeyer F, Wittemer-Rump SM, Seibyl J, Reininger C (2011) Cerebral amyloid-β PET with florbetaben (18F) in patients with Alzheimer's disease and healthy controls: a multicentre phase 2 diagnostic study. Lancet Neurol 10:424–435
Cerasuolo J, Izzo A (2017) Persistent impairment in working memory following severe hyperglycemia in newly diagnosed type 2 diabetes. Endocrinology, diabetes & metabolism case reports 2017
Chai G-S, Jiang X, Ni Z-F, Ma Z-W, Xie A-J, Cheng X-S, Wang Q, Wang J-Z, Liu G-P (2013) Betaine attenuates Alzheimer-like pathological changes and memory deficits induced by homocysteine. J Neurochem 124:388–396
Chang CH, Lin CH, Lane HY (2020) d-glutamate and Gut Microbiota in Alzheimer's Disease. Int J Mol Sci 21:2676
Charlton M (2006) Branched-chain amino acid enriched supplements as therapy for liver disease. J Nutr 136:295S–298S
Chong J, Soufan O, Li C, Caraus I, Li S, Bourque G, Wishart DS, Xia J (2018) MetaboAnalyst 4.0: towards more transparent and integrative metabolomics analysis. Nucleic Acids Res 46:W486–W494
Chong J, Wishart DS, Xia J (2019) Using MetaboAnalyst 4.0 for comprehensive and integrative metabolomics data analysis. Current protocols in bioinformatics (68): e86
Chu H, Zhang A, Han Y, Lu S, Kong L, Han J, Liu Z, Sun H, Wang X (2016) Metabolomics approach to explore the effects of Kai-Xin-san on Alzheimer's disease using UPLC/ESI-Q-TOF mass spectrometry. J Chromatogr B 1015:50–61
Ciavardelli D, Piras F, Consalvo A, Rossi C, Zucchelli M, Di Ilio C, Frazzini V, Caltagirone C, Spalletta G, Sensi SL (2016) Medium-chain plasma acylcarnitines, ketone levels, cognition, and gray matter volumes in healthy elderly, mildly cognitively impaired, or Alzheimer's disease subjects. Neurobiol Aging 43:1–12
Donohue MC, Moghadam SH, Roe AD, Sun CK, Edland SD, Thomas RG, Petersen RC, Sano M, Galasko D, Aisen PS (2015) Longitudinal plasma amyloid beta in Alzheimer's disease clinical trials. Alzheimers Dement 11:1069–1079
Dranca F, Oroian M (2019) Ultrasound-assisted extraction of pectin from Malus domestica ΓÇÿF-âlticeniΓÇÖApple Pomace. Processes 7:488
Drobny A, Meloh H, Wåchtershäuser E, Hellmann B, Mueller A S, van der Klis J D, Fitzenberger E, Wenzel U (2019) Betaine-rich sugar beet molasses protects from homocysteine-induced reduction of survival in Caenorhabditis elegans. Eur J Nutr 1–8
Enns GM (2008) Neurologic damage and neurocognitive dysfunction in urea cycle disorders. Semin Pediatr Neurol 15:132–139
Fielding RA, Gunstad J, Gustafson DR, Heymsfield SB, Launer LJ, Kral JG, Penninger J, Phillips DI, Scarmeas N (2013) The paradox of overnutrition in aging and cognition. Ann N Y Acad Sci 1287:31–43
Gong Y, Liu Y, Zhou L, Di X, Li W, Li Q, Bi K (2015) A UHPLC-TOF/MS method based metabonomic study of total ginsenosides effects on Alzheimer disease mouse model. J Pharm Biomed Anal 115:174–182
Gonzáílez-Domìnguez R, Garcia-Barrera T, Gòmez-Ariza JL (2014) Using direct infusion mass spectrometry for serum metabolomics in Alzheimer's disease. Anal Bioanal Chem 406:7137–7148
González-Domìnguez R, García A, Garcia-Barrera T, Barbas C, Gòmez-Ariza JL (2014a) Metabolomic profiling of serum in the progression of Alzheimer's disease by capillary electrophoresis-mass spectrometry. Electrophoresis 35:3321–3330
Gonzalez-Dominguez R, Garcia-Barrera T, Gòmez-Ariza JL (2015a) Application of a novel metabolomic approach based on atmospheric pressure photoionization mass spectrometry using flow injection analysis for the study of Alzheimer's disease. Talanta 131:480–489
González-Domìnguez R, Garcia-Barrera T, Gòmez-Ariza JL (2014b) Metabolomic study of lipids in serum for biomarker discovery in Alzheimer's disease using direct infusion mass spectrometry. J Pharm Biomed Anal 98:321–326
