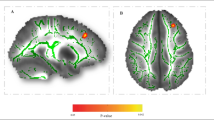
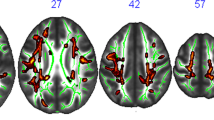
Abstract
Magnetic Resonance Spectroscopy provides measures of brain chemistry that are sensitive to cardiorespiratory fitness and body composition. The concentration of N-acetyl aspartic acid (NAA) is of interest because it is a marker of neuronal integrity. The ratio of NAA to creatine, a standard reference metabolite, has been shown to correlate with measures of both cardiorespiratory fitness and body composition. However, previous studies have explored these effects in isolation, making it impossible to know which of these highly correlated measures drive the correlations with NAA/Cr. As a result, the mechanisms underlying their association remain to be established. We therefore conducted a comprehensive study to investigate the relative contributions of cardiorespiratory fitness and percent body fat in predicting NAA/Cr. We demonstrate that NAA/Cr in white matter is correlated with percent body fat, and that this relationship largely subsumes the correlation of NAA/Cr with cardiorespiratory fitness. These results underscore the association of body composition with axonal integrity and suggests that this relationship drives the association of NAA/Cr with physical fitness in young adults.



Similar content being viewed by others
References
American College of Sports Medicine (2014) ACSMʼs guidelines for exercise testing and prescription, 9th edn. Wolters Kluwer/Lippincott, Williams, & Wilkins Health, Philadelphia
Bettcher BM et al (2013) Body mass and white matter integrity: the influence of vascular and inflammatory markers. PLoS One 8:UNSP e77741. https://doi.org/10.1371/journal.pone.0077741
Black JE, Isaacs KR, Anderson BJ, Alcantara AA, Greenough WT (1990) Learning causes synaptogenesis, whereas motor-activity causes angiogenesis, in cerebellar cortex of adult-rats. Proc Natl Acad Sci USA 87:5568–5572. https://doi.org/10.1073/pnas.87.14.5568
Chapman SB, Aslan S, Spence JS, Defina LF, Keebler MW, Didehbani N, Lu H (2013) Shorter term aerobic exercise improves brain, cognition, and cardiovascular fitness in aging. Front Aging Neurosci 5:75. https://doi.org/10.3389/fnagi.2013.00075
Charlton RA et al (2006) White matter damage on diffusion tensor imaging correlates with age-related cognitive decline. Neurology 66:217–222. https://doi.org/10.1212/01.wnl.0000194256.15247.83
Colcombe S, Kramer AF (2003) Fitness effects on the cognitive function of older adults: A meta-analytic study. Psychol Sci 14:125–130. https://doi.org/10.1111/1467-9280.t01-1-01430
Cook RL et al (2017) Relationship between obesity and cognitive function in young women: the food, mood and mind study, J Obes Artn 5923862. https://doi.org/10.1155/2017/5923862
Coplan JD et al (2014) Reduced hippocampal N-acetyl-aspartate (NAA) as a biomarker for overweight. Neuroimage-Clin 4:326–335. https://doi.org/10.1016/j.nicl.2013.12.014
Daugherty AM et al (2018) Multi-modal fitness and cognitive training to enhance fluid intelligence. Intelligence 66:32–43. https://doi.org/10.1016/j.intell.2017.11.001
Erickson KI et al (2012) Beyond vascularization: aerobic fitness is associated with N-acetylaspartate and working memory. Brain Behav 2:32–41. https://doi.org/10.1002/brb3.30
Firth J, Stubbs B, Vancampfort D, Schuch F, Lagopoulos J, Rosenbaum S, Ward PB (2018) Effect of aerobic exercise on hippocampal volume in humans: A systematic review and meta-analysis. Neuroimage 166:230–238. https://doi.org/10.1016/j.neuroimage.2017.11.007
