Abstract
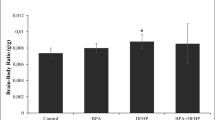
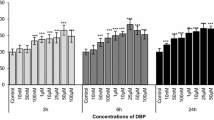
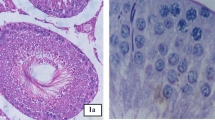
Due to its ability to cross blood brain barrier and placenta, dibutyl phthalate (di-n-butyl phthalate, DBP) is expected to cause severe side effects to the central nervous system of animals and humans. A little data is available about the potential DBP neurotoxicity; therefore, this work was designed to investigate the brain tissue injury induced by DBP exposure. Forty Wister albino rats were allocated randomly into 4 groups (10 rats each). Group 1 served as control and the rats administered with physiological saline (0.9% NaCl) orally for 12 weeks. Groups 2, 3 and 4 were orally treated with DPB (100, 250 and 500 mg/kg) respectively for 12 weeks. DBP-intoxicated rats showed a disturbance in the oxidative status in cerebral cortex, striatum and brainstem, as represented by the elevated oxidants [malondialdehyde (MDA), nitric oxide (NO), 8-hydroxy-2-deoxyguanosine (8-OHdG)] and the decreased antioxidant molecules [reduced glutathione (GSH), superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx) and glutathione reductase (GR)]. DBP also enhanced a pro-inflammatory state through increasing the release of tumor necrosis factor- α (TNF-α) and interleukin-1β (IL-1β). The increase of these cytokines was associated with the increase of pro-apoptotic proteins [Bcl-2 associated X protein (Bax) and caspase-3] and the decrease of the anti-apoptotic protein, B cell lymphoma 2 (Bcl-2). In addition, the levels of norepinephrine (NE), dopamine (DA) and acetylcholine esterase (AChE) activity were decreased. This was accompanied by the alterations in the major excitatory and inhibitory amino acids neurotransmitters levels. The present findings indicated that DBP could exert its neuronal damage through oxidative stress, DNA oxidation, neuroinflammation, activation of apoptotic proteins and altering the monoaminergic, cholinergic and amino acids transmission.






Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Aktan F (2004) iNOS-mediated nitric oxide production and its regulation. Life Sci 75:639–653. https://doi.org/10.1016/j.lfs.2003.10.042
Al Omairi NE, Radwan OK, Alzahrani YA, Kassab RB (2018) Neuroprotective efficiency of Mangifera indica leaves extract on cadmium-induced cortical damage in rats. Metab Brain Dis 33:1121–1130. https://doi.org/10.1007/s11011-018-0222-6
Al-Olayan EM, El-Khadragy MF, Abdel Moneim AE (2015) The protective properties of melatonin against aluminium-induced neuronal injury. Int J Exp Pathol 96:196–202. https://doi.org/10.1111/iep.12122
Al-Olayan EM, El-Khadragy MF, Omer SA, Shata MT, Kassab RB, Abdel Moneim AE (2016) The beneficial effect of cape gooseberry juice on carbon tetrachloride- induced neuronal damage. CNS Neurol Disord Drug Targets 15:344–350
Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O (2012) Oxidative stress and antioxidant defense. World Allergy Organ J 5:9–19. https://doi.org/10.1097/WOX.0b013e3182439613
Calafat AM, Brock JW, Silva MJ, Gray LE Jr, Reidy JA, Barr DB, Needham LL (2006) Urinary and amniotic fluid levels of phthalate monoesters in rats after the oral administration of di(2-ethylhexyl) phthalate and di-n-butyl phthalate. Toxicology 217:22–30. https://doi.org/10.1016/j.tox.2005.08.013
Carbone S, Szwarcfarb B, Ponzo O, Reynoso R, Cardoso N, Deguiz L, Moguilevsky JA, Scacchi P (2010) Impact of gestational and lactational phthalate exposure on hypothalamic content of amino acid neurotransmitters and FSH secretion in peripubertal male rats. Neurotoxicology 31:747–751. https://doi.org/10.1016/j.neuro.2010.06.006