González-Domìnguez R, Garcia-Barrera T, Gòmez-Ariza JL (2015a) Metabolite profiling for the identification of altered metabolic pathways in Alzheimer's disease. J Pharm Biomed Anal 107:75–81
González-Domìnguez R, Garcìa-Barrera T, Gòmez-Ariza JL (2015b) Metabolite profiling for the identification of altered metabolic pathways in Alzheimer's disease. J Pharm Biomed Anal 107:75–81
Gonzalez-Dominguez R, Garcia-Barrera T, Vitorica J, Gòmez-Ariza JL (2015b) Metabolomic research on the role of interleukin-4 in Alzheimer's disease. Metabolomics 11:1175–1183
González-Domìnguez R, Garcia-Barrera T, Vitorica J, Gòmez-Ariza JL (2015c) Application of metabolomics based on direct mass spectrometry analysis for the elucidation of altered metabolic pathways in serum from the APP/PS1 transgenic model of Alzheimer's disease. J Pharm Biomed Anal 107:378–385
Gonzalez-Dominguez R, Javier Ruperez F, Garcia-Barrera T, Barbas C, Luis G-AJ (2016) Metabolomic-driven elucidation of serum disturbances associated with Alzheimer's disease and mild cognitive impairment. Curr Alzheimer Res 13:641–653
Graham SF, Chevallier OP, Elliott CT, Hölscher C, Johnston J, McGuinness B, Kehoe PG, Passmore AP, Green BD (2015) Untargeted metabolomic analysis of human plasma indicates differentially affected polyamine and L-arginine metabolism in mild cognitive impairment subjects converting to Alzheimer's disease. PloS one 10:e0119452
Graham SF, Holscher C, McClean P, Elliott CT, Green BD (2013) 1 H NMR metabolomics investigation of an Alzheimer's disease (AD) mouse model pinpoints important biochemical disturbances in brain and plasma. Metabolomics 9:974–983
Griffin JW, Bradshaw PC (2017) Amino acid catabolism in Alzheimer's disease brain: friend or foe? Oxidative medicine and cellular longevity 2017
Guleria A, Kumar A, Kumar U, Raj R, Kumar D (2018) NMR based metabolomics: an exquisite and facile method for evaluating therapeutic efficacy and screening drug toxicity. Curr Top Med Chem 18:1827–1849
Guleria A, Pratap A, Dubey D, Rawat A, Chaurasia S, Sukesh E, Phatak S, Ajmani S, Kumar U, Khetrapal CL (2016) NMR based serum metabolomics reveals a distinctive signature in patients with lupus nephritis. Sci Rep 6:35309
He Q, Li Q, Zhao J, Wu T, Ji L, Huang G, Ma F (2016) Relationship between plasma lipids and mild cognitive impairment in the elderly Chinese: a case-control study. Lipids Health Dis 15:146
Hu ZP, Browne ER, Liu T, Angel TE, Ho PC, Chan ECY (2012) Metabonomic profiling of TASTPM transgenic Alzheimer's disease mouse model. J Proteome Res 11:5903–5913
Inoue S, Kanda T, Imamura M, Quan GX, Kojima K, Tanaka H, Tomita M, Hino R, Yoshizato K, Mizuno S (2005) A fibroin secretion-deficient silkworm mutant, Nd-sD, provides an efficient system for producing recombinant proteins. Insect Biochem Mol Biol 35:51–59
Irizarry MC (2004) Biomarkers of Alzheimer disease in plasma. NeuroRx 1:226–234
Jiang N, Yan X, Zhou W, Zhang Q, Chen H, Zhang Y, Zhang X (2008) NMR-based metabonomic investigations into the metabolic profile of the senescence-accelerated mouse. J Proteome Res 7:3678–3686
Kaddurah-Daouk R, Zhu H, Sharma S, Bogdanov M, Rozen SG, Matson W, Oki NO, Motsinger-Reif AA, Churchill E, Lei Z (2013) Alterations in metabolic pathways and networks in Alzheimer's disease. Transl Psychiatry 3:e244
Kantarci K, Jack CR Jr, Xu YC, Campeau NG, ÒBrien PC, Smith GE, Ivnik RJ, Boeve BF, Kokmen E, Tangalos EG (2001) Mild cognitive impairment and Alzheimer disease: regional diffusivity of water. Radiology 219:101–107