Fitzpatrick S, Gilbert S, Serpell L (2013) Systematic review: are overweight and obese individuals impaired on behavioural tasks of executive functioning? Neuropsychol Rev 23:138–156. https://doi.org/10.1007/s11065-013-9224-7
Fuller NJ, Laskey MA, Elia M (1992) Assessment of the composition of major body regions by dual-energy X-ray absorptiometry (Dexa), with special reference to limb muscle mass. Clin Physiol 12:253–266. https://doi.org/10.1111/j.1475-097X.1992.tb00831.x
Gazdzinski S, Kornak J, Weiner MW, Meyerhoff DJ, Nat R (2008) Body mass index and magnetic resonance markers of brain integrity in adults. Ann Neurol 63:652–657. https://doi.org/10.1002/ana.21377
Gazdzinski S, Millin R, Kaiser LG, Durazzo TC, Mueller SG, Weiner MW, Meyerhoff DJ (2010) BMI and neuronal integrity in healthy, cognitively normal elderly: a proton magnetic resonance spectroscopy study. Obesity 18:743–748. https://doi.org/10.1038/oby.2009.325
Gonzales MM, Tarumi T, Eagan DE, Tanaka H, Vaghasia M, Haley AP (2012) Indirect effects of elevated body mass index on memory performance through altered cerebral metabolite concentrations. Psychosom Med 74:691–698. https://doi.org/10.1097/PSY.0b013e31825fflde
Gonzales MM et al (2013) Aerobic fitness and the brain: increased N-acetyl-aspartate and choline concentrations in endurance-trained middle-aged adults. Brain Topogr 26:126–134. https://doi.org/10.1007/s10548-012-0248-8
Gonzales MM et al (2014) Central adiposity and the functional magnetic resonance imaging response to cognitive challenge. Int J Obes 38:1193–1199. https://doi.org/10.1038/ijo.2014.5
Gustafson D, Rothenberg E, Blennow K, Steen B, Skoog I (2003) An 18-year follow-up of overweight and risk of Alzheimer disease. Arch Intern Med 163:1524–1528. https://doi.org/10.1001/archinte.163.13.1524
Haley AP, Gonzales MM, Tarumi T, Tanaka H (2013) Dyslipidemia links obesity to early cerebral neurochemical alterations. Obesity 21:2007–2013. https://doi.org/10.1002/oby.20332
Hayes SM et al (2015) Physical activity is positively associated with episodic memory in aging. J Int Neuropsychol Soc 21:780–790. https://doi.org/10.1017/S1355617715000910
Hillman CH, Erickson KI, Kramer AF (2008) Be smart, exercise your heart: exercise effects on brain and cognition. Nat Rev Neurosci 9:58–65. https://doi.org/10.1038/nrn2298
Hua K et al (2008) Tract probability maps in stereotaxic spaces: analyses of white matter anatomy and tract-specific quantification. Neuroimage 39:336–347. https://doi.org/10.1016/j.neuroimage.2007.07.053
Iadecola C (2013) The pathobiology of vascular dementia. Neuron 80:844–866. https://doi.org/10.1016/j.neuron.2013.10.008
Kantarci K et al (2013) MRI and MRS predictors of mild cognitive impairment in a population-based sample. Neurology 81:126–133. https://doi.org/10.1212/WNL.0b013e31829a3329
Kaur S, Birdsill AC, Steward K, Pasha E, Kruzliak P, Tanaka H, Haley AP (2017) Higher visceral fat is associated with lower cerebral N-acetyl-aspartate ratios in middle-aged adults. Metab Brain Dis 32:727–733. https://doi.org/10.1007/s11011-017-9961-z
Kim KW, Seo H, Kwak MS, Kim D (2017) Visceral obesity is associated with white matter hyperintensity and lacunar infarct. Int J Obes 41:683–688. https://doi.org/10.1038/ijo.2017.13
Kramer AF, Colcombe S (2018) Fitness effects on the cognitive function of older adults: a meta-analytic study-revisited. Perspect Psychol Sci 13:213–217. https://doi.org/10.1177/1745691617707316