Chighizola C, Meroni PL (2012) The role of environmental estrogens and autoimmunity. Autoimmun Rev 11:A493–A501. https://doi.org/10.1016/j.autrev.2011.11.027
Cook I (2008) Biomarkers in psychiatry: potentials, pitfalls, and pragmatics. Prim Psychiatry 15:54–59
de Mello Santos T, da Silveira LTR, Rinaldi JC, Scarano WR, Domeniconi RF (2017) Alterations in prostate morphogenesis in male rat offspring after maternal exposure to Di-n-butyl-phthalate (DBP). Reprod Toxicol 69:254–264. https://doi.org/10.1016/j.reprotox.2017.03.010
Elkhadragy MF, Kassab RB, Metwally DM, Almeer R, Abdel-Gaber R, al-Olayan EM, Essawy EA, Amin HK, Abdel Moneim AE (2018) Protective effects of Fragaria ananassa methanolic extract in a rat model of cadmium chloride-induced neurotoxicity. Biosci Rep. https://doi.org/10.1042/BSR2018086
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35:495–516. https://doi.org/10.1080/01926230701320337
Escudero-Lourdes C (2016) Toxicity mechanisms of arsenic that are shared with neurodegenerative diseases and cognitive impairment: role of oxidative stress and inflammatory responses. Neurotoxicology 53:223–235. https://doi.org/10.1016/j.neuro.2016.02.002
Factor VM, Kiss A, Woitach JT, Wirth PJ, Thorgeirsson SS (1998) Disruption of redox homeostasis in the transforming growth factor-alpha/c-myc transgenic mouse model of accelerated hepatocarcinogenesis. J Biol Chem 273:15846–15853
Faniband M, Lindh CH, Jonsson BA (2014) Human biological monitoring of suspected endocrine-disrupting compounds. Asian J Androl 16:5–16. https://doi.org/10.4103/1008-682X.122197
Franken C, Lambrechts N, Govarts E, Koppen G, den Hond E, Ooms D, Voorspoels S, Bruckers L, Loots I, Nelen V, Sioen I, Nawrot TS, Baeyens W, van Larebeke N, Schoeters G (2017) Phthalate-induced oxidative stress and association with asthma-related airway inflammation in adolescents. Int J Hyg Environ Health 220:468–477. https://doi.org/10.1016/j.ijheh.2017.01.006
Fujii M, Shinohara N, Lim A, Otake T, Kumagai K, Yanagisawa Y (2003) A study on emission of phthalate esters from plastic materials using a passive flux sampler. Atmos Environ 37(39–40):5495–5504
Gao M, Dong Y, Zhang Z, Song W, Qi Y (2017) Growth and antioxidant defense responses of wheat seedlings to di-n-butyl phthalate and di (2-ethylhexyl) phthalate stress. Chemosphere 172:418–428. https://doi.org/10.1016/j.chemosphere.2017.01.034
Giribabu N, Sainath SB, Sreenivasula Reddy P (2014) Prenatal di-n-butyl phthalate exposure alters reproductive functions at adulthood in male rats. Environ Toxicol 29:534–544. https://doi.org/10.1002/tox.21779
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138
Heinrikson RL, Meredith SC (1984) Amino acid analysis by reverse-phase high-performance liquid chromatography: precolumn derivatization with phenylisothiocyanate. Anal Biochem 136:65–74
Heudorf U, Mersch-Sundermann V, Angerer J (2007) Phthalates: toxicology and exposure. Int J Hyg Environ Health 210:623–634. https://doi.org/10.1016/j.ijheh.2007.07.011
Kassab RB, El-Hennamy RE (2017) The role of thymoquinone as a potent antioxidant in ameliorating the neurotoxic effect of sodium arsenate in female rat. Egyptian J Basic Appl Sci 4:160–167. https://doi.org/10.1016/j.ejbas.2017.07.002
Kavlock R et al (2006) NTP-CERHR expert panel update on the reproductive and developmental toxicity of di(2-ethylhexyl) phthalate. Reprod Toxicol 22:291–399
Kovacic P, Edwards C (2010) Integrated approach to the mechanisms of thyroid toxins: electron transfer, reactive oxygen species, oxidative stress, cell signaling, receptors, and antioxidants. J Recept Signal Transdut Res 30:133–142. https://doi.org/10.3109/10799891003702678