Kim E, Jung YS, Kim H, Kim JS, Park M, Jeong J, Lee SK, Yoon HG, Hwang GS, Namkoong K (2014) Metabolomic signatures in peripheral blood associated with Alzheimer's disease amyloid-induced neuroinflammation. J Alzheimers Dis 42:421–433
Kumar U, Jain A, Guleria A, Misra DP, Goel R, Danda D, Misra R, Kumar D (2020) Circulatory glutamine/glucose ratio for evaluating disease activity in Takayasu arteritis: a NMR based serum metabolomics study. J Pharm Biomed Anal 180:113080
Li N, Zhou L, Li W, Liu Y, Wang J, He P (2015) Protective effects of ginsenosides Rg1 and Rb1 on an Alzheimer's disease mouse model: a metabolomics study. J Chromatogr B 985:54–61
Liang Q, Liu H, Zhang T, Jiang Y, Xing H, Zhang AH (2016) Discovery of serum metabolites for diagnosis of progression of mild cognitive impairment to Alzheimer's disease using an optimized metabolomics method. RSC advances 6:3586–3591
Lin C-N, Huang C-C, Huang K-L, Lin K-J, Yen T-C, Kuo H-C (2019) A metabolomic approach to identifying biomarkers in blood of Alzheimer's disease. Annals clinic trans neurol 6:537–545
Madeira C, Vargas-Lopes C, Brandão CO, Reis T, Laks J, Panizzutti R, Ferreira ST (2018) Elevated glutamate and glutamine levels in the cerebrospinal fluid of patients with probable Alzheimer's disease and depression. Front psych 9:561
Mapstone M, Cheema AK, Fiandaca MS, Zhong X, Mhyre TR, MacArthur LH, Hall WJ, Fisher SG, Peterson DR, Haley JM (2014a) Plasma phospholipids identify antecedent memory impairment in older adults. Nat Med 20:415–418
Mapstone M, Cheema AK, Fiandaca MS, Zhong X, Mhyre TR, MacArthur LH, Hall WJ, Fisher SG, Peterson DR, Haley JM (2014b) Plasma phospholipids identify antecedent memory impairment in older adults. Nat Med 20:415–418
Marjanska M, Curran GL, Wengenack TM, Henry PG, Bliss RL, Poduslo JF, Jack CR, U-ƒurbil K +, Garwood M (2005) Monitoring disease progression in transgenic mouse models of Alzheimer's disease with proton magnetic resonance spectroscopy. Proc Natl Acad Sci 102:11906–11910
Oskouie AA, Yekta RF, Tavirani MR, Kashani MS, Goshadrou F (2018) Lavandula angustifolia effects on rat models of Alzheimer's disease through the investigation of serum metabolic features using NMR metabolomics. Avicenna j med biotechnol 10:83
Petersen RC, Doody R, Kurz A, Mohs RC, Morris JC, Rabins PV, Ritchie K, Rossor M, Thal L, Winblad B (2001) Current concepts in mild cognitive impairment. Arch Neurol 58:1985–1992
Rawlings AM, Sharrett AR, Albert MS, Coresh J, Windham BG, Power MC, Knopman DS, Walker K, Burgard S, Mosley TH (2019) The association of late-life diabetes status and hyperglycemia with incident mild cognitive impairment and dementia: the ARIC study. Diabetes Care 42:1248–1254
Reisberg B, Doody R, Stöffler A, Schmitt F, Ferris S, Möbius HJ (2003) Memantine in moderate-to-severe Alzheimer's disease. N Engl J Med 348:1333–1341
Salek RM, Xia J, Innes A, Sweatman BC, Adalbert R, Randle S, McGowan E, Emson PC, Griffin JL (2010) A metabolomic study of the CRND8 transgenic mouse model of Alzheimer's disease. Neurochem Int 56:937–947
Scheltens P, Blennow K, Breteler MM, de Strooper B, Frisoni GB, Salloway S, Van der Flier WM (2016) Alzheimer-disease. Lancet 388:505–517
Schneider LS, Dagerman KS, Higgins JP, McShane R (2011) Lack of evidence for the efficacy of memantine in mild Alzheimer disease. Arch Neurol 68:991–998
Serkova NJ, Niemann CU (2006) Pattern recognition and biomarker validation using quantitative 1H-NMR-based metabolomics. Expert Rev Mol Diagn 6:717–731
Shea TB, Rogers E, Remington R (2012) Nutrition and dementia: are we asking the right questions? J Alzheimers Dis 30:27–33
Stebbins GT, Murphy CM (2009) Diffusion tensor imaging in Alzheimer's disease and mild cognitive impairment. Behav Neurol 21:39–49