Kullberg J et al (2009) Whole-body adipose tissue analysis: comparison of MRI, CT and dual energy X-ray absorptiometry. Br J Radiol 82:123–130. https://doi.org/10.1259/bjr/80083156
Kullmann S, Schweizer F, Veit R, Fritsche A, Preissl H (2015) Compromised white matter integrity in obesity. Obes Rev 16:273–281. https://doi.org/10.1111/obr.12248
Lampe L et al (2019) Visceral obesity relates to deep white matter hyperintensities via inflammation. Ann Neurol 85:194–203. https://doi.org/10.1002/ana.25396
Lancaster JL et al (2000) Automated Talairach Atlas labels for functional brain mapping. Hum Brain Mapp 10:120–131. https://doi.org/10.1002/1097-0193(200007)10:3<120::Aid-Hbm30>3.0.Co;2-8
Larsen RJ, Newman M, Nikolaidis A (2016) Reduction of variance in measurements of average metabolite concentration in anatomically-defined brain regions. J Magn Reson 272:73–81. https://doi.org/10.1016/j.jmr.2016.09.005
Mazess RB, Barden HS, Bisek JP, Hanson J (1990) Dual-energy X-Ray absorptiometry for total-body and regional bone-mineral and soft-tissue composition. Am J Clin Nutr 51:1106–1112
Meng XL, Rosenthal R, Rubin DB (1992) Comparing correlated correlation-coefficients. Psychol Bull 111:172–175. https://doi.org/10.1037//0033-2909.111.1.172
Miyamoto N, Maki T, Pham LD, Hayakawa K, Seo JH, Mandeville ET, Mandeville JB, Kim KW, Lo EH, Arai K (2013) Oxidative stress interferes with white matter renewal after prolonged cerebral hypoperfusion in mice. Stroke 44:3516–3521. https://doi.org/10.1161/Strokeaha.113.002813
Moffett JR, Ross B, Arun P, Madhavarao CN, AMA N (2007) N-acetylaspartate in the CNS: From neurodiagnostics to neurobiology. Prog Neurobiol 81:89–131. https://doi.org/10.1016/j.pneurobio.2006.12.003
Narayana PA (2005) Magnetic resonance spectroscopy in the monitoring of multiple sclerosis. J Neuroimaging 15:46s–57s. https://doi.org/10.1177/1051228405284200
Nikolaidis A, Baniqued PL, Kranz MB, Scavuzzo CJ, Barbey AK, Kramer AF, Larsen RJ (2017) Multivariate associations of fluid intelligence and NAA. Cereb Cortex 27:2607–2616. https://doi.org/10.1093/cercor/bhw070
Paul EJ et al (2016) Dissociable brain biomarkers of fluid intelligence. Neuroimage 137:201–211. https://doi.org/10.1016/j.neuroimage.2016.05.037
Pereira AC et al (2007) An in vivo correlate of exercise-induced neurogenesis in the adult dentate gyrus. Proc Natl Acad Sci U S A 104:5638–5643. https://doi.org/10.1073/pnas.0611721104
Prickett C, Brennan L, Stolwyk R (2015) Examining the relationship between obesity and cognitive function: A systematic literature review. Obes Res Clin Pract 9:93–113. https://doi.org/10.1016/j.orcp.2014.05.001
Provencher SW (1993) Estimation of metabolite concentrations from localized in-vivo proton Nmr-spectra. Magn Reson Med 30:672–679. https://doi.org/10.1002/mrm.1910300604
Rae CD (2014) A guide to the metabolic pathways and function of metabolites observed in human brain H-1 magnetic resonance spectra. Neurochem Res 39:1–36. https://doi.org/10.1007/s11064-013-1199-5
Ross AJ, Sachdev PS, Wen W, Valenzuela MJ, Brodaty H (2005) Cognitive correlates of H-1 MRS measures in the healthy elderly brain. Brain Res Bull 66:9–16. https://doi.org/10.1016/j.brainresbull.2005.01.015
Shepherd JA, Ng BK, Sommer MJ, Heymsfield SB (2017) Body composition by DXA. Bone 104:101–105. https://doi.org/10.1016/j.bone.2017.06.010
Simpson R, Devenyi GA, Jezzard P, Hennessy TJ, Near J (2017) Advanced processing and simulation of MRS data using the FID appliance (FID-A)an open source, MATLAB-based toolkit. Magn Reson Med 77:23–33. https://doi.org/10.1002/mrm.26091