Lee E, Ahn MY, Kim HJ, Kim IY, Han SY, Kang TS, Hong JH, Park KL, Lee BM, Kim HS (2007) Effect of di(n-butyl) phthalate on testicular oxidative damage and antioxidant enzymes in hyperthyroid rats. Environ Toxicol 22:245–255. https://doi.org/10.1002/tox.20259
Li Y, Zhuang M, Li T, Shi N (2009) Neurobehavioral toxicity study of dibutyl phthalate on rats following in utero and lactational exposure. J Appl Toxicol 29:603–611. https://doi.org/10.1002/jat.1447
Li XJ, Jiang L, Chen L, Chen HS, Li X (2013) Neurotoxicity of dibutyl phthalate in brain development following perinatal exposure: a study in rats. Environ Toxicol Pharmacol 36:392–402. https://doi.org/10.1016/j.etap.2013.05.001
Lien Y et al (2015) Prenatal exposure to phthalate esters and behavioral syndromes in children at 8 years of age: Taiwan maternal and infant ohort study. Environ Health Perspect 123:95–100
Liu PS, Tseng FW, Liu JH (2009) Comparative suppression of phthalate monoesters and phthalate diesters on calcium signalling coupled to nicotinic acetylcholine receptors. J Toxicol Sci 34:255–263
Lodovici M, Casalini C, Briani C, Dolara P (1997) Oxidative liver DNA damage in rats treated with pesticide mixtures. Toxicology 117:55–60
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lu KY, Tseng FW, Wu CJ, Liu PS (2004) Suppression by phthalates of the calcium signaling of human nicotinic acetylcholine receptors in human neuroblastoma SH-SY5Y cells. Toxicology 200:113–121. https://doi.org/10.1016/j.tox.2004.03.018
Mahaboob Basha P, Radha MJ (2017) Gestational di-n-butyl phthalate exposure induced developmental and teratogenic anomalies in rats: a multigenerational assessment. Environ Sci Pollut Res Int 24:4537–4551. https://doi.org/10.1007/s11356-016-8196-6
Mao G, Zhou Z, Chen Y, Wang W, Wu X, Feng W, Cobbina SJ, Huang J, Zhang Z, Xu H, Yang L, Wu X (2016) Neurological toxicity of individual and mixtures of low dose arsenic, mono and di (n-butyl) phthalates on sub-chronic exposure to mice. Biol Trace Elem Res 170:183–193. https://doi.org/10.1007/s12011-015-0457-6
Marc DT, Ailts JW, Campeau DC, Bull MJ, Olson KL (2011) Neurotransmitters excreted in the urine as biomarkers of nervous system activity: validity and clinical applicability. Neurosci Biobehav Rev 35:635–644. https://doi.org/10.1016/j.neubiorev.2010.07.007
Mariana M, Feiteiro J, Verde I, Cairrao E (2016) The effects of phthalates in the cardiovascular and reproductive systems: a review. Environ Int 94:758–776. https://doi.org/10.1016/j.envint.2016.07.004
Min A, Liu F, Yang X, Chen M (2014) Benzyl butyl phthalate exposure impairs learning and memory and attenuates neurotransmission and CREB phosphorylation in mice. Food Chem Toxicol 71:81–89. https://doi.org/10.1016/j.fct.2014.05.021
Nishikimi M, Appaji N, Yagi K (1972) The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem Biophys Res Commun 46:849–854
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Pagel P, Blome J, Wolf HU (2000) High-performance liquid chromatographic separation and measurement of various biogenic compounds possibly involved in the pathomechanism of Parkinson's disease. J Chromatogr B Biomed Sci Appl 746:297–304
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Peng L (2015) Mice brain tissue injury induced by diisononyl phthalate exposure and the protective application of vitamin E. J Biochem Mol Toxicol 29:311–320. https://doi.org/10.1002/jbt.21700