Tajiri K, Shimizu Y (2013) Branched-chain amino acids in liver diseases. World J Gastroenterol: WJG 19:7620–7629
Trushina E, Dutta T, Persson XM, Mielke MM, Petersen RC (2013) Identification of altered metabolic pathways in plasma and CSF in mild cognitive impairment and Alzheimer's disease using metabolomics. PLoS One 8:e63644
Trygg J, Wold S (2002) Orthogonal projections to latent structures (O-PLS). J Chem: J Chem Soc 16:119–128
Tsuruoka M, Hara J, Hirayama A, Sugimoto M, Soga T, Shankle WR, Tomita M (2013) Capillary electrophoresis-mass spectrometry-based metabolome analysis of serum and saliva from neurodegenerative dementia patients. Electrophoresis 34:2865–2872
van der Lee SJ, Teunissen CE, Pool R, Shipley MJ, Teumer A, Chouraki V, van Lent DM, Tynkkynen J, Fischer K, Hernesniemi J (2018) Circulating metabolites and general cognitive ability and dementia: evidence from 11 cohort studies. Alzheimers Dement 14:707–722
Von Arnim CA, Gola U, Biesalski HK (2010) More than the sum of its parts? Nutrition in Alzheimer's disease. Nutrition 26:694–700
Walton HS, Dodd PR (2007) Glutamate-glutamine cycling in Alzheimer's disease. Neurochem Int 50:1052–1066
Wang G, Zhou Y, Huang FJ, Tang HD, Xu XH, Liu JJ, Wang Y, Deng YL, Ren RJ, Xu W (2014) Plasma metabolite profiles of Alzheimer's disease and mild cognitive impairment. J Proteome Res 13:2649–2658
Wu J, Fu B, Lei H, Tang H, Wang Y (2016) Gender differences of peripheral plasma and liver metabolic profiling in app/Ps1 transgenic ad mice. Neuroscience 332:160–169
Xu WL, Atti AR, Gatz M, Pedersen NL, Johansson B, Fratiglioni L (2011) Midlife overweight and obesity increase late-life dementia risk: a population-based twin study. Neurology 76:1568–1574
Acknowledgments
Authors would also like to acknowledge the Department of Medical Education, Govt. of Uttar Pradesh for supporting the High Field NMR Facility at Centre of Biomedical Research, Lucknow, India. SS acknowledges the receipt of PDF Fellowship from Cognitive Science Research Initiative Program of DST, India and SKS acknowledges RA fellowship from The Indian Council of Medical Research (ICMR), New Delhi, India. AK acknowledges the financial assistance from Department of Science and Technology, Govt of India support scheme DST/CSRI/PDF-63/2018 under Cognitive Science Research Initiative program DK acknowledges the Department of Science and Technology for financial assistance under SERB EMR Scheme (Ref. No.: EMR/2016/001756). UK acknowledges receipt of the SRF fellowship [ICMR sanction no.3/1/3/JRF-2014/HRD-100 (32508)]. Other authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Contributions
Smita Singh (SS), Rameshwar Nath Chaurasia (RNC) and Anup Singh (AS) helped in the clinical screening of patients, imaging analysis and clinical data collection. Umesh Kumar (UK), Sandeep Kumar Singh (SKS) and Payal Arya (PA) processed plasma samples, prepared the NMR samples and recorded the NMR spectra. Dinesh Kumar (DK) and Abhai Kumar (AK) analyzed Metabolomics data, prepared the Figures and drafted the manuscript.
Corresponding authors
Ethics declarations
None.
Conflict of interest
Authors declare that they have no conflict of interest
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 1301 kb)
Rights and permissions
About this article
Cite this article
Kumar, U., Kumar, A., Singh, S. et al. An elaborative NMR based plasma metabolomics study revealed metabolic derangements in patients with mild cognitive impairment: a study on north Indian population. Metab Brain Dis 36, 957–968 (2021). https://doi.org/10.1007/s11011-021-00700-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00700-z