Smith E, Hay P, Campbell L, Trollor JN (2011) A review of the association between obesity and cognitive function across the lifespan: implications for novel approaches to prevention and treatment. Obes Rev 12:740–755. https://doi.org/10.1111/j.1467-789X.2011.00920.x
Sripetchwandee J, Chattipakorn N, Chattipakorn SC (2018) Links between obesity-induced brain insulin resistance, brain mitochondrial dysfunction, and dementia. Front Endocrinol 9:ARTN 496. https://doi.org/10.3389/fendo.2018.00496
Stanek KM, Grieve SM, Brickman AM, Korgaonkar MS, Paul RH, Cohen RA, Gunstad JJ (2011) Obesity is associated with reduced white matter integrity in otherwise healthy adults. Obesity 19:500–504. https://doi.org/10.1038/oby.2010.312
Thomas S, Reading J, Shephard R J (1992) Revision of the Physical Activity Readiness Questionnaire (PAR-Q). Canadian Journal of Sport Sciences 17(4), 338–345
Tzourio-Mazoyer N et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15:273–289. https://doi.org/10.1006/nimg.2001.0978
Voss MW et al (2013) The influence of aerobic fitness on cerebral white matter integrity and cognitive function in older adults: results of a one-year exercise intervention. Hum Brain Mapp 34:2972–2985. https://doi.org/10.1002/hbm.22119
Williams VJ, Hayes JP, Forman DE, Salat DH, Sperling RA, Verfaellie M, Hayes SM (2017) Cardiorespiratory fitness is differentially associated with cortical thickness in young and older adults. Neuroimage 146:1084–1092. https://doi.org/10.1016/j.neuroimage.2016.10.033
Acknowledgements
The research is based upon work supported by the Office of the Director of National Intelligence (ODNI), Intelligence Advanced Research Projects Activity (IARPA), via Contract 2014-13121700004 to the University of Illinois at Urbana-Champaign (PI: Barbey). The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsements, either expressed or implied, of the ODNI, IARPA, or the U.S. Government. The U.S. Government is authorized to reproduce and distribute reprints for Governmental purposes notwithstanding any copyright annotation thereon.
We thank Nancy Dodge, Holly Tracy, Tracy Henigman, and Courtney Allen for assistance in performing the experiments.
Author information
Authors and Affiliations
Contributions
Ryan J. Larsen: Conceptualization, Methodology, Software, Formal analysis, Investigation, Resources, Data Curation, Writing-Original Draft, Writing – Review & Editing, Visualization, Supervision. Lauren B. Raine: Writing – Review & Editing Charles H. Hillman: Conceptualization, Writing – Review & Editing, Project administration, Funding acquisition Arthur F. Kramer: Conceptualization, Writing – Review & Editing, Project administration, Funding acquisition Neal J. Cohen: Conceptualization, Writing – Review & Editing, Project administration, Funding acquisition Aron K. Barbey: Conceptualization, Writing – Review & Editing, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest to report.
Informed consent
This study was performed in accordance with the Institutional Review Board at the University of Illinois, and informed consent was obtained from all study participants.
Data Sharing
Study data are available at the Illinois Data Bank, https://doi.org/10.13012/B2IDB-9371397_V1.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Larsen, R.J., Raine, L.B., Hillman, C.H. et al. Body mass and cardiorespiratory fitness are associated with altered brain metabolism. Metab Brain Dis 35, 999–1007 (2020). https://doi.org/10.1007/s11011-020-00560-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-020-00560-z