Rocha BA, Asimakopoulos AG, Barbosa F Jr, Kannan K (2017) Urinary concentrations of 25 phthalate metabolites in Brazilian children and their association with oxidative DNA damage. Sci Total Environ 586:152–162. https://doi.org/10.1016/j.scitotenv.2017.01.193
Schettler T (2006) Human exposure to phthalates via consumer products. Int J Androl 29:134–139; discussion 181-135. https://doi.org/10.1111/j.1365-2605.2005.00567.x
Shaw CA, Taylor EL, Megson IL, Rossi AG (2005) Nitric oxide and the resolution of inflammation: implications for atherosclerosis. Mem Inst Oswaldo Cruz 100 Suppl 1:67–71. https://doi.org/10.1590/S0074-02762005000900012
Shono T, Taguchi T (2014) Short-time exposure to mono-n-butyl phthalate (MBP)-induced oxidative stress associated with DNA damage and the atrophy of the testis in pubertal rats. Environ Sci Pollut Res Int 21:3187–3190. https://doi.org/10.1007/s11356-013-2332-3
Tabb MM, Blumberg B (2006) New modes of action for endocrine-disrupting chemicals. Mol Endocrinol 20:475–482. https://doi.org/10.1210/me.2004-0513
Teixeira D, Marques C, Pestana D, Faria A, Norberto S, Calhau C, Monteiro R (2016) Effects of xenoestrogens in human M1 and M2 macrophage migration, cytokine release, and estrogen-related signaling pathways. Environ Toxicol 31:1496–1509. https://doi.org/10.1002/tox.22154
Uttara B, Singh AV, Zamboni P, Mahajan RT (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7:65–74. https://doi.org/10.2174/157015909787602823
Wang IJ, Karmaus WJ (2017) Oxidative stress-related genetic variants may modify associations of phthalate exposures with asthma. Int J Environ Res Public Health 14. https://doi.org/10.3390/ijerph14020162
Wojtowicz AK, Szychowski KA, Wnuk A, Kajta M (2017) Dibutyl phthalate (DBP)-induced apoptosis and neurotoxicity are mediated via the aryl hydrocarbon receptor (AhR) but not by estrogen receptor alpha (ERalpha), estrogen receptor beta (ERbeta), or peroxisome proliferator-activated receptor gamma (PPARgamma) in mouse cortical neurons. Neurotox Res 31:77–89. https://doi.org/10.1007/s12640-016-9665-x
Yan B, Guo J, Liu X, Li J, Yang X, Ma P, Wu Y (2016) Oxidative stress mediates dibutyl phthalateinduced anxiety-like behavior in Kunming mice. Environ Toxicol Pharmacol 45:45–51. https://doi.org/10.1016/j.etap.2016.05.013
Yavasoglu NU, Koksal C, Dagdeviren M, Aktug H, Yavasoglu A (2014) Induction of oxidative stress and histological changes in liver by subacute doses of butyl cyclohexyl phthalate. Environ Toxicol 29:345–353. https://doi.org/10.1002/tox.21813
Zhou D, Wang H, Zhang J, Gao X, Zhao W, Zheng Y (2010) Di-n-butyl phthalate (DBP) exposure induces oxidative damage in testes of adult rats. Syst Biol Reprod Med 56:413–419. https://doi.org/10.3109/19396368.2010.509902
Zhou D, Wang H, Zhang J (2011) Di-n-butyl phthalate (DBP) exposure induces oxidative stress in epididymis of adult rats. Toxicol Ind Health 27:65–71. https://doi.org/10.1177/0748233710381895
Zhu YP, Chen L, Wang XJ, Jiang QH, Bei XY, Sun WL, Xia SJ, Jiang JT (2017) Maternal exposure to di-n-butyl phthalate (DBP) induces renal fibrosis in adult rat offspring. Oncotarget 8(19):31101–31111. https://doi.org/10.18632/oncotarget.16088
Zuo HX et al (2014) Di-(n-butyl)-phthalate-induced oxidative stress and depression-like behavior in mice with or without ovalbumin immunization. Biomed Environ Sci 27:268–280. https://doi.org/10.3967/bes2014.001
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kassab, R.B., Lokman, M.S. & Essawy, E.A. Neurochemical alterations following the exposure to di-n-butyl phthalate in rats. Metab Brain Dis 34, 235–244 (2019). https://doi.org/10.1007/s11011-018-0341-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-018-0341